Abstract
Background and Aim
Nitrogen (N) and carbon (C) isotopic signatures (δ15N and δ13C) serve as powerful tools for understanding temporal changes in ecosystem processes, but how these signatures change across boreal forest chronosequences is poorly understood.
Methods
The δ15N, δ13C, and C/N ratio of foliage of eight dominant plant species, including trees, understory shrubs, and a moss, as well as humus, were examined across a 361 years fire-driven chronosequence in boreal forest in northern Sweden.
Results
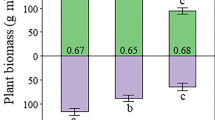
The δ13C and C/N ratio of plants and humus increased along the chronosequence, suggesting increasing plant stress through N limitation. Despite increasing biological N fixation by cyanobacteria associated with feather mosses, δ15N showed an overall decline, and δ15N of the feather moss and associated vascular plants diverged over time from that of atmospheric N2.
Conclusions
Across this chronosequence the N fixed by cyanobacteria is unlikely to be used by mosses and vascular plants without first undergoing mineralization and mycorrhizal transport, which would cause a change in δ15N signature due to isotopic fractionation. The decreasing trend of δ15N suggests that as the chronosequence proceeds, the plants may become more dependent on N transferred from mycorrhizal fungi or from N deposition.




Similar content being viewed by others
References
Albert CH, Thuiller W, Yoccoz NG, Soudant A, Boucher F, Saccone P, Lavorel S (2010) Intraspecific functional variability: extent, structure and sources of variation. J Ecol 98:604–613
Bates JW (2009) Mineral nutrition and substratum ecology. In: Goffnet B, Shaw AJ (eds) Bryophyte biology. Cambridge University Press, Cambridge, pp 299–356
Bisbee K, Gower S, Norman J, Nordheim E (2001) Environmental controls on ground cover species composition and productivity in a boreal black spruce forest. Oecologia 129:261–270
Bitterlich W (1984) The relascope idea: relative measurements in forestry. Commonwealth Agricultural Bureau, Slough
Brenner DL, Amundson R, Baisden WT, Kendall C, Harden J (2001) Soil N and 15N variation with time in a California annual grassland ecosystem. Geochim Cosmochim Acta 65:4171–4186
Brooks JR, Flanagan LB, Buchmann N, Ehleringer JR (1997) Carbon isotope composition of boreal plants: functional grouping of life forms. Oecologia 110:301–311
Chapin FS, Walker LR, Fastie CL, Sharman LC (1994) Mechanisms of primary succession following deglaciation at Glacier Bay, Alaska. Ecol Monogr 64:149–175
Cook GD (2001) Effects of frequent fires and grazing on stable nitrogen isotope ratios of vegetation in northern Australia. Austral Ecol 26:630–636
Craine JM, Elmore AJ, Aidar MPM, Bustamante M, Dawson TE, Hobbie EA, Kahmen A, Mack MC, McLauchlan KK, Michelsen A, Nardoto GB, Pardo LH, Penuelas J, Reich PB, Schuur EAG, Stock WD, Templer PH, Virginia RA, Welker JM, Wright IJ (2009) Global patterns of foliar nitrogen isotopes and their relationships with climate, mycorrhizal fungi, foliar nutrient concentrations, and nitrogen availability. New Phytol 183:980–992
Crews T, Kurina L, Vitousek P (2001) Organic matter and nitrogen accumulation and nitrogen fixation during early ecosystem development in Hawaii. Biogeochemistry 52:259–279
Crutsinger GM, Sanders NJ, Albrectsen BR, Abreu IN, Wardle DA (2008) Ecosystem retrogression leads to increased insect abundance and herbivory across an island chronosequence. Funct Ecol 22:816–823
DeLuca TH, Nilsson MC, Zackrisson O (2002a) Nitrogen mineralization and phenol accumulation along a fire chronosequence in northern Sweden. Oecologia 133:206–214
DeLuca TH, Zackrisson O, Nilsson MC, Sellstedt A (2002b) Quantifying nitrogen-fixation in feather moss carpets of boreal forests. Nature 419:917–920
DeLuca T, Zackrisson O, Gentili F, Sellstedt A, Nilsson M-C (2007) Ecosystem controls on nitrogen fixation in boreal feather moss communities. Oecologia 152:121–130
DeLuca TH, Zackrisson O, Gundale MJ, Nilsson MC (2008) Ecosystem feedbacks and nitrogen fixation in boreal forests. Science 320:1181–1181
Farquhar G, Richards R (1984) Isotopic composition of plant carbon correlates with water-use efficiency of wheat genotypes. Aust J Plant Physiol 11:539–552
Farquhar GD, Ehleringer JR, Hubick KT (1989) Carbon isotope discrimination and photosynthesis. Annu Rev Plant Physiol Plant Mol Biol 40:503–537
Grogan P, Burns TD, Chapin Iii FS (2000) Fire effects on ecosystem nitrogen cycling in a Californian bishop pine forest. Oecologia 122:537–544
Gundale MJ, Gustafsson H, Nilsson MC (2009) The sensitivity of nitrogen fixation by a feathermoss-cyanobacteria association to litter and moisture variability in young and old boreal forests. Can J For Res 39:2542–2549
Gundale MJ, Hyodo F, Nilsson MC, Wardle DA (2012) Nitrogen niches revealed through species and functional group removal in a boreal shrub community. Ecology:(in press)
Hobbie EA, Colpaert JV (2003) Nitrogen availability and colonization by mycorrhizal fungi correlate with nitrogen isotope patterns in plants. New Phytol 157:115–126
Hobbie EA, Macko SA, Shugart HH (1998) Patterns in N dynamics and N isotopes during primary succession in Glacier Bay, Alaska. Chem Geol 152:3–11
Hobbie EA, Macko SA, Shugart HH (1999) Insights into nitrogen and carbon dynamics of ectomycorrhizal and saprotrophic fungi from isotopic evidence. Oecologia 118:353–360
Hobbie EA, Macko SA, Williams M (2000) Correlations between foliar d15N and nitrogen concentrations may indicate plant-mycorrhizal interactions. Oecologia 122:273–283
Hobbie EA, Jumpponen A, Trappe J (2005) Foliar and fungal 15N: 14N ratios reflect development of mycorrhizae and nitrogen supply during primary succession: testing analytical models. Oecologia 146:258–268
Högberg P (1997) 15N natural abundance in soil-plant systems. New Phytol 137:179–203
Houle D, Bilodeau Gauthier S, Paquet S, Planas D, Warren A (2006) Identification of two genera of N2-fixing cyanobacteria growing on three feather moss species in boreal forests of Quebec, Canada. Can J Bot 84:1025–1029
Houlton BZ, Bai E (2009) Imprint of denitrifying bacteria on the global terrestrial biosphere. Proc Natl Acad Sci USA 106:21713–21716
Houlton BZ, Sigman DM, Schuur EAG, Hedin LO (2007) A climate-driven switch in plant nitrogen acquisition within tropical forest communities. Proc Natl Acad Sci USA 104:8902–8906
Hubick K, Farquhar G, Shorter R (1986) Correlation between water-use efficiency and carbon isotope discrimination in diverse peanut (Arachis) germplasm. Aust J Plant Physiol 13:803–816
Hultine KR, Marshall JD (2000) Altitude trends in conifer leaf morphology and stable carbon isotope composition. Oecologia 123:32–40
Hyodo F, Wardle DA (2009) Effect of ecosystem retrogression on stable nitrogen and carbon isotopes of plants, soils and consumer organisms in boreal forest islands. Rapid Commun Mass Spectrom 23:1892–1898
Kielland K (1994) Amino acid absorption by arctic plants: implications for plant nutrition and nitrogen cycling. Ecology 75:2373–2383
Kohzu A, Tateishi T, Yamada A, Koba K, Wada E (2000) Nitrogen isotope fractionation during nitrogen transport from ectomycorrhizal fungi, Suiillus granulatus, to the host plant, Pinus densiflora. Soil Sci Plant Nutr 46:733–739
Krab E, Cornelissen J, Lang S, van Logtestijn R (2008) Amino acid uptake among wide-ranging moss species may contribute to their strong position in higher-latitude ecosystems. Plant Soil 304:199–208
Lagerström A, Nilsson MC, Zackrisson O, Wardle DA (2007) Ecosystem input of nitrogen through biological fixation in feather mosses during ecosystem retrogression. Funct Ecol 21:1027–1033
Larsen CPS (1997) Spatial and temporal variations in boreal forest fire frequency in northern Alberta. J Biogeogr 24:663–673
Lindahl BD, Ihrmark K, Boberg J, Trumbore SE, Hogberg P, Stenlid J, Finlay RD (2007) Spatial separation of litter decomposition and mycorrhizal nitrogen uptake in a boreal forest. New Phytol 173:611–620
Markham JH (2009) Variation in moss-associated nitrogen fixation in boreal forest stands. Oecologia 161:353–359
Martinelli LA, Piccolo MC, Townsend AR, Vitousek PM, Cuevas E, McDowell W, Robertson GP, Santos OC, Treseder K (1999) Nitrogen stable isotopic composition of leaves and soil: tropical versus temperate forests. Biogeochemistry 46:45–65
Matzek V, Vitousek P (2003) Nitrogen fixation in bryophytes, lichens, and decaying wood along a soil-age gradient in Hawaiian montane rain forest. Biotropica 35:12–19
McKane RB, Johnson LC, Shaver GR, Nadelhoffer KJ, Rastetter EB, Fry B, Giblin AE, Kielland K, Kwiatkowski BL, Laundre JA, Murray G (2002) Resource-based niches provide a basis for plant species diversity and dominance in arctic tundra. Nature 415:68–71
Menge DNL, Hedin LO (2009) Nitrogen fixation in different biogeochemical niches along a 120000-year chronosequence in New Zealand. Ecology 90:2190–2201
Menge DNL, Troy Baisden W, Richardson SJ, Peltzer DA, Barbour MM (2011) Declining foliar and litter δ15N diverge from soil, epiphyte and input δ15N along a 120 000 years temperate rainforest chronosequence. New Phytol 190:941–952
Michelsen A, Quarmby C, Sleep D, Jonasson S (1998) Vascular plant 15N natural abundance in heath and forest tundra ecosystems is closely correlated with presence and type of mycorrhizal fungi in roots. Oecologia 115:406–418
Nadelhoffer K, Fry B (1994) Nitrogen isotope studies in forest ecosystems. In: Lajtha K, Michener RH (eds) Stable isotopes in ecology and environmental science. Blackwell Scientific Publications, Oxford, pp 22–44
Nadelhoffer K, Shaver G, Fry B, Giblin A, Johnson L, McKane R (1996) 15N natural abundances and N use by tundra plants. Oecologia 107:386–394
Näsholm T, Kielland K, Ganeteg U (2009) Uptake of organic nitrogen by plants. New Phytol 182:31–48
Nilsson MC, Wardle DA (2005) Understory vegetation as a forest ecosystem driver: evidence from the northern Swedish boreal forest. Front Ecol Environ 3:421–428
Robinson D (2001) δ15N as an integrator of the nitrogen cycle. Trends Ecol Evol 16:153–162
Selmants PC, Hart SC (2008) Substrate age and tree islands influence carbon and nitrogen dynamics across a retrogressive semiarid chronosequence. Global Biogeochem Cycles 22:GB1021
Shearer G, Kohl DH (1989) Estimates of N2 fixation in ecosystems: the need for and basis of the 15N natural abundance method. In: Rundel PW, Ehleringer JR, Nagy KA (eds) Stable isotopes in ecological research. Springer, New York, pp 342–374
Skre O, Oechel WC (1979) Moss production in a black spruce Picea mariana forest with permafrost near Fairbanks, Alaska, as compared with two permafrost-free stands. Holarct Ecol 2:249–254
Takebayashi Y, Koba K, Sasaki Y, Fang YT, Yoh M (2010) The natural abundance of 15N in plant and soil-available N indicates a shift of main plant N resources to NO -3 from NH +4 along the N leaching gradient. Rapid Commun Mass Spectrom 24:1001–1008
Vitousek PM (2004) Nutrient cycling and limitation: Hawai'i as a model system. Princeton University Press, Princeton
Vitousek PM, Howarth RW (1991) Nitrogen limitation on land and in the sea—how can it occur? Biogeochemistry 13:87–115
Vitousek PM, Shearer G, Kohl DH (1989) Foliar 15N natural abundance in Hawaiian rainforest: patterns and possible mechanisms. Oecologia 78:383–388
Vitousek PM, Field CB, Matson PA (1990) Variation in foliar δ13C in Hawaiian Metrosideros polymorpha—a case of internal resistance. Oecologia 84:362–370
Wallander H, Nylund J-E (1991) Effects of excess nitrogen on carbohydrate concentration and mycorrhizal development of Pinus sylvestris L. seedlings. New Phytol 119:405–411
Wardle DA, Zackrisson O, Hornberg G, Gallet C (1997) The influence of island area on ecosystem properties. Science 277:1296–1299
Wardle DA, Jonsson M, Bansal S, Bardgett RD, Gundale MJ, Metcalfe DB (2012) Linking vegetation change, carbon sequestration and biodiversity: insights from island ecosystems in a long-term natural experiment. J Ecol 100:16–30
Zackrisson O (1977) Influence of forest fires on north Swedish boreal forest. Oikos 29:22–32
Zackrisson O, DeLuca TH, Nilsson MC, Sellstedt A, Berglund LM (2004) Nitrogen fixation increases with successional age in boreal forests. Ecology 85:3327–3334
Acknowledgements
We thank Morgan Karlsson for sample collection in this study. We also thank Keisuke Koba and two anonymous reviewers for valuable comments on an earlier draft of this manuscript. This study was supported by grants from Vetenskapsrådet to DW, FORMAS to MCN, and the Research Institute for Humanity and Nature (Japan) and JSPS, as well as by Special Coordination funds for Promoting Sciences and Technology from the MEXT to FH. FH was supported as a postdoctoral researcher by the Japan Society of the Promotion of Science Fellowships for Research Abroad.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Benjamin L. Turner.
Rights and permissions
About this article
Cite this article
Hyodo, F., Kusaka, S., Wardle, D.A. et al. Changes in stable nitrogen and carbon isotope ratios of plants and soil across a boreal forest fire chronosequence. Plant Soil 364, 315–323 (2013). https://doi.org/10.1007/s11104-012-1339-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-012-1339-8



