Abstract
Background and Aims
Forest trees directly contribute to carbon cycling in forest soils through the turnover of their fine roots. In this study we aimed to calculate root turnover rates of common European forest tree species and to compare them with most frequently published values.
Methods
We compiled available European data and applied various turnover rate calculation methods to the resulting database. We used Decision Matrix and Maximum-Minimum formula as suggested in the literature.
Results
Mean turnover rates obtained by the combination of sequential coring and Decision Matrix were 0.86 yr−1 for Fagus sylvatica and 0.88 yr−1 for Picea abies when maximum biomass data were used for the calculation, and 1.11 yr−1 for both species when mean biomass data were used. Using mean biomass rather than maximum resulted in about 30 % higher values of root turnover. Using the Decision Matrix to calculate turnover rate doubled the rates when compared to the Maximum-Minimum formula. The Decision Matrix, however, makes use of more input information than the Maximum-Minimum formula.
Conclusions
We propose that calculations using the Decision Matrix with mean biomass give the most reliable estimates of root turnover rates in European forests and should preferentially be used in models and C reporting.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Turnover of tree fine roots is one of the major carbon (C) pathways in forests. The cause of the large C flux through this biomass pool is the rather limited lifespan of tree roots less than 2 mm in diameter. Given the estimated size of the C flux associated with the limited lifespan (synonyms: ‘longevity’ or ‘turnover time’, inverse of ‘turnover rate’) of fine roots, thought to reach 0.5 to 3 t C ha−1 yr−1 in steady-state forest ecosystems (Gill and Jackson 2000; Brunner and Godbold 2007), we clearly need to have a good understanding of the turnover rate at which fine roots die and contribute to soil C pools. Indeed, in the light of ongoing and projected climate change and the implementation of C reporting in many countries, belowground C dynamics have to be taken into account. Given the role of scientists in this debate, it is down to those who study root dynamics to provide the knowledge basis that permits modellers and C reporters to utilise the most realistic turnover values. Currently, root turnover rates are commonly utilised to parameterise biogeochemical models, which require fine root turnover rate data input e.g. Biome-BGC, LPJ, or LPJ-GUESS (e.g. Pietsch et al. 2005; Sitch et al. 2003; Smith et al. 2001). The turnover rates as input can be derived from published scientific literature, based on supposed relationships between leaf lifespan and fine root lifespan (i.e. lower turnover rates for evergreen species with long-lived leaves) with values between 0.18 and 1.02 yr−1 (Pietsch et al. 2005; Cienciala and Tatarinov 2006; Tatarinov and Cienciala 2006) or simplified to just one value 0.7 yr−1 as suggested recently by Hickler et al. (2008). Using the most appropriate turnover rates will improve the capacity of these models to assess the change in belowground C pools in forests.
Fine root turnover rate is dependent on the fine root biomass and the annual production of fine roots, but also on the various methods and calculations (e.g. Jourdan et al. 2008; Gaul et al. 2009; Finer et al. 2011; Yuan and Chen 2010). However, there is quite some uncertainty regarding which fine root turnover rates would be most suitable for end users. This is illustrated by the ongoing debate among scientists about how the turnover rate of the fine roots can be estimated best and which method is the most suitable (e.g. Strand et al. 2008; Trumbore and Gaudinski 2003; Majdi et al. 2005; Jourdan et al. 2008). Starting from the most recent developments, stable C-isotopes and radiocarbon (13C, 14C) may be used to estimate root carbon longevity, either by using labelling techniques or natural abundances in the atmosphere (e.g. Matamala et al. 2003; Gaudinski et al. 2001, 2010; Endrulat et al. 2010). A more widely used method to estimate the lifespan of fine roots is the use of minirhizotrons (e.g. Johnson et al. 2001; Majdi and Andersson 2005). This technique allows for a direct observation of individual roots and their development. Both methods suffer from several drawbacks, the main weakness of isotopic analysis for root age determination is the uncertain age of organic compounds used to construct fine roots (Sah et al. 2011). Meanwhile, minirhizotron studies are not able to determine the exact time of root death. In addition, the installation of the minirhizotron tubes can change water and temperature regimes as well as soil matrix resistance to root penetration. Moreover, fine root growth is often stimulated by the conditions along the minirhizotron tube. Unsurprisingly, direct comparisons of these two methods result in a discrepancy in root longevity estimates (Tierney and Fahey 2002; Strand et al. 2008; Gaul et al. 2009), sometimes explained by different fractions of fine roots under observation, i.e. the short-lived and the long-lived fine roots, likely to be recorded by these two methods (Gaudinski et al. 2010).
Alternatively, instead of direct observations of individual root longevity, the mean lifespan can be calculated by dividing the ‘pool’ (biomass) by its ‘input’ (annual production). Because the turnover rate is the inverse of lifespan, it can be calculated by dividing the ‘annual production’ by the ‘belowground standing crop’ (=biomass) (Gill and Jackson 2000). There are several methods used to obtain estimates of annual fine root production. A widely used method to directly measure the production of fine roots is the use of ingrowth cores (e.g. Persson 1980a, 1980b; Vogt and Persson 1991). This method measures the amount of fine roots which grow into a defined volume of root-free soil over a defined period of time. The advantage of this method is its relative ease and speed of application when estimating root production (Vogt and Persson 1991). More recently, root nets were applied instead of ingrowth cores to minimise soil disturbance during the installation (Hirano et al. 2009; Lukac and Godbold 2010). An alternative method to indirectly measure the production of fine roots is the sequential coring technique (e.g. Stober et al. 2000; Ostonen et al. 2005). Here, several series of soil cores are sampled at defined intervals over a period of at least 1 year. Fine roots are extracted from the soil cores and the differences of the dry mass of living (biomass) and dead (necromass) fine roots between two time points recorded. Taking advantage of data generated by sequential coring, several methods exist to calculate the production from the change of the fine-root biomass and necromass data. The production can be calculated by the ‘Maximum-Minimum’ formula (McClaugherty et al. 1982), by the ‘Decision Matrix’ formula (Fairley and Alexander 1985), or by the ‘Compartment Flow’ formula (Santantonio and Grace 1987). Whereas the ‘Maximum-Minimum’ formula uses only biomass data, the other two methods require both biomass and necromass data. The ‘Compartment Flow’ formula further requires decomposition data of fine root litter (e.g. Silver et al. 2005, Osawa and Aizawa 2012). Thus, the values of fine root turnover rates can vary not only due to measurement methods but also due to calculation methods applied (e.g. Publicover and Vogt 1993; Vogt et al. 1998; Strand et al. 2008). A true comparison of the various turnover rates may only be possible by using observations from same sites where various methods were applied (e.g. Haynes and Gower 1995; Ostonen et al. 2005; Hendricks et al. 2006; Gaul et al. 2009). As for the popularity of different measurement methods, many more estimates of root turnover rates are available from sequential coring and ingrowth cores than from the minirhizotron method (Finer et al. 2011).
We took advantage of the European COST network FP0803 “Belowground carbon turnover in European forests” bringing together root researchers from 30 European countries, to investigate on the sources of variation in turnover estimates available in the literature. In particular, as our group covered most of the European research groups that have worked on fine root turnover in the last decades, we were able to reunite/mine detailed datasets needed to evaluate the effect of calculation methods on fine root turnover rates in the European context. This implies that the implications of our work are restricted to European tree species and growth conditions. Our objectives were 1) to evaluate the pure effect of calculation methods on mean turnover rates of European forests and their ranges; 2) to evaluate how other factors such as soil stratification contribute to the calculated/perceived variation in turnover rates of European forests, and 3) to propose turnover rates and ranges for end users for the most common European forests.
Materials and methods
Data origin
Our study was carried out on data of fine root biomass and necromass of European forest tree species, extracted from published studies, found through regular literature research in library databases or supplied by members of our COST network. A large proportion of the data originates from doctoral theses due to the availability of raw data in this type of publication. We only included datasets where data collection was carried out for at least one full year. Fine root production was measured either directly by the use of the ingrowth core method or indirectly by the use of the sequential coring method (see Ostonen et al. 2005). Fine root biomass was defined as the amount of living fine roots occurring in the soil at any given time. Sequential coring was used to establish fine root biomass in most studies, apart from the case of the ingrowth core method where biomass usually was estimated from a single coring. We did not consider data originating from minirhizotron studies as these are reviewed elsewhere (Børja et al. in preparation). Finally, the dataset created for this study included 17 studies with 31 datasets for sequential coring and 7 for ingrowth core studies. The most abundant data sets obtained by sequential coring were available for Fagus sylvatica and Picea abies with 13 and 11 data sets, respectively (Table 3). Data sets of other tree species, e.g. Pinus sylvestris, Populus spp., and Quercus spp., were present only in three or fewer data sets. More than 80 % of the data were from forests with adult trees (‘steady state’ conditions). Data sets originating from ingrowth cores were available only for F. sylvatica, P. abies, and P. sylvestris, and with only two to three data sets per tree species (Table 4).
Calculations of fine-root production
Fine root production was calculated either with the ‘Maximum-Minimum’ formula or the ‘Decision Matrix’. The ‘Compartment Flow’ method was not applied because decomposition data of root litter were not sufficiently available. As a pre-requisite of annual fine root production calculation, a single sampling campaign must have lasted at least 12 months. Studies of less than 12 months (e.g. one vegetation period) or not of required level of detail were not considered (e.g. Konôpka et al. 2005; Konôpka 2009; López et al. 2001). At least two measurements from the same month in two consecutive years are the minimum requirement for the calculation of root production.
The Maximum-Minimum (MM) formula calculates the annual fine-root production (Pa) by subtracting the lowest biomass (Bmin) from the highest biomass value (Bmax) irrespectively of other biomass values recorded during a full year (McClaugherty et al. 1982). Necromass data are not required for this method:
The Decision Matrix (DM) calculates the annual fine-root production (Pa) by summing all calculated productions (P) between each pair of consecutive sampling dates throughout a full year:
The production (P) between two sampling dates is calculated either by adding the differences in biomass (∆B) and necromass (∆N), by adding only the differences in biomass (∆B), or by equalling P to zero (Fairley and Alexander 1985). The conditions with which of the P formulas to be used are as follows:
The Decision Matrix used as the basis for calculations is shown in Table 1. To calculate the annual production, all production values from interim periods are summed up from the start of sequential coring until the same time point in the following year (see also Table 2a, b). In the present study, all differences in biomass and necromass were taken into account during the calculation, assuming that the living and dead pool are continuously changing. However, some authors suggest summing up only the statistically significant differences (e.g. Stober et al. 2000). We propose that accounting for all differences between root biomass in two sampling dates constitutes a better approach. The size (and therefore the significance) of the difference is clearly dependent on the duration of the interim period, as well as on the season. Including significantly different observations would skew the data coverage towards long-gap observations only.
Calculations of fine-root turnover rates
The turnover rate TBmax of fine roots was calculated by dividing the annual fine root production (Pa) by the highest biomass value (maximum biomass Bmax) according to Gill and Jackson (2000) (compare also Table 2c):
As an alternative, the turnover rate TBmean was calculated by dividing the annual fine root production (Pa) by the mean biomass (Bmean) according to McClaugherty et al. (1982) (compare also Table 2c):
Assessment of other factors generating variation in fine-root turnover rates
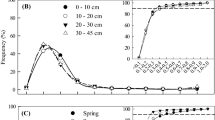
Utilizing the raw datasets in our database, we analysed several other factors for their influence of fine root turnover, based on subsamples of the database for sequential coring only. These factors included 1) soil stratification, 2) soil depth, 3) root diameter, 4) observation length, 5) start of observation period and 6) number of samplings per year. For the soil stratification approach we used 13 sites with detailed root data for the various soil layers. Briefly, for the layer-per-layer approach we computed fine root production per layer and summed this as a fine root production for the entire profile. Turnover rate was then computed as production divided by average fine root biomass for the entire profile. For the whole profile approach, instead, we used the summed bio- and necromass values for the entire profile to compute fine root production and then divided by average fine root biomass to calculate the fine root turnover rate. For this comparison of the two approaches, sequential coring data, decision matrix calculations and mean biomass values were taken from Hertel (1999), Richter (2007), Makkonen and Helmisaari (1999), Bakker (1999), and Ostonen et al. (2005). In the reports relative to seven sites we had fine root data separately assessed for sub diameter classes (<1 mm, 1–2 mm) and so we could do the calculations for each diameter class and compare them with the total diameter class (<2 mm). And, utilizing studies with longest data series, we explored whether and how different observations lengths (1 yr, 1–2 yr, 2–3 yr), the start of the observation period (spring, summer, autumn, winter), and the number of samplings per year may influence the turnover values.
Statistics
For statistical analyses, simple linear regression and Mann–Whitney U test, the software StatView 5.0 (SAS Institute, Cary, NY, USA) was used, with the significance level of p < 0.05. The data was tested for normal distribution and for homogeneity of variances among groups.
Results
Fine-root turnover rate
Turnover rates obtained by the combination of sequential coring, Decision Matrix method, and the maximum biomass data varied from 0.19 to 2.04 yr−1 for F. sylvatica and from 0.44 to 1.36 yr−1 for P. abies (Table 3), with mean values for F. sylvatica and P. abies of 0.86 and 0.88 yr−1, respectively (Table 5). Using the mean biomass instead of the maximum biomass, the turnover rates varied from 0.23 to 2.92 yr−1 for F. sylvatica and from 0.56 to 1.77 yr−1 for P. abies (Table 3), with mean values of 1.11 yr−1 for both F. sylvatica and P. abies (Table 5). For other tree species, less than three data sets were available, e.g. only 2 data sets were available for P. sylvestris, and both had turnover rates higher than 1.5 yr−1 (Table 3).
Turnover rates obtained by the combination of sequential coring, Maximum-Minimum method, and maximum biomass data were consistently below 0.7 yr−1 for F. sylvatica and P. abies (Table 3), with mean turnover rates of 0.41 yr−1 and 0.44 yr−1, respectively (Table 5). The mean turnover rate of P. sylvestris was 0.48 yr−1 and did fall in a similar range (Table 5). Using the mean biomass instead of the maximum biomass, the turnover rates ranged from 0.26 to 0.95 yr−1 for F. sylvatica and P. abies (Table 3), with mean turnover rates of 0.53 yr−1 for F. sylvatica and 0.57 yr−1 for P. abies (Table 5).
Using the ingrowth core method, in maximum three data sets were available per tree species (Table 4). Mean turnover rates obtained by ingrowth cores, the Decision Matrix method, and the maximum biomass were 1.00, 0.72, and 0.76 yr−1 for F. sylvatica, P. abies, and P. sylvestris, respectively (Table 5). Using the Maximum-Minimum method and the maximum biomass, the mean turnover rates were with 1.00, 0.62, and 0.72 yr−1, respectively, in a similar range (Table 5). Using the mean biomass instead of the maximum biomass, the mean turnover rates were higher, 2.58, 1.15, and 1.40 yr−1 for F. sylvatica, P. abies, and P. sylvestris, respectively, using the Decision Matrix, and 2.58, 0.98, and 1.31 yr−1 for F. sylvatica, P. abies, and P. sylvestris, respectively, using the Maximum-Minimum formula (Table 5).
We compared the difference in turnover rate estimates based on maximum or mean biomass as the denominator. On average in our dataset, using mean biomass rather than maximum resulted in about 30 % higher estimate of root turnover rate T (\( {{\text{T}}_{\text{Bmean}}} = {1}.{3}\;{{\text{T}}_{\text{Bmax}}} - 0.00{1} \); r 2 = 0.98, p < 0.001; Fig. 1).
Soil stratification and root turnover rate
Our results show that a layer-per-layer approach yields a higher turnover rate than a ‘one soil layer’ approach (Fig. 2). Using average data for the whole of the soil profile, as opposed to using data for individual layers, does not capture all observed differences in root biomass and therefore results in a lower estimate of NPP and thus significantly lower turnover rate T (\( {{\text{T}}_{\text{whole profile}}} = 0.{88}\;{{\text{T}}_{{{\text{layer}} - {\text{per}} - {\text{layer}}}}} - 0.{17} \); r 2 = 0.91, p < 0.001; Fig. 2).
Relationship between turnover rates calculated per whole soils profiles or per individual soil layers (summed versus individual layers). Turnover rates were calculated the whole data set of sequential coring and using the Decision Matrix method and maximum biomass data (data from Hertel 1999; Richter 2007; Makkonen and Helmisaari 1999; Bakker 1999; Ostonen et al. 2005). Mean soil depth is 44 cm, and the average number of individual soil layers is four
Comparison between the Decision Matrix and the Maximum-Minimum method
Mean turnover rates calculated with the Decision Matrix were significantly higher than rates calculated with the Maximum-Minimum method (p < 0.001, Fig. 3). The Decision Matrix methods yielded T approximately double the Maximum-Minimum method. The turnover rates were significantly different when using mean biomass data (1.14 yr−1 from the Decision Matrix versus 0.57 yr−1 from the Maximum-Minimum method) as well as when using maximum biomass data (0.88 yr−1 versus 0.43 yr−1). Using mean biomass data resulted in significantly higher turnover rates compared to the use of maximum biomass data (p = 0.006, Fig. 3), with a mean difference of about 30 %.
Discussion
Decision Matrix versus Maximum-Minimum method
By analysing our European data set, we found about two times higher root turnover rates when using the Decision Matrix method compared to the Maximum-Minimum method. The observed discrepancy is best described by the fact that Decision Matrix accumulates differences between all observations—the larger the number of interim observations (e.g. monthly observations) the larger the potential for accounting all the peaks and troughs. The Maximum-Minimum method, on the other hand, makes use only of the annual net gain in biomass. On the basis of our comparison, we suggest that the Maximum-Minimum method should be used with caution; by definition, root turnover rates calculated by this method are bound between 0 and 1. Although this range may cover some ecosystems, it cannot correctly capture reality in systems where faster root turnover has been observed (e.g. Lukac et al. 2003) as it was shown for fast growing species (Jourdan et al. 2008). The Maximum-Minimum method is therefore only suitable for ecosystems with strong annual fluctuation of fine root biomass where turnover rate is not expected to exceed 1. In a forest ecosystem where root production and root death occur continuously and on a similar level all year round (e.g. ‘steady state’), no differences between maximum and minimum biomass will be observed. Such an observation will result in a zero estimate of root production and subsequently a zero estimate of root turnover rate (see also Kurz and Kimmins 1987).
Moving on to the Decision Matrix method, the weak point of this method is—as with all methods using dead roots—the difficulty of quantifying root necromass. The potentially rapid disappearance of root necromass may lead to underestimates (Hendricks et al. 2006). One of possible reasons for the rapid disappearance is belowground herbivory (Stevens et al. 2002). Nevertheless, we propose that if necromass observations are available or can be obtained, the Decision Matrix should be favoured over the Minimum-Maximum formula. The former considers both living and dead fine roots, the calculation is thus based on more information, reducing the scope for significant errors. One potential source of error, however, is if all the differences in fine root biomass and necromass between sampling periods are subjected to the calculations regardless of their statistical significance. This may lead to overestimation.
Even though root coring methods—whether sequential or ingrowth—do deliver dependable and comparable measurements of fine root turnover, the application of the minirhizotron technique to estimate fine-root production and turnover is still favoured over the sequential coring or the ingrowth core method in certain situations (Hendricks et al. 2006). Turnover rate estimates obtained by minirhizotron studies can be higher than 1 and the method allows for repeated observation of the same roots. However, in some forest ecosystems, application of minirhizotron methods to measure fine-root production is hampered, e.g. in stony or shallow soils or on steep slopes. Sequential coring and ingrowth core methods are suitable even for these environments, giving them an advantage in terms of comparability of resulting data.
Maximum biomass versus mean biomass
By definition, the denominator in the root turnover calculation equation is the representation of biomass present in the soil. An assumption inherent to all root turnover calculation method is that annual fine root production (obtained by whatever method) equals to fine root mortality and the system is at steady state on an annual basis. Over the course of a year, new growth replaces roots which have died. The proportion of roots which have been replaced can therefore be calculated as root production over biomass. At the present, both maximum and mean root biomass are used, with about two-thirds of studies using maximum biomass (Gill and Jackson 2000). They justified the use of the maximum biomass as “…because it is an extensively used model of root turnover and because of its heuristic value”. When constructing models of root allocation in forests, a case can be made for maximum biomass to be the preferred parameter over mean or minimum values due to the importance of setting an upper limit for the allocation rate. Fine root allocation rate may depend on sink strength (C demand), but might ultimately be limited by the maximum fraction of GPP which trees can allocate to root systems (Astrid Meyer, personal communication; see also Farrar and Jones 2000; Gower et al. 1996; Poorter et al. 2012). Having said that, and bearing in mind that the root turnover calculation assumes an ecosystem at steady state, a mean value is indicative of the long-term average as it evens out seasonal variation in biomass. Maximum biomass, on the other hand, is substantially more susceptible to between-year fluctuations due to climatic variation, which occur even if a forest ecosystem is at a steady state. Thus, we propose that mean biomass rather than the maximum is more representative of the annual live biomass present in the soil. The use of mean biomass in our calculations increased the turnover rates by about 30 % compared to the use of the maximum biomass.
An additional factor significantly affecting the results of the turnover calculations is the use of summed up values of biomass, necromass, and productivity for the whole soil profile versus using these data for individual soil layers (horizons). We acknowledge that using individual horizons should be preferable as the root turnover rate may be affected by differing physical and chemical characteristics of individual horizons. We established that basing root turnover rate calculation on individual horizon data increases the overall turnover rate—probably because it allows for better capture of biomass and necromass variations over time. We are, however, aware that root biomass and production observation on a horizon basis constitutes a significant technical challenge and contend that using whole-soil data is acceptable. Further factors potentially influencing the turnover rate, e.g. soil depth, length of study, or root diameter class have also been tested in this study, however, the available European dataset for these parameters was limited and did not allow further deductions. Thus, besides the uncertainties due to climatic and calculation reasons, many other external factors may potentially affect the estimates of root turnover rates. At present, no available technique can solve this predicament and we put forward that our root turnover rates represent the best approximation obtained by using sequential soil or ingrowth cores.
Turnover rates of European tree species
Our review of published studies from European forest stands revealed that most data for fine-root turnover rate originate from sequential coring, with the prevalence of Fagus sylvatica or Picea abies as the species of interest. Studies performed in forest stands with other dominating tree species such as Quercus spp., Pinus spp. were far less abundant. Similarly, turnover rate studies where ingrowth cores were used instead of employing the sequential coring method to measure fine-root production, were far less abundant. Whereas in our study the data sets of F. sylvatica derived mainly from Central Europe, the data sets of P. abies originated from Central as well as from Northern Europe. Trees from Southern European countries were represented only by a few data sets, and no conclusive turnover rates can be suggested for this environment yet. Overall, we propose that only the fine root turnover rates in our study for the following species may be recommended for further use in biogeochemical models with a reasonable degree of accuracy: F. sylvatica and P. abies. We established a turnover rate of 1.11 yr−1 for both F. sylvatica and P. abies, using the Decision Matrix formula and the mean biomass data from sequential coring.
Turnover rates applied in biogeochemical models
One of the aims of the present study was to deliver suitable fine-root turnover data of European tree species, which may be used by modellers to construct ecosystem or biogeochemical models. Such models are applied in many European countries to report the change of belowground C in European forests as a reporting requirement for the Kyoto protocol signatories. A brief overview of the models applied so far shows that a wide variety of root turnover rates are used, some resembling measured values, others less so. In one of the first applications, the fine-root turnover rate was set to 1.0 yr−1 for deciduous broad-leaf and deciduous needle-leaf trees and to 0.26 yr−1 for evergreen needle-leaf trees (White et al. 2000, using the Biome-BGC model). The distinct difference between deciduous trees and evergreen needle-leaf trees mainly originated from the notion that fine-root turnover rate is equal to leaf turnover rate. A compilation of the various turnover rates applied in European modelling studies is shown in Table 6. Most recent studies applied a universal fine-root turnover rate of 0.7 yr−1 to all forest tree species (Hickler et al. 2008, using the LPJ-GUESS model). This assumption is based on Vogt et al. (1996) and on Li et al. (2003) (Thomas Hickler, personal communication). Li et al. (2003) found a linear relationship between fine root production and fine root biomass, with the turnover rate 0.64 yr−1 which was lower than the original estimate of 0.73 y−1 from a previous analysis (Kurz et al. 1996). Using ‘universal’ turnover rates, however, should be discouraged if country-based C budgets have to be reported within the frame to the Kyoto protocol and species-specific and biome based values of root turnover rate are available.
Conclusions
The present synthesis on fine-root turnover of European forests reveals that only Fagus sylvatica and Picea abies have sufficient data availability to suggest mean turnover rates obtained by soil coring to be used by National C reporters (0.86 ± 0.16 yr−1 for F. sylvatica, 0.88 ± 0.11 yr−1 for P. abies, when maximum biomass data are used; 1.11 ± 0.21 yr−1 for F. sylvatica, 1.11 ± 0.14 yr−1 for P. abies, when mean biomass data are used). Data sets of other European forests or obtained by alternative methods such as ingrowth cores were too small to allow for distinct conclusions on the turnover rates. Based on our calculations, we put forward that usage of mean rather than maximum root biomass in turnover calculations is preferable as it better reflects long-term quantity of biomass.
Abbreviations
- B:
-
Biomass
- BGC:
-
Biogeochemical cycles
- C:
-
Carbon
- DM:
-
Decision Matrix
- GPP:
-
Gross primary production
- GUESS:
-
General ecosystem simulator
- LPJ:
-
Lund-Potsdam-Jena model
- MM:
-
Maximum-Minimum
- MRT:
-
Mean residence time
- N:
-
Necromass
- NPP:
-
Net primary production
- P:
-
Production
- SOM:
-
Soil organic matter
- T:
-
Turnover rate
References
Bakker MR (1999) The effect of lime and gypsum applications on a sessile oak (Quercus petraea (M.) Liebl.) stand at La Croix Scaille (French Ardennes) II. Fine root dynamics. Plant Soil 206:109–121
Børja I, De Wit HA, Steffenrem A, Majdi H (2008) Stand age and fine root biomass, distribution and morphology in a Norway spruce chronosequence in southeast Norway. Tree Physiol 28:773–784
Brunner I, Godbold DL (2007) Tree roots in a changing world. J For Res 12:78–82
Brunner I, Graf Pannatier E, Frey B, Rigling A, Landolt W, Zimmermann S, Dobbertin M (2009) Morphological and physiological responses of Scots pine fine roots to water supply in a dry climatic region in Switzerland. Tree Physiol 29:541–550
Cienciala E, Tatarinov FA (2006) Application of BIOME-BGC model to managed forests. 2. Comparison with long-term observations of stand production for major tree species. For Ecol Manag 237:252–266
Eldhuset TD, Lange H, de Wit HA (2006) Fine root biomass, necromass and chemistry during seven years of elevated aluminium concentrations in the soil solution of a middle-aged Picea abies stand. Sci Total Environ 369:344–356
Endrulat T, Saurer M, Buchmann N, Brunner I (2010) Incorporation and remobilization of 13C within the fine-root systems of individual Abies alba trees in a temperate coniferous stand. Tree Physiol 30:1515–1527
Fairley RI, Alexander IJ (1985) Methods of calculating fine root production in forests. In: Fitter AH, Atkinson D, Read DJ (eds) Ecological interactions in soil: plants, microbes and animals. Blackwell, Oxford, pp 37–42
Farrar JF, Jones DL (2000) The control of carbon acquisition by roots. New Phytol 147:43–53
Finer L, Ohashi M, Noguchi K, Hirano Y (2011) Fine root production and turnover in forest ecosystems in relation to stand and environmental characteristics. For Ecol Manag 262:2008–2023
Fritz HW (1999) Feinwurzel-Verteilung, -Vitalität, -Produktion und -Umsatz von Fichten (Picea abies (L.) Karst.) auf unterschiedlich versauerten Standorten. Ber Forsch Zent Waldökosyst, Reihe A 165:1–138
Gaudinski JB, Trumbore SE, Davidson EA, Cook AC, Makrewitz D, Richter DD (2001) The age of fine-root carbon in three forests of the eastern United States measured by radiocarbon. Oecologia 129:420–429
Gaudinski JB, Tom MS, Riley WJ, Dawson TE, Joslin JD, Majdi H (2010) Measuring and modeling the spectrum of fine-root turnover times in three forests using isotopes, minirhizotrons, and the Radix model. Glob Biogeochem Cycles 24:GB3029
Gaul D, Hertel D, Leuschner C (2009) Estimating fine root longevity in a temperate Norway spruce forest using three independent methods. Funct Plant Biol 36:11–19
Genenger M, Zimmermann S, Hallenbarter D, Landolt W, Frossard E, Brunner I (2003) Fine root growth and element concentrations of Norway spruce as affected by wood ash and liquid fertilisation. Plant Soil 255:253–264
Gill RA, Jackson R (2000) Global patterns of root turnover for terrestrial ecosystems. New Phytol 147:13–31
Gower ST, Pongracic S, Landsberg JJ (1996) A global trend in belowground carbon allocation: Can we use the relationship at smaller scales? Ecology 77:1750–1755
Haynes BE, Gower ST (1995) Belowground carbon allocation in unfertilized and fertilized red pine plantations in Northern Wisconsin. Tree Physiol 15:317–325
Hendricks JJ, Hendrick RL, Wilson CA, Mitchell RJ, Pecot SD, Guo D (2006) Assessing the patterns and controls of fine root dynamics: an empirical test and methodological review. J Ecol 94:40–57
Hertel D (1999) Das Feinwurzelsystem von Rein- und Mischbeständen der Rotbuche: Struktur, Dynamik und interspezifische Konkurrenz. Diss Bot 317:1–185
Hickler T, Smith B, Sykes MT, Davis M, Sugita S, Walker K (2004) Using a generalized vegetation model to simulate vegetation dynamics in northeastern USA. Ecology 85:519–530
Hickler T, Prentice IC, Smith B, Sykes MT, Zaehle S (2006) Implementing plant hydraulic architecture within the LPJ dynamic global vegetation model. Glob Ecol Biogeogr 15:567–577
Hickler T, Smith B, Prentice C, Mjöfors K, Miller P, Arnetz A, Sykes MT (2008) CO2 fertilization in temperate FACE experiments not representative of boreal and tropical forests. Glob Change Biol 14:1531–1542
Hirano Y, Noguchi K, Ohashi M, Hishi T, Makita N, Fujii S, Finér L (2009) A new method for placing and lifting root meshes for estimating fine root production in forest ecosystems. Plant Root 3:26–31
Johnson MG, Tingey DT, Phillips DL, Storm MJ (2001) Advancing fine root research with minirhizotrons. Environ Exp Bot 45:263–289
Jourdan C, Silva EV, Gonçalves JLM, Ranger J, Moreira RM, Laclau JP (2008) Fine root production and turnover in Brazilian Eucalyptus plantations under contrasting nitrogen fertilization regimes. For Ecol Manag 256:396–404
Konôpka B (2009) Differences in fine root traits between Norway spruce (Picea abies (L.) Karst.) and European beech (Fagus sylvatica L.)—a case study in the Kysucké Beskydy Mts. J For Sci 55:556–566
Konôpka B, Yuste JC, Janssens IA, Ceulemans R (2005) Comparison of fine root dynamics in Scots pine and Pedunculate oak in sandy soil. Plant Soil 276:33–45
Kurz WA, Kimmins JP (1987) Analysis of some sources of error in methods used to determine fine root production in forest ecosystems: a simulation approach. Can J For Res 17:909–912
Kurz WA, Beukema SJ, Apps MJ (1996) Estimation of root biomass and dynamics for the carbon budget model of the Canadian forest sector. Can J For Res 26:1973–1979
Li Z, Kurz WA, Apps MJ, Beukema SJ (2003) Belowground biomass dynamics in the carbon budget model of the Canadian forest sector: recent improvements and implications for the estimation of NPP and NEP. Can J For Res 33:106–126
López B, Sabaté S, Gracia CA (2001) Annual and seasonal changes in fine root biomass of a Quercus ilex L. forest. Plant Soil 230:125–13
Lukac M, Godbold DL (2010) Fine root biomass and turnover in southern taiga estimated by root inclusion nets. Plant Soil 331:505–513
Lukac M, Calfapietra C, Godbold DL (2003) Production, turnover and mycorrhizal colonization of root systems of three Populus species grown under elevated CO2 (POPFACE). Glob Change Biol 9:838–848
Majdi H, Andersson P (2005) Fine root production and turnover in a Norway spruce stand in northern Sweden: effects of nitrogen and water manipulation. Ecosystems 8:191–199
Majdi H, Pregitzer K, Morén AS, Nylund JE, Ågren GI (2005) Measuring fine root turnover in forest ecosystems. Plant Soil 276:1–8
Makkonen K, Helmisaari HS (1999) Assessing fine-root biomass and production in a Scots pine stand—comparison of soil core and root ingrowth core methods. Plant Soil 210:43–50
Matamala R, Gonzalez-Meler MA, Jastrow JD, Norby RJ, Schlesinger WH (2003) Impacts of fine root turnover on forest NPP and soil C sequestration potential. Science 302:1385–1387
McClaugherty CA, Aber JD, Melillo JM (1982) The role of fine roots in the organic-matter and nitrogen budgets of two forested ecosystems. Ecology 63:1481–1490
Miguel Pérez C (2010) Respuestas ecofisiologicas y estructurales a la recurrencia, duracion e intensidad de la sequia en plantaciones y bosques mixtos de Quercus ilex, Quercus pubescens y Quercus cerrioides. Dissertation, Institut de Recerca i Tecnologia Agroalimentaries IRTA, Universitat Autonoma de Barcelona UAB
Osawa A, Aizawa R (2012) A new approach to estimate fine root production, mortality, and decomposition using litter bag experiments and soil core techniques. Plant Soil. doi:10.1007/s11104-011-1096-6
Ostonen I, Lohmus K, Pajuste K (2005) Fine root biomass, production and its proportion of NPP in a fertile middle-aged Norway spruce forest: comparison of soil core and ingrowth core methods. For Ecol Manag 21:264–277
Persson H (1980a) Fine-root production, mortality and decomposition in forest ecosystems. Vegetatio 41:101–109
Persson H (1980b) Fine-root dynamics in a Scots pine stand, with and without near optimum nutrient and water regimes. Acta Phytogeogr Suec 68:101–110
Persson HÅ, Stadenberg I (2010) Fine-root dynamics in a Norway spruce forest (Picea abies (L.) Karst) in Eastern Sweden. Plant Soil 330:329–344
Pietsch SA, Hasenauer H, Thornton PE (2005) BGC-model parameters for tree species growing in central European forests. For Ecol Manag 211:264–295
Poorter H, Niklas KJ, Reich PB, Oleksyn J, Poot P, Mommer L (2012) Biomass allocation to leaves, stems and roots: meta-analyses of interspecific variation and environmental control. New Phytol 193:30–50
Publicover DA, Vogt KA (1993) A comparison of methods for estimating forest fine root production with respect to sources of error. Can J For Res 23:1179–1186
Richter A (2007) Fine root growth and vitality of European beech in acid forest soils with a low base saturation. Dissertation, ETH Zürich
Sah SP, Jungner H, Oinonen M, Kukkola M, Helmisaari H-S (2011) Does the age of fine root carbon indicate the age of fine roots in boreal forests? Biogeochemistry 104:91–102
Santantonio D, Grace JC (1987) Estimating fine-root production and turnover from biomass and decomposition data: a compartment-flow model. Can J For Res 17:900–908
Silver WL, Thompson AW, McGroddy ME, Varner RK, Dias JD, Silva H, Crill PM, Keller M (2005) Fine root dynamics and trace gas fluxes in two lowland tropical forest soils. Glob Change Biol 11:290–306
Sitch S, Smith B, Prentice IC, Arneth A, Bondeau A, Cramer W, Kaplan J, Levis S, Lucht W, Sykes M, Thonicke K, Venevsky S (2003) Evaluation of ecosystem dynamics, plant geography and terrestrial carbon cycling in the LPJ Dynamic Global Vegetation Model. Glob Change Biol 9:161–185
Smith B, Prentice IC, Sykes MT (2001) Representation of vegetation dynamics in the modelling of terrestrial ecosystems: comparing two contrasting approaches within European climate space. Glob Ecol Biogeogr 10:621–637
Stevens GN, Mitchell RJ, Jones RH (2002) Rapid fine root disappearance in a pine woodland: a substantial carbon flux. Can J For Res 32:2225–2230
Stober C, George E, Persson H (2000) Root growth and response to nitrogen. In: Schulze ED (ed) Carbon and nitrogen cycling in European forest ecosystems. Springer, Berlin, pp 99–121
Strand AE, Pritchard SG, McCormack ML, Davis MA, Oren R (2008) Irreconcilable differences: fine-root life spans and soil carbon persistence. Science 319:456–458
Tatarinov FA, Cienciala E (2006) Application of BIOME-BGC model to managed forests 1. Sensitivity analysis. For Ecol Manag 237:267–279
Tierney G, Fahey T (2002) Fine root turnover in a northern hardwood forest: a direct comparison of the radiocarbon and minirhizotron methods. Can J For Res 32:1692–1697
Trumbore SE, Gaudinski JB (2003) The secret lives of roots. Science 302:1344–1345
Vogt KA, Persson H (1991) Measuring growth and development of roots. In: Lassoie JP, Hinkley TM (eds) Techniques and approaches in forest tree ecophysiology. CRC Press, Boca Raton, pp 477–501
Vogt KA, Vogt DJ, Palmiotto PA, Boon P, O’Hara J, Asbjornson H (1996) Review of root dynamics in forest ecosystems grouped by climate, climatic forest type and species. Plant Soil 187:159–219
Vogt KA, Vogt DJ, Bloomfield J (1998) Analysis of some direct and indirect methods for estimating root biomass and production of forest at ecosystem level. Plant Soil 200:71–89
White MA, Thornton PE, Running SW, Nemai RR (2000) Parametrization and sensitivity analysis of the BIOME-BGC terrestrial ecosystem Model: net primary production controls. Earth Interact 4:1–85
Wu K (2000) Fine root production and turnover and its contribution to nutrient cycling in two beech (Fagus sylvatica L.) forest ecosystems. Ber Forsch Zent Waldökosyst, Reihe A 170:1–130
Yuan ZH, Chen HYH (2010) Fine root biomass, production, turnover rates, and nutrient contents in boreal forest ecosystems in relation to species, climate, fertility, and stand age: literature review and meta-analyses. Crit Rev Plant Sci 29:204–221
Acknowledgements
This study is a result of the activity of a Working Group of the COST Action FP0803 “Belowground carbon turnover in European forests”. We thank Astrid Meyer and Thomas Hickler for helpful comments on the calculations and the models, and Beat Frey for critical reading. We also would like to thank Carmen Biel, Karna Hansson, Peter Zeleznik, Thomas Leski, Inger-Kappel Schmidt, Antonino di Iorio, Kyotaro Noguchi, and Yoni Ephrat, who helped us in any way to write up this review. Claude Herzog helped with statistical tests.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Alexia Stokes.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Brunner, I., Bakker, M.R., Björk, R.G. et al. Fine-root turnover rates of European forests revisited: an analysis of data from sequential coring and ingrowth cores. Plant Soil 362, 357–372 (2013). https://doi.org/10.1007/s11104-012-1313-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-012-1313-5