Abstract
Aims
It was shown previously that Arabidopsis (Arabidopsis thaliana) desaturase 2 (ADS2) cDNA was isolated and it was shown that the expression of ADS2 was organ-dependent and up-regulated by low temperature. However, little is known about the role of ADS2 gene in heavy metal resistance in plants. In this study, we showed that ADS2 gene is involved in the regulation of cadmium (Cd) and lead (Pb) resistance.
Methods
For heavy metal resistance tests, seeds were germinated and grown on 1/2 MS media supplemented with the indicated concentrations of metal ions. To quantify root length, plants were grown vertically in plates. For heavy metal treatments, two-week old wild-type seedlings grown on MS media were treated with cadmium (Cd) or lead (Pb) for 24 h, and then sampled for metal content measurement and qPCR analysis.
Results
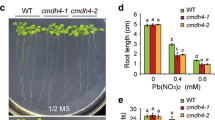
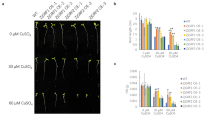
ADS2 was strongly repressed by Cd(II), and ads2-1 mutant plants showed increased Cd(II) resistance. A lower Cd content was detected in ads2-1 plants than in wild-type plants subjected to Cd(II) treatment, which was associated with activation in expression of AtPDR8 gene, a pump excluding Cd(II) and/or Cd(II)-containing toxic compounds from the cytoplasm, suggesting that ADS2-mediated Cd(II) resistance is AtPDR8 dependent. We also found that ads2-1 plants showed increased Pb(II) sensitivity, and ADS2 was strongly repressed by hydrogen peroxide (H2O2) but not by Pb(II). The ads2-1 mutant showed increased sensitivity to oxidative stresses mediated by H2O2 and paraquat, and higher levels of H2O2 accumulation were observed in leaves of ads2-1 plants than those of wild-type plants when subjected to Pb(II) and H2O2, indicating that ADS2 mediates Pb(II) resistance indirectly by impaired ROS scavenging.
Conclusions
ADS2 gene mediates Cd(II) and Pb(II) resistance, at least in part, through two distinct mechanisms, an AtPDR8-dependent mechanism and a ROS detoxification system-mediated mechanism, respectively.










Similar content being viewed by others
References
Alonso JM, Stepanova AN, Leisse TJ, Kim CJ, Chen H, Shinn P, Stevenson DK, Zimmerman J, Barajas P, Cheuk R, Gadrinab C, Heller C, Jeske A, Koesema E, Meyers CC, Parker H, Prednis L, Ansari Y, Choy N, Deen H, Geralt M, Hazari N, Hom E, Karnes M, Mulholland C, Ndubaku R, Schmidt I, Guzman P, Aguilar-Henonin L, Schmid M, Weigel D, Carter DE, Marchand T, Risseeuw E, Brogden D, Zeko A, Crosby WL, Berry CC, Ecker JR (2003) Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301:653–657
Cao SQ, Chen ZY, Liu GQ, Jiang L, Yuan HB, Ren G, Bian XH, Jian HY, Ma XL (2009) The Arabidopsis Ethylene-Insensitive 2 gene is required for lead resistance. Plant Physiol Biochem 47:308–312
Clemens S (2001) Molecular mechanisms of plant metal tolerance and homeostasis. Planta 212:475–486
Fukuchi-Mizutani M, Tasaka Y, Tamaka Y, Ashikari T, Kusumi T, Murata N (1998) Characterization of delta 9 acyl-lipid desaturase homologues from Arabidopsis thaliana. Plant Cell Physiol 39:247–253
Gallego SM, Benavides MP, Tomaro ML (2002) Involvement of antioxidant defense system in the adaptive response to heavy metal ions in Helianthus annuus L. cells. Plant Growth Regul 36:17–21
Goyer RA (1997) Toxic and essential metal interactions. Annu Rev Nutr 17:7–50
Hall JL (2002) Cellular mechanisms for heavy metal detoxification and tolerance. J Exp Bot 53:1–11
Heilmann MS, Pidkowich T, Girke Shanklin J (2004) Switching desaturase enzyme specificity by alternate subcellular targeting. Proc Natl Acad Sci USA 101:10266–10271
Jasinski M, Sudre D, Schansker G, Schellenberg M, Constant S, Martinoia E, Bovet L (2008) AtOSA1, a member of Abc1-like family as a new factor in cadmium and oxidative stress response. Plant Physiol 147:719–731
Kim DY, Bovet L, Kushnir S, Noh EW, Martinoia E, Lee Y (2006) AtATM3 is involved in heavy metal resistance in Arabidopsis. Plant Physiol 140:922–932
Kim DY, Bovet L, Maeshima M, Martinoia E, Lee Y (2007) The ABC transporter AtPDR8 is a cadmium extrusion pump conferring heavy metal resistance. Plant J 50:207–218
Kunst L, Browse J, Somerville C (1989) A mutant of Arabidopsis deficient in desaturation of palmitic acid in leaf lipids. Plant Physiol 90:943–947
Kurepa J, Smalle J, Va M, Montagu N, Inez D (1998) Oxidative stress tolerance and longevity in Arabidopsis: the late-flowering mutant gigantea is tolerant to paraquat. Plant J 14:759–764
Lanphear BP (1998) The paradox of lead poisoning prevention. Science 281:1617–1618
Lee M, Lee K, Lee J, Noh EW, Lee Y (2005) AtPDR12 contributes to lead resistance in Arabidopsis. Plant Physiol 138:827–836
Lim CE, Choi JN, Kim IA, Lee SA, Hwang YS, Lee CH, Lim J (2008) Improved resistance to oxidative stress by a loss-of-function mutation in the Arabidopsis UGT71C1 gene. Mol Cells 25:368–375
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Okuda T, Matsuda Y, Yamanaka A, Sagisaka S (1991) Abrupt increase in the level of hydrogen peroxide in leaves of winter wheat is caused by cold treatment. Plant Physiol l97:1265–1267
Ortiz DF, Kreppel L, Spaser DM (1995) Transport of metalbinding peptides by HMT1, a fission yeast ABC-type vacuolar membrane protein. J Biol Chem 270:4721–4727
Raskin I, Smith RD, Salt DE (1997) Phytoremediation of metals: using plants to remove pollutants from the environment. Curr Opin Biotechnol 8:221–226
Schutzendubel A, Schwanz P, Teichmann T, Gross K, Langenfeld-Heyser R, Godbold DL, Polle L (2001) Cadmium-induced changes in antioxidative systems, hydrogen peroxide content, and differentiation in Scots pine roots. Plant Physiol 127:887–898
Sharma R, Rensing C, Rosen BP, Mitra B (2000) The ATP hydrolytic activity of purified ZntA, a Pb(II)/Cd(II)/Zn(II)-translocating ATPase from Escherichia coli. J Biol Chem 275:3873–3878
Shiraishi E, Inouhe M, Joho M, Tohoyama H (2000) The cadmium-resistant gene, CAD2, which is a mutated putative copper-transporter gene (PCA1), controls the intracellular cadmium-level in the yeast S. cerevisiae. Curr Genet 37:79–86
Song WY, Martinoia E, Lee J, Kim D, Kim DY, Vogt E, Shim D, Choi KS, Hwang I, Lee Y (2004) A novel family of cys-rich membrane proteins mediates cadmium resistance in Arabidopsis. Plant Physiol 135:1027–1039
Vijayan P, Browse J (2002) Photoinhibition in mutants of Arabidopsis deficient in thylakoid unsaturation. Plant Physiol 129:876–885
Williams LE, Pittman JK, Hall JL (2000) Emerging mechanisms for heavy metal transport in plants. Biochim Biophys Acta 1465:104–126
Xiao S, Gao W, Chen QF, Ramalingam S, Chye ML (2008) Overexpression of membrane-associated acyl-CoA-binding protein ACBP1 enhances lead tolerance in Arabidopsis. Plant J 54:141–151
Yuan JS, Reed A, Chen F, Stewart CN (2006) Statistical analysis of real-time PCR data. BMC Bioinformatics 7:85
Zimmermann P, Hirsch-Hoffmann M, Hennig L, Gruissem W (2004) GENEVESTIGATOR. Arabidopsis microarray database and analysis toolbox. Plant Physiol 136:2621–2632
Zsigmond L, Rigó G, Szarka A, Székely G, Ötvös K, Darula Z, Medzihradszky KF, Koncz C, Koncz S, Szabados L (2008) Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport. Plant Physiol 146:1721–1737
Acknowledgements
We thank Zhenyi Chen, Hongyong Jian, Jiajia Sun and Yongbin Ren for their technical assistances. This work was supported by the National Natural Science Foundation of China (grant nos. 20777014 and 31070241), the National Transgenic Plant R&D Project of China (2009ZX08009-063B), the Great Project of Natural Science Foundation at Anhui Provincial Education Department (KJ2010ZD04), and the Funds for Creative Research Groups of Hefei University of Technology (2009HGCX0233).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Robert Reid.
Bin Yu and Xiaohui Bian contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yu, B., Bian, X., Qian, J. et al. Arabidopsis desaturase 2 gene is involved in the regulation of cadmium and lead resistance. Plant Soil 358, 289–300 (2012). https://doi.org/10.1007/s11104-012-1186-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-012-1186-7