Abstract
Background and aims
Phosphorus (P) nutrition is very important during early maize seedling growth. Remobilization of endogenous seed P and uptake of exogenous P are therefore of prime importance during this period. Our objectives were to study the effect of the availability of endogenous and exogenous P on i) remobilization of endogenous seed P, ii) the beginning of exogenous P uptake and its intensity, iii) their interaction and effect on seedling development.
Methods
Seeds with high and low reserves of endogenous seed P were cultivated at three rates of availability of exogenous P (0, 100, 1,000 μM) over a growth period of 530 cumulated degree days after sowing. Exogenous P was labeled with radioactive P (32P) to distinguish the two fluxes of P in seedlings, one due to remobilization of seed P and the other to uptake of exogenous P.
Results
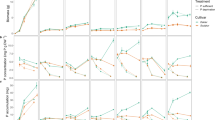
Initially, 86% of endogenous seed P was localized in the scutellum, mainly in the form of phytate, regardless of initial endogenous seed P. At 89 cumulated degree days after sowing (base temperature: 10°C), 98% of seed phytate was hydrolyzed in all treatments. In treatments with available exogenous P, significant uptake of exogenous P started at 71 cumulated degree days after sowing. Efficient uptake of exogenous P depended on its availability, but was independent of phytate hydrolysis and seedling P status. Significant loss of P from germinating seeds due to efflux was observed and was also independent of the availability of exogenous P.
Conclusions
Our results show that hydrolysis of seed P was not influenced by the availability of exogenous P, and conversely, that uptake of exogenous P was not influenced by endogenous P in the seed. This suggests that remobilization of endogenous seed P and uptake of exogenous P by seedling roots are controlled independently.







Similar content being viewed by others
Abbreviations
- LS seeds:
-
Seeds with low endogenous seed P
- HS seeds:
-
Seeds with high endogenous seed P
- LS seedlings:
-
Seedlings grown from LS seeds
- HS seedlings:
-
Seedlings grown from HS seeds
- 0P:
-
No exogenous P
- LP:
-
Low exogenous P availability
- HP:
-
High exogenous P availability
- DW:
-
Dry weight
- Endo-P:
-
Endogenous seed P
- Exo-P:
-
Exogenous P uptake
References
Anghinoni I, Barber SA (1980) Phosphorus influx and growth characteristics of corn roots as influenced by phosphorus supply. Agron J 72(4):685–688
Assuero SG, Mollier A, Pellerin S (2004) The decrease in growth of phosphorus-deficient maize leaves is related to a lower cell production. Plant Cell Environ 27(7):887–895
Barry DAJ, Miller MH (1989) Phosphorus nutritional-requirement of maize seedlings for maximum yield. Agron J 81(1):95–99
Bewley JD (1997) Seed germination and dormancy. Plant Cell 9(7):1055–1066
Bhadoria PS, El Dessougi H, Liebersbach H, Claassen N (2004) Phosphorus uptake kinetics, size of root system and growth of maize and groundnut in solution culture. Plant Soil 262(1–2):327–336
Bieleski RL, Ferguson IB (1983) Physiology and metabolism of phosphate and its compounds. In: Läuchli A, Bieleski RL (eds) Encyclopedia of Plant Physiology. New Series, vol 15A. Springer, Berlin, pp 422–449
Bonhomme R, Derieux M, Edmeades GO (1994) Flowering of diverse maize cultivars in relation to temperature and photoperiod in multilocation field trials. Crop Sci 34(1):156–164
Centeno C, Viveros A, Brenes A, Canales R, Lozano A, de la Cuadra C (2001) Effect of several germination conditions on total P, phytate P, phytase, and acid phosphatase activities and inositol phosphate esters in rye and barley. J Agric Food Chem 49(7):3208–3215
Colomb B, Kiniry JR, Debaeke P (2000) Effect of soil phosphorus on leaf development and senescence dynamics of field-grown maize. Agron J 92(3):428–435
Eagles HA, Hardacre AK (1979) Genetic variation in maize (Zea mays L.) for germination and emergence at 10°C. Euphytica 28(2):287–295
Egli I, Davidsson L, Juillerat MA, Barclay D, Hurrell RF (2002) The influence of soaking and germination on the phytase activity and phytic acid content of grains and seeds potentially useful for complementary feeding. J Food Sci 67(9):3484–3488
Elliott GC, Lynch J, Läuchli A (1984) Influx and efflux of P in roots of intact maize plants -double labeling with 32P and 33P. Plant Physiol 76(2):336–341
Fardeau JC (1993) Available soil phosphate—Its representation by a functional multiple compartment model. Agronomie 13(4):317–331
Fincher GB, Stone BA (1986) Cell walls and their components in cereal grain technology. In: Pomeranz Y (ed) Advences in cereal science and technology, vol 8. American Association of Cereal Chemists, St. Paul, pp 207–295
Gavito ME, Miller MH (1998) Early phosphorus nutrition, mycorrhizae development, dry matter partitioning and yield of maize. Plant Soil 199(2):177–186
Grant CA, Flaten DN, Tomasiewicz DJ, Sheppard SC (2001) The importance of early season phosphorus nutrition. Can J Plant Sci 81(2):211–224
Hall JR, Hodges TK (1966) Phosphorus metabolism of germinating oat seeds. Plant Physiol 41:1459–1464
Hegeman CE, Grabau EA (2001) A novel phytase with sequence similarity to purple acid phosphatases is expressed in cotyledons of germinating soybean seedlings. Plant Physiol 126(4):1598–1608
Laboure AM, Gagnon J, Lescure AM (1993) Purification and characterization of a phytase (myo-Inositol-hexakisphosphate phosphohydrolase) accumulated in maize (Zea mays) seedlings during germination. Biochem J 295:413–419
Lestienne I, Icard-Verniere C, Mouquet C, Picq C, Treche S (2005) Effects of soaking whole cereal and legume seeds on iron, zinc and phytate contents. Food Chem 89(3):421–425
Lockhart HB, Hurt HD (1986) Nutrition of oats. In: Webster FH (ed) Oats: chemistry and technology. American Association of Cereal Chemists, Inc., St Paul, Minnesota, USA, pp 297–308
Lorenz AJ, Scott MP, Lainkey KR (2007) Quantitative determination of phytate and inorganic phosphorus for maize breeding. Crop Sci 47(2):600–606
Lott JNA, Greenwood JS, Batten GD (1995) Mechanisms and regulation of mineral nutrient storage during seed development. In: Kigel J, Galili G (eds) Seed Development and Germination. Marcel Dekker, New York, pp 215–235
Modi AT, Asanzi NM (2008) Seed performance of maize in response to phosphorus application and growth temperature is related to phytate-phosphorus occurrence. Crop Sci 48(1):286–297
Mollier A, Pellerin S (1999) Maize root system growth and development as influenced by phosphorus deficiency. J Exp Bot 50(333):487–497
Nadeem M, Mollier A, Morel C, Vives A, Prud’homme L, Pellerin S (2011) Relative contribution of seed phosphorus reserves and exogenous phosphorus uptake to maize (Zea mays L.) nutrition during early growth stages. Plant Soil 346(1):231–244
Palomo L, Claassen N, Jones DL (2006) Differential mobilization of P in the maize rhizosphere by citric acid and potassium citrate. Soil Biol Biochem 38(4):683–692
Park SH, Sung JK, Lee SY, Park JH, Lee JY, Jang BC, Lee BH, Kim TW (2006) Early growth, carbohydrate, and phytic acid contents of germinating rice seeds under NaCl stress. Korean J Crop Sci 51(2):137–141
Pellerin S, Mollier A, Plenet D (2000) Phosphorus deficiency affects the rate of emergence and number of maize adventitious nodal roots. Agron J 92(4):690–697
Plénet D, Etchebest S, Mollier A, Pellerin S (2000) Growth analysis of maize field crops under phosphorus deficiency:I. Leaf growth. Plant and Soil 223(1–2):117–130
Römer W, Augustin J, Schilling G (1988) The relationship between phosphate absorption and root length in 9 wheat cultivars. Plant Soil 111(2):199–201
Römer W, Schilling G (1986) Phosphorus requirements of the wheat plant in various stages of its life-cycle. Plant Soil 91(2):221–229
Rubio G, Sorgona A, Lynch JP (2004) Spatial mapping of phosphorus influx in bean root systems using digital autoradiography. J Exp Bot 55(406):2269–2280
Schjørring JK, Jensén P (1984) Phosphorus nutrition of barley, buckwheat and rape seedlings. I. Influence of seed-borne P levels and external P levels on growth, P content and 32P/31P-fractionation in shoots and roots. Physiol Plant 61(4):577–583
Sung HG, Shin HT, Ha JK, Lai HL, Cheng KJ, Lee JH (2005) Effect of germination temperature on characteristics of phytase production from barley. Bioresour Technol 96(11):1297–1303
Van Veldhoven PP, Mannaerts GP (1987) Inorganic and organic phosphate measurements in the nanomolar range. Anal Biochem 161(1):45–48
Wyss M, Brugger R, Kronenberger A, Remy R, Fimbeld R, Oesterhelt G, Lehman M, Van Loon AP (1999) Biochemical characterization of fungal phytases (myo-inositol hexakisphosphate phosphohydrolyses): Catalytic properties. Appl Environ Microbiol 65:367–373
Acknowledgement
This study was funded by a project grant from the Higher Education Commission (HEC), Pakistan and benefited from the financial support from the INRA (French National Institute for Agricultural Research). M. Nadeem thanks the Pakistan Higher Education Commission for funding his PhD studentship at the University of Bordeaux I, France. The authors acknowledge the technical help and the useful advice offered by Anne Gallet-Budynek and Sylvie Milin.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Hans Lambers.
Rights and permissions
About this article
Cite this article
Nadeem, M., Mollier, A., Morel, C. et al. Maize (Zea mays L.) endogenous seed phosphorus remobilization is not influenced by exogenous phosphorus availability during germination and early growth stages. Plant Soil 357, 13–24 (2012). https://doi.org/10.1007/s11104-011-1111-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-011-1111-5