Abstract
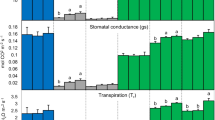
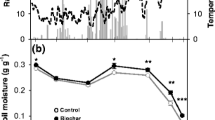
The application of pyrogenic carbon, biochar, to agricultural soils is currently discussed as a win-win strategy to sequester carbon in soil, thus improving soil fertility and mitigate global warming. Our aim was to investigate if biochar may improve plant eco-physiological responses under sufficient water supply as well as moderate drought stress. A fully randomized greenhouse study was conducted with the pseudo-cereal Chenopodium quinoa Willd, using three levels of biochar addition (0, 100 and 200 t ha−1) to a sandy soil and two water treatments (60% and 20% of the water holding capacity of the control), investigating growth, water use efficiency, eco-physiological parameters and greenhouse gas (GHG) fluxes. Biochar application increased growth, drought tolerance and leaf-N- and water-use efficiency of quinoa despite larger plant–leaf areas. The plants growing in biochar-amended soil accumulated exactly the same amount of nitrogen in their larger leaf biomass than the control plants, causing significantly decreased leaf N-, proline- and chlorophyll-concentrations. In this regard, plant responses to biochar closely resembled those to elevated CO2. However, neither soil- nor plant–soil-respiration was higher in the larger plants, indicating less respiratory C losses per unit of biomass produced. Soil-N2O emissions were significantly reduced with biochar. The large application rate of 200 t ha−1 biochar did not improve plant growth compared to 100 t ha−1; hence an upper beneficial level exists. For quinoa grown in a sandy soil, biochar application might hence provide a win-win strategy for increased crop production, GHG emission mitigation and soil C sequestration.




Similar content being viewed by others
Notes
Calculated with the total dry matter and leaf area at the harvest date, but without the few leaves that were harvested earlier for proline- Rubisco- and osmotic potential analyses.
Abbreviations
- BC:
-
Biochar
- WUE:
-
Water use efficiency
- NUE:
-
Nitrogen use efficiency
- WHC:
-
Water holding capacity
- SOC:
-
Soil organic carbon
References
Ainsworth EA, Rogers A (2007) The response of photosynthesis and stomatal conductance to rising [CO2]: mechanisms and environmental interactions. Plant Cell Environ 30:258–270. doi:10.1111/j.1365-3040.2007.01641.x
Amonette JE, Joseph S (2009) Characteristics of biochar: microchemical properties. In: Lehmann J, Joseph S (eds) Biochar for environmental management—science and technology. Earthscan, London, pp 33–52
Barker DJ, Sullivan CY, Moser LE (1993) Water deficit effects on osmotic potential, cell wall elasticity, and proline in five forage grasses. Agron J 85:270–275
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Blackwell P, Riethmuller G, Collins M (2009) Biochar application to soil. In: Lehmann J, Joseph S (eds) Biochar for environmental management: science and technology. Earthscan, London, pp 207–226
Chan KY, Van Zwieten L, Meszaros I, Downie A, Joseph S (2007) Agronomic values of greenwaste biochar as a soil amendment. Aust J Soil Res 45:629–634. doi:10.1071/SR07109
Chan KY, Van Zwieten L, Meszaros I, Downie A, Joseph S (2008) Using poultry litter biochars as soil amendments. Aust J Soil Res 46:437–444. doi:10.1071/SR08036
Chan KY, Xu Z (2009) Biochar: nutrient properties and their enhancement. In: Lehmann J, Joseph S (eds) Biochar for environmental management: science and technology. Earthscan, London, pp 67–84
Cheng C-H, Lehmann J, Thies JE, Burton AJ, Engelhard M (2006) Oxidation of black carbon by biotic and abiotic processes. Org Geochem 37:1477–1488
Cheng C-H, Lehmann J, Thies JE, Burton SD (2008) Stability of black carbon in soils across a climatic gradient. J Geophys Res 113: doi:10.1029/2007JG000642
Clough TJ, Condron LM (2010) Biochar and the nitrogen cycle: Introduction. J Environ Qual 39:1218–1223.
Cotrufo MF, Ineson P, Scott A (1998) Elevated CO2 reduces the nitrogen concentration of plant tissues. Global Change Biol 4:43–54
Downie A, Crosky A, Munroe P (2009) Physical properties of biochar. In: Lehmann J, Joseph S (eds) Biochar for environmental management—science and technology. Earthscan, London, pp 13–32
Elad Y, David DR, Harel YM, Borenshtein M, Kalifa HB, Silber A, Graber ER (2010) Induction of systemic resistance in plants by biochar, a soil-applied carbon sequestering agent. Phytopathology 100:913–921
Galwey NW (1989) Quinoa. Biologist 36:5
Gaskin JW, Speir RA, Harris K, Das KC, Lee RD, Morris LA, Fisher DS (2010) Effect of peanut hull and pine chip biochar on soil nutrients, corn nutrient status, and yield. Agron J 102:623–633
Gaunt JL, Lehmann J (2008) Energy balance and emissions associated with biochar sequestration and pyrolysis bioenergy production. Environ Sci Technol 42:4152–4158
Glaser B (2007) Prehistorically modified soils of central Amazonia: a model for sustainable agriculture in the twenty-first century. Phil Trans R Soc London B 362:187–196
Glaser B, Haumaier L, Guggenberger G, Zech W (2001) The ‘Terra Preta’ phenomenon: a model for sustainable agriculture in the humid tropics. Naturwissenschaften 88:37–41
Glaser B, Lehmann J, Zech W (2002) Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal—a review. Biol Fertil Soils 35:219–230
Golluscio RA (2007) On the link between nitrogen productivity and residence time: two opposite nitrogen use strategies? J Arid Environ 68:165–169
Gonzalez JA, Gallardo M, Hila LM, Rosa M, Prado FE (2009) Physiological responses of quinoa (Chenopodium quinoa Willd.) to drought and waterlogging stresses: dry matter partitioning. Bot Stud 50:35–42
Graber ER, Harel YM, Kolton M, Cytryn E, Silber A, David DR, Tsechansky L, Borenshtein M, Elad Y (2010) Biochar impact on development and productivity of pepper and tomato grown in fertigated soilless media. Plant and Soil 337:481–496
Granier F (1988) Extraction of plant proteins for two-dimensional electrophoresis. Electrophoresis 9:112–718
Granli T, Bøckmann OC (1994) Nitrous oxide from agriculture. Norweg J Agr Sci Supp 12:1–128
Groffman PM, Tiedje JM (1991) Relationships between denitrification, CO2 production and air-filled porosity in soils of different texture and drainage. Soil Biol Biochem 23(3):299–302
Hansen J, Sato M, Kharecha P, Beerling D, Berner R, Masson-Delmotte V, Pagani M, Raymo M, Royer DL, Zachos JC (2008) Target atmospheric CO2: where should humanity aim? Open Atm Sci J 2:217–231
Hutchinson GL, Mosier AR (1981) Improved soil cover method for field measurement of nitrous oxide fluxes. Soil Sci Soc Am J 45:311–316
Ibarra-Caballero J, Villanueva-Verduz C, Molina-Galan J, Sanchez-de-Jimenez E (1988) Proline accumulation as a symptom of drought stress in maize: a tissue differentiation requirement. J Exp Bot 39:889–897
IPCC (2007a) Climate change 2007: Climate change impacts, adaptation and vulnerability. Working Group II Contribution to the Intergovernmental Panel on Climate Change Fourth Assessment Report—Summary for Policymakers. IPCC, Bern
IPCC (2007b) Climate change 2007: the physical science basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge
Ishida H, Nishimori Y, Sugisawa M, Makino A, Mae T (1997) The large subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase is fragmented. Plant Cell Physiol 38:471–479
Jacobsen S-E, Stølen O (1993) Quinoa—morphology and phenology and prospects for its production as a new crop in Europe. Eur J Agron 2:19–29
Jensen CR, Jacobsen S-E, Andersen MN, Núñez N, Andersen SD, Rasmussen L, Mogensen VO (2000) Leaf gas exchange and water relation characteristics of field quinoa (Chenopodium quinoa Willd.) during soil drying. Eur J Agron 13:11–25
Kim J-S, Sparovek G, Longo RM, De Melo WJ, Crowley D (2007) Bacterial diversity of terra preta and pristine forest soil from the Western Amazon. Soil Biol Biochem 39:684–690
Kimetu JM, Lehmann J, Kinyangi JM, Cheng CH, Thies J, Mugendi DN, Pell A (2009) Soil organic C stabilization and thresholds in C saturation. Soil Biol Biochem 41:2100–2104
Kolb SE, Fermanich KJ, Dornbush ME (2009) Effect of charcoal quantity on microbial biomass and activity in temperate soils. Soil Sci Soc Am J 73:1173–1181. doi:10.2136/sssaj2008.0232
Kuzyakov Y, Subbotina I, Chen H, Bogomolova I, Xu X (2009) Black carbon decomposition and incorporation into soil microbial biomass estimated by 14C labeling. Soil Biol Biochem 41:210–219. doi:10.1016/j.soilbio.2008.10.016
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 27:680–685
Laird DA, Brown RC, Amonette JE, Lehmann J (2009) Review of the pyrolysis platform for coproducing bio-oil and biochar. Biofuels Bioprod Biorefin 3:547–562
Lal R (2004) Soil carbon sequestration impacts on global climate change and food security. Science 304:1623–1627. doi:10.1126/science.1097396
Lal R (2009) Challenges and opportunities in soil organic matter research. Eur J Soil Sci 60:158–169
Lehmann J (2006) Bio-char sequestration in terrestrial ecosystems—a review. Mitigat Adaptat Strat Glob Chan 11:403–427. doi:10.1007/s11027-005-9006-5
Lehmann J (2007a) Bio-energy in the black. Front Ecol Environ 5:381–387
Lehmann J (2007b) A handful of carbon. Nature 447:143–144
Lehmann J, Czimczik C, Laird D, Sohi S (2009) Stability of biochar in soil. In: Lehmann J, Joseph S (eds) Biochar for environmental management—science and technology. Earthscan, London, pp 183–205
Liang B, Lehmann J, Sohi SP, Thies JE, O'Neill B, Trujillo L, Gaunt J, Solomon D, Grossman J, Neves EG, Luizão FJ (2010) Black carbon affects the cycling of non-black carbon in soil. Org Geochem. doi:10.1016/j.orggeochem.2009.09.007
Liang B, Lehmann J, Solomon D, Kinyangi J, Grossman J, O'Neill B, Skjemstad JO, Thies J, Luizao FJ, Petersen J, Neves EG (2006) Black carbon increases cation exchange capacity in soils. Soil Sci Soc Am J 70:1719–1730. doi:10.2136/sssaj2005.0383
Loftfield N, Flessa H, Augustin J, Beese F (1997) Automated gas chromatographic system for rapid analysis of the atmospheric trace gases methane, carbon dioxide, and nitrous oxide. J Environ Qual 26:560–564
Long SP, Zhu X-G, Naidu SL, Ort DR (2006) Can improvement in photosynthesis increase crop yields? Plant Cell Environ 29:315–330
Major J, Lehmann J, Rondon M, Goodale C (2010) Fate of soil-applied black carbon: downward migration, leaching and soil respiration. Global Change Biol 16:1366–1379. doi:10.1111/j.1365-2486.2009.02044.x
Marris E (2006) Putting the carbon back: black is the new green. Nature 442:624–626
Mathews JA (2008) How carbon credits could drive the emergence of renewable energies. Energ Pol 36:3633–3639
McHenry MP (2009) Agricultural bio-char production, renewable energy generation and farm carbon sequestration in Western Australia: certainty, uncertainty and risk. Agric Ecosys Environ 129:1–7
Morgan JA, Pataki DE, Körner C, Clark H, Del Grosso SJ, Grünzweig JM, Knapp AK, Mosier AR, Newton PCD, Niklaus PA, Nippert JB, Nowak RS, Parton WJ, Polley HW, Shaw MR (2004) Water relations in grassland and desert ecosystems exposed to elevated atmospheric CO2. Oecologia 140:11–25
Mosier AR, Mack L (1980) Gas chromatographic system for precise, rapid analysis of nitrous oxide. Soil Sci Soc Am J 44:1121–1123
Nösberger J, Long SP, Norby RJ, Stitt M, Hendrey G, Blum H (2006) Managed ecosystems and CO2: case studies, processes, and perspectives, vol 187. Ecological studies. Springer, Berlin, pp 457
Novak JM, Busscher WJ, Watts DW, Laird DA, Ahmedna MA, Niandou MAS (2010) Short-term CO2 mineralization after additions of biochar and switchgrass to a Typic Kandiudult. Geoderma 154:281–288
Nowak RS, Ellsworth DS, Smith SD (2004) Functional responses of plants to elevated atmospheric CO2—do photosynthetic and productivity data from FACE experiments support early predictions? New Phytol 162:253–280
Owensby CE, Ham JM, Knapp AK, Auen LM (1999) Biomass production and species composition change in a tallgrass prairie ecosystem after long-term exposure to elevated atmospheric CO2. Global Change Biol 5:497–506
Reich PB, Tjoelker MG, Machado J-L, Oleksyn J (2006) Universal scaling of respiratory metabolism, size and nitrogen in plants. Nature 439:457–461
Rondon M, Lehmann J, Ramírez J, Hurtado M (2007) Biological nitrogen fixation by common beans (Phaseolus vulgaris L.) increases with bio-char additions. Biol Fertil Soils 43:699–708
Schulte PJ, Brooks JR (2003) Branch junctions and the flow of water through xylem in Douglas-fir and ponderosa pine stems. J Exp Bot 54:1597–1605. doi:10.1093/jxb/erg169
Smith MS, Tiedje JM (1979) The effect of roots on soil denitrification. Soil Sci Soc Am J 43:951–955. doi:10.2136/sssaj1979.03615995004300050027x
Sperry JS, Hacke UG (2002) Desert shrub water relations with respect to soil characteristics and plant functional type. Funct Ecol 16:367–378
Spokas KA, Koskinen WC, Baker JM, Reicosky DC (2009) Impacts of woodchip biochar additions on greenhouse gas production and sorption/degradation of two herbicides in a Minnesota soil. Chemosphere 77:574–581
Spokas KA, Baker JM, Reicosky DC (2010) Ethylene: potential key for biochar amendment impacts. Plant Soil 333:443–452
Steiner C, Glaser B, Teixeira WG, Lehmann J, Blum WEH, Zech W (2008) Nitrogen retention and plant uptake on a highly weathered central Amazonian Ferralsol amended with compost and charcoal. J Plant Nutr Soil Sci 171:893–899. doi:10.1002/jpln.200625199
Steiner C, Teixeira M, Zech W (2007) Soil respiration curves as soil fertility indicators in perennial central Amazonian plantations treated with charcoal, and mineral or organic fertilisers. Trop Sci 47:218–230. doi:10.1002/ts.216
Stitt M, Krapp A (1999) The interaction between elevated carbon dioxide and nitrogen nutrition: the physiological and molecular background. Plant Cell Environ 22:583–621
Taghizadeh-Toosi A, Clough TJ, Condron LM, Sherlock RR, Anderson CR, Craigie RA (2011) Biochar incorporation into pasture soil suppresses in situ nitrous oxide emissions from ruminant urine patches. J Environ Qual, in press (open access) https://www.soils.org/publications/jeq/view/40-42/q10-0419.pdf
van Zwieten L, Singh B, Joseph S, Kimber S, Cowie A, Chan KY (2009) Biochar and emissions of non-CO2 greenhouse gases from soil. In: Lehmann J, Joseph S (eds) Biochar for environmental management—science and technology. Earthscan, London, pp 227–249
van Zwieten L, Kimber S, Morris S, Downie A, Berger E, Rust J, Scheer C (2010) Influence of biochars on flux of N2O and CO2 from Ferrosol. Aust J Soil Res 48:555–568
Wardle DA, Nilsson M-C, Zackrisson O (2008) Fire-derived charcoal causes loss of forest humus. Science 320:629. doi:10.1126/science.1154960
Yanai Y, Toyota K, Okazaki M (2007) Effects of charcoal addition on N2O emissions from soil resulting from rewetting air-dried soil in short-term laboratory experiments. Soil Sci Plant Nutr 53:181–188
Acknowledgements
The authors want to thank Christoph Forreiter for critical reading of the manuscript and Judy Libra for proof reading. The authors acknowledge the technical assistance of Nicol Strasilla and Gerlinde Lehr with proline and RuBisCO extractions and greenhouse gas analyses and Gerhard Mayer for his assistance at the ion-chromatograph. Thanks to Johanna Kreiling for technical assistance, and to the Department of Applied Microbiology, in particular to Stefan Ratering, for help with the GC analyses.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Johannes Lehmann.
Rights and permissions
About this article
Cite this article
Kammann, C.I., Linsel, S., Gößling, J.W. et al. Influence of biochar on drought tolerance of Chenopodium quinoa Willd and on soil–plant relations. Plant Soil 345, 195–210 (2011). https://doi.org/10.1007/s11104-011-0771-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-011-0771-5