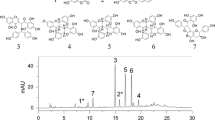
Abstract
Panax quinquefolium (American ginseng) is a perennial understory herb that has been widely used as a medicinal plant in China and other countries. Autotoxicity has been reported to be one of the major problems hindering the consecutive cultivation of American ginseng. However, the potential autotoxic compounds produced by the root of American ginseng are less well known. Here, we report the isolation and characterization of five groups of autotoxic compounds from aqueous extracts of the fibrous roots of American ginseng. Ether extracts of the water-soluble compounds were further analyzed and separated into seven fractions. Among them, the most autotoxic fraction (Fraction V) was subjected to GC/MS analysis, and 44 compounds were identified. Based on literature information, 14 individual compounds were selected and their autotoxic effects on seedling growth were further tested. The results revealed that, of these 14 compounds, 9 phenolic compounds significantly reduced the growth of seedlings in a concentration-dependent manner, while 5 aliphatic compounds showed modest inhibition at all three concentrations tested. Furthermore, we verified the existence of the autotoxic compounds in the plow layer soil of commercially cultivated American ginseng fields, and the concentration of these compounds as determined by HPLC analysis was in line with the concentration determined to be bioactive. Taken together, our study established a functional link between the compounds produced by American ginseng and their autotoxic effects.

Similar content being viewed by others
References
Banwart WL, Porter PM, Granato TC, Hassett JJ (1985) HPLC separation and wavelength area ratios of more than 50 phenolic acids and flavonoids. J Chem Ecol 11:383–395 doi:10.1007/BF01411424
Bertin C, Yang X, Weston LA (2003) The role of root exudates and allelochemicals in the rhizosphere. Plant Soil 256:67–83 doi:10.1023/A:1026290508166
Blum U, Gerig TM (2005) Relationships between phenolic acid concentrations, transpiration, water utilization, leaf area expansion, and uptake of phenolic acids: nutrient culture studies. J Chem Ecol 31:1907–1932 doi:10.1007/s10886-005-5934-5
Boggs KW, Story JM (1987) The population age structure of spotted knapweed (Centaurea maculosa) in Montana. Weed Sci 35:194–198
Canals RM, Emeterio LS, Peralta J (2005) Autotoxicity in Lolium rigidum: analyzing the role of chemically mediated interactions in annual plant populations. J Theor Biol 235:402–407 doi:10.1016/j.jtbi.2005.01.020
Chon SU, Choi SK, Jung S, Jang HG, Pyo BS, Kim SM (2002) Effects of alfalfa leaf extracts and phenolic allelochemicals on early seedling growth and root morphology of alfalfa and barnyard grass. Crop Prot 21:1077–1082 doi:10.1016/S0261-2194(02)00092-3
Chon SU, Kim JD (2002) Biological activity and quantification of suspected allelochemicals from alfalfa plant parts. J Agron Crop Sci 188:281–285 doi:10.1046/j.1439-037X.2002.00574.x
Dalton BR (1999) The occurrence and behavior of plant phenolic acids in soil environment and their potential involvement in allelochemical interference interactions: methodological limitations in establishing conclusive proof of allelopathy. CRC, Boca Raton
Dalton BR, Weed SB, Blum U (1987) Plant phenolic acids in soils: a comparison of extraction procedures. Soil Sci Soc Am J 51:1515–1521
Einhellig FA (1996) Interactions involving allelopathy in cropping systems. Agron J 88:886–893
Ervin GN, Wetzel RG (2000) Allelochemical autotoxicity in the emergent wetland macrophyte Juncus effusus (Juncaceae). Am J Bot 87:853–860 doi:10.2307/2656893
Kalinova J, Vrchotova N, Triska J (2007) Exudation of allelopathic substances in buckwheat (Fagopyrum esculentum Moench). J Agric Food Chem 55:6453–6459 doi:10.1021/jf070795u
Kong C, Hu F, Xu T, Lu Y (1999) Allelopathic potential and chemical constituents of volatile oil from ageratum conyzoides. J Chem Ecol 25:2347–2356 doi:10.1023/A:1020882109682
Machrafi Y, Prevost D, Beauchamp CJ (2006) Toxicity of phenolic compounds extracted from bark residues of different ages. J Chem Ecol 32:2595–2615 doi:10.1007/s10886-006-9157-1
Mahall BE, Callaway RM (1992) Root communication mechanisms and intracommunity distributions of two mojave desert shrubs. Ecology 73:2145–2151 doi:10.2307/1941462
Mahfuzur R, Zamir KP (2007) Biological control of damping-off on American ginseng (Panax quinquefolius) by Clonostachys rosea f. catenulata (=Gliocladium catenulatum). Can J Plant Pathol 29:203–207
Perry LG, Thelen GC, Ridenour WM, Weir TL, Callaway RM, Paschke MW, Vivanco JM (2005) Dual role for an allelochemical: (+/−)-catechin fromCentaurea maculosa root exudates regulates conspecific seedling establishment. J Ecol 93:1126–1135 doi:10.1111/j.1365-2745.2005.01044.x
Porter PM, Banwart WL, Hassett JJ (1985) HPLC isolation and GC-MS identification of genistein, daidzein, and coumestrol from unhydrolyzed soybean root extracts. Environ Exp Bot 25:229–232 doi:10.1016/0098-8472(85)90006-1
Rice EL (1984) Allelopathy. Academic, Orlando
Seal AN, Pratley JE, Haig T, An M (2004) Identification and quantitation of compounds in a series of allelopathic and non-allelopathic rice root exudates. J Chem Ecol 30:1647–1662 doi:10.1023/B:JOEC.0000042074.96036.14
Sène M, Gallet C, Doré T (2001) Phenolic compounds in a sahelian sorghum (Sorghum bicolor) genotype (CE145–66) and associated soils. J Chem Ecol 27:81–92 doi:10.1023/A:1005620000835
Singh HP, Batish DR, Kohli RK (1999) Autotoxicity: concept, organisms, and ecological significance. Crit Rev Plant Sci 18:757–772 doi:10.1080/07352689991309478
Weller DM, Raaijmakers JM, Gardener BBM, Thomashow LS (2002) Microbial populations responsible for specific soil suppressiveness to plant pathogens. Annu Rev Phytopathol 40:309–348 doi:10.1146/annurev.phyto.40.030402.110010
Whitehead DC, Dibb H, Hartley RD (1981) Extractant pH and the release of phenolic compounds from soils, plant roots and leaf litter. Soil Biol Biochem 13:343–348 doi:10.1016/0038-0717(81)90074-2
Whitehead DC, Dibb H, Hartley RD (1983) Bound phenolic compounds in water extracts of soils, plant roots and leaf litter. Soil Biol Biochem 15:133–136 doi:10.1016/0038-0717(83)90092-5
Zamir KP (1997) Fungal pathogens of American ginseng (Panax quinquefolium) in British Colombia. Can J Plant Pathol 19:301–306
Zhao YJ, Wang YP, Shao D, Yang JS, Liu D (2005a) Autotoxicity of Panax quinquefolium L. Allelophathy J 15:67–74
Zhao YJ, Wang YP, Yang JS, Liu D (2005b) A study on the rotation of crops among Panax quinquefolium, Perilla frutescens and Coix lacryma-jobi (in Chinese with English abstract). Zhongguo Zhong Yao Za Zhi 30:12–15
Acknowledgments
The authors are grateful to the staff of the Department of Medicinal Analysis, Institute of Materia Medica, Chinese Academy of Medical Sciences and Peking Union Medical College, for performing EI-MS, NMR, and thank Dr. Xiang-ning Jiang, the College of Biological Sciences and Biotechnology, Beijing Forestry University, for analysis of GC-MS. This work was supported by the Beijing Natural Science Foundation (6072025), Beijing Science and technology project (D0206001040591) of China and the National Key Technology R&D Program (2006BA109B03).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Petra Marschner.
Rights and permissions
About this article
Cite this article
He, C.N., Gao, W.W., Yang, J.X. et al. Identification of autotoxic compounds from fibrous roots of Panax quinquefolium L.. Plant Soil 318, 63–72 (2009). https://doi.org/10.1007/s11104-008-9817-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-008-9817-8