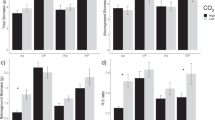
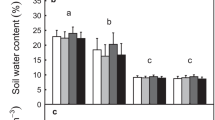
Abstract
Photosynthetic stimulation by elevated [CO2] is largely regulated by nitrogen and phosphorus availability in the soil. During a 6 year Free Air CO2 Enrichment (FACE) experiment with poplar trees in two short rotations, inorganic forms of soil nitrogen, extractable phosphorus, microbial and total nitrogen were assessed. Moreover, in situ and potential nitrogen mineralization, as well as enzymatic activities, were determined as measures of nutrient cycling. The aim of this study was to evaluate the effects of elevated [CO2] and fertilization on: (1) N mineralization and immobilization processes; (2) soil nutrient availability; and (3) soil enzyme activity, as an indication of microbial and plant nutrient acquisition activity. Independent of any treatment, total soil N increased by 23% in the plantation after 6 years due to afforestation. Nitrification was the main process influencing inorganic N availability in soil, while ammonification being null or even negative. Ammonium was mostly affected by microbial immobilization and positively related to total N and microbial biomass N. Elevated [CO2] negatively influenced nitrification under unfertilised treatment by 44% and consequently nitrate availability by 30% on average. Microbial N immobilization was stimulated by [CO2] enrichment and probably enhanced the transformation of large amounts of N into organic forms less accessible to plants. The significant enhancement of enzyme activities under elevated [CO2] reflected an increase in nutrient acquisition activity in the soil, as well as an increase of fungal population. Nitrogen fertilization did not influence N availability and cycling, but acted as a negative feed-back on phosphorus availability under elevated CO2.










Similar content being viewed by others
References
Allen AS, Andrews JA, Finzi AC, Matamala R, Richter DD, Schlesinger WH (2000) Effects of Free Air CO2 Enrichment (FACE) on belowground processes in a Pinus taeda forest. Ecol Appl 10:437–448
Anderson JM, Ingram JSI (1993) Colorimetric determination of ammonium. In: Anderson JM, Ingram JSI (eds) Tropical soil biology and fertility, a handbook of methods, 2nd edn. Anderson, CAB International, Wallingford, UK, pp 73–74
Bandick AK, Dick RP (1999) Field management effects on soil enzyme activities. Soil Biol Biochem 31:1471–1479
BassiriRad H, Griffin KL, Reynolds JF, Strain BR (1997) Changes in root \({\text{NH}}_4 ^ + \) and \({\text{NO}}_3 ^ - \) absorption rates of loblolly and ponderosa pine in response to CO2 enrichment. Plant Soil 190:1–9
Calfapietra C, Gielen B, Sabatti M, De Angelis P, Scarascia-Mugnozza G, Ceulemans R (2001) Growth performance of Populus exposed to “free air carbon dioxide enrichment” during the first growing season in the POPFACE experiment. Ann For Sci 58:819–828
Calfapietra C, Gielen B, Galema ANJ, Lukac M, De Angelis P, Moscatelli MC, Ceulemans R, Scarascia-Mugnozza G (2003) Free-air CO2 enrichment (FACE) enhances biomass production in a short-rotation poplar plantation. Tree Physiol 23:805–814
Calfapietra C, De Angelis P, Gielen B, Lukac M, Moscatelli MC, Avino G, Lagomarsino A, Polle A, Ceulemans R, Scarascia Mugnozza G, Hoosbeek MR, Cotrufo MF (2007) Increased nitrogen-use efficiency of a short-rotation poplar plantation in elevated CO2 concentration. Tree Physiol 27:1153–1163
Cardon G (1996) Influence of rhizodepositions under elevated CO2 on plant nutrition and soil organic matter. Plant Soil 187:277–288
Carnol M, Hogenboom L, Jach ME, Remacle J, Ceulemans R (2002) Elevated atmospheric CO2 in open top chambers increases net nitrification and potential denitrification. Glob Change Biol 8:590–598
Cataldo DA, Haroon M, Schrader LE, Young V (1975) Rapid colorimetric determination of nitrate in plant tissue by nitration of salycilic acid. Commun Soil Sci Plan 6:71–80
Cotrufo MF, Ineson P, Scott A (1998) Elevated CO2 reduces the nitrogen concentration of plant tissues. Glob Change Biol 4:43–54
Cotrufo MF, De Angelis P, Polle A (2005) Leaf litter production and decomposition in a poplar short-rotation coppice exposed to free air CO2 enrichment (POPFACE). Glob Change Biol 11:1–12
De Lucia EH, Callaway RM, Thomas EM, Schlesinger WH (1997) Mechanisms of phosphorus acquisition for ponderosa pine seedlings under high CO2 and temperature. Ann Bot 79:111–120
Demetz M, Insam H (1999) Phosphorus availability in a forest soil determined with a respiratory assay compared to chemical methods. Geoderma 89:259–271
Dick WA, Tabatabai MA (1993) Significance and potential use of soil enzymes. In: Blaine F (ed) Soil microbial ecology. Marcel Dekker, NY
Elsgaard L, Andersen GH, Eriksen J (2002) Measurement of arylsulphatase activity in agricultural soils using a simplified assay. Soil Biol Biochem 34:79–82
Eno CF (1960) Nitrate production in the field by incubating the soil in polyethylene bags. Soil Sci Soc Am Proc 24:277–299
Finzi AC, Norby RJ, Calfapietra C, Gallet-Budynek A, Gielen B, Holmes WE, Hoosbeek MR, Iversen CM, Jackson RB, Kubiske ME, Ledford J, Liberloo M, Oren R, Polle A, Pritchard S, Zak DR, Schlesinger WH, Ceulemans R (2007) Increases in nitrogen uptake rather than nitrogen-use efficiency support higher rates of temperate forest productivity under elevated CO2. P Natl Acad Sci 104(35):14014–14019
Garzillo AMV, Badalucco L, De Cesare F, Grego S, Buonocore V (1996) Synthesis and characterization of an acid phosphatase-polyresorcinol complex. Soil Biol Biochem 28:1155–1161
Godbold DL, Hoosbeek MR, Lukac M, Cotrufo MF, Janssens IA, Ceulemans R, Polle A, Velthorst EJ, Scarascia-Mugnozza G, De Angelis P, Miglietta F, Peressotti A (2006) Mycorrhizal hyphal turnover as a dominant process for carbon input into soil organic matter. Plant Soil 281:15–24
Harley PC, Thomas RB, Reynolds JF, Strain BR (1992) Modelling of photosynthesis of cotton grown in elevated CO2. Plant Cell Environ 15:271–282
Haynes RJ (1986) The decomposition process: mineralization, immobilization, humus formation and degradation. In: Haynes RJ (ed) Mineral nitrogen in the plant–soil system. Academic, Orlando, pp 55–126
Hoosbeek MR, Lukac M, van Dam D, Godbold DL, Velthorst EJ, Biondi FA, Peressotti A, Cotrufo MF, De Angelis P, Scarascia Mugnozza G (2004) More new carbon in the mineral soil of a poplar plantation under Free Air Carbon Enrichment (POPFACE): cause of increased priming effect? Glob Biogeochem Cycles 18, GB1040, DOI 10.1029/2003GB00212
Hoosbeek MR, Li Y, Scarascia Mugnozza G (2006) Free atmospheric CO2 enrichment (FACE) increased labile and total carbon in the mineral soil of a short rotation Poplar plantation. Plant Soil 281:247–254
Hopkins DW, O’Donnell AG, Shiel RS (1988) The effect of fertilisation on soil nitrifier activity in experimental grassland plots. Biol Fertil Soils 5:344–349
Hungate BA, Dijkstra P, Johnson DW, Hinkle CR, Drake B (1999) Elevated CO2 increases nitrogen fixation and decreases soil nitrogen mineralization in Florida scrub oak. Glob Chang Biol 5:1–9
Janssens IA, Crookshanks M, Taylor G, Ceulemans R (1998) Elevated atmospheric CO2 increases fine root production, respiration, rhizosphere respiration and soil CO2 efflux in Scots pine seedlings. Glob Chang Biol 4:871–878
Joergensen RG, Brookes PC (1990) Ninhydrin-reactive nitrogen measurements of microbial biomass in 0.5 M K2SO4 soil extracts. Soil Biol Biochem 22:1023–1027
Joergensen RG, Anderson TH, Wolters V (1995) C and N relationships of the soil microbial biomass in soils of beech (Fagus sylvatica L.) forests. Biol Fert Soils 19:141–147
Jonasson S, Michelsen A, Schmidt IK, Nielsen EV, Callaghan TV (1996) Microbial biomass C, N and P in two arctic soils and responses to addition of NPK fertilizer and sugar: implications for plant nutrient uptake. Oecologia 106:507–515
Lagomarsino A, Moscatelli MC, De Angelis P, Grego S (2006) Labile substrate quality as the main driving force of microbial mineralization activity in a poplar plantation soil under elevated CO2 and nitrogen fertilization. Sci Total Environ 372:256–265
Lewis JD, Strain BR (1996) The role of mycorrhizas in the response of Pinus taeda seedlings to elevated CO2. New Phytol 133:431–443
Liberloo M, Gielen B, Calfapietra C, Veys C, Pigliacelli R, Scarascai-Mugnozza G, Ceulemans R (2004) Growth of a poplar short rotation coppice under elevated atmospheric CO2 concentrations (EUROFACE) depends on fertilization and species. Ann For Sci 61:299–307
Liberloo M, Calfapietra C, Lukac M, Godbold D, Luo ZB, Polle A, Hoosbeek MR, Kull O, Marek M, Raine C, Rubino M, Taylor G, Scarascia-Mugnozza G, Ceulemans R (2006) Woody biomass production during the second rotation of a bio-energy Populus plantation increases in a future high CO2 world. Glob Chang Biol 12:1094–1106
Liberloo M, Tulva I, Raïm O, Kull O, Ceulemans R (2007) Photosynthetic stimulation under long-term CO2 enrichment and fertilization is sustained across a closed Populus canopy profile (EUROFACE). New Phytol 173:537–549
Lukac M, Calfapietra C, Godbold DL (2003) Root production and turnover in Populus grown under elevated CO2 using a free air enrichment system POPFACE. Glob Change Biol 9:838–848
Magill AH, Aber JD (2000) Variation in soil net mineralization rates with dissolved organic carbon dditions. Soil Biol Biochem 32:597–601
Marinari S, Calfapietra C, De Angelis P, Scarascia Mugnozza G, Grego S (2006) Impact of elevated CO2 and nitrogen fertilization on foliar elemental composition in a short rotation poplar plantation. Environ Pollut 47:507–515
Miglietta F, Peressotti A, Vaccari FP, Zaldei A, De Angelis P, Scarascia-Mugnozza G (2001) Free-air CO2 enrichment (FACE) of a poplar plantation: the POPFACE fumigation system. New Phytol 150:465–476
Miller M, Palojarvi A, Rangger A, Reeslev M, Kjioller A (1998) The use of fluorogenic substrates to measure fungal presence and activity in soil. Appl Environ Microbiol 64:613–617
Morgan A, Knight WG, Dudley LM, Hunt HW (1994) Enhanced root system C-sink activity, water relations and aspects of nutrient acquisition in mycotrophic Bouteloua gracilis subjected to CO2 enrichment. Plant Soil 165:139–146
Moscatelli MC, Lagomarsino A, De Angelis P, Grego S (2005a) Seasonality of soil biological properties in a poplar plantation growing under elevated CO2. Appl Soil Ecol 30:162–173
Moscatelli MC, Lagomarsino A, De Angelis P, Grego S (2005b) Soil microbial indices as bioindicators of environmental changes in a poplar plantation. Ecol Indic 5:171–179
Olsen SR, Sommers LE (1982) Phosphorous. In: Page AL, Miller RH, Keeney DR (eds) Methods of soil analysis. Part 2, chemical and microbiological properties. Am Soc Agron, Madison, Wisconsin (USA), pp 403–430
Parham JA, Deng SP (2000) Detection, quantification and characterization of b-glucosaminidase activity in soil. Soil Biol Biochem 32:1183–1190
Phillips RL, Zak DR, Holmes WE, White DC (2002) Microbial community composition and function beneath temperate trees exposed to elevated atmospheric carbon dioxided and ozone. Oecologia 131:236–244
Rothstein DE, Zak DR, Pregitzer KS, Curtis PS (2000) Kinetics of nitrogen uptake by Populus tremuloides in relation to atmospheric CO2 and soil nitrogen availability. Tree Physiol 20:265–270
Rutherford PM, Juma NG (1992) Influence of soil texture on protozoa-induced mineralization of bacterial carbon and nitrogen. Can J Soil Sci 72:183–200
Schimel JP, Firestone MK (1989) Nitrogen incorporation and flow through a coniferous forest soil profile. Soil Biol Biochem 53:779–784
Sinsabaugh RL, Moorhead DL (1994) Resource allocation to extracellular enzyme production: a model for nitrogen and phosphorus control of litter decomposition. Soil Biol Biochem 26:1305–1311
Syvertsen JP, Graham JH (1999) Phosphorus supply and arbuscular mycorrhizas increase growth and net gas exchange responses of two Citrus spp. grown at elevated [CO2]. Plant Soil 208:209–219
Tabatabai MA, Bremner JM (1970) Arylsulfatase activity of soils. Soil Sci Soc Am J 34:225–229
Trehan SP (1996) Immobilization of \(^{15} {\text{NH}}_4 ^ + \) in three soils by chemical and biological processes. Soil Biol Biochem 28:1021–1027
Treseder KK (2004) A meta-analysis of mycorrhizal responses to nitrogen, phosphorus, and atmospheric CO2 in field studies. New Phytol 164:347–355
Treseder KK, Allen MF (2000) Research review: mycorrhizal fungi have a potential role in soil carbon storage under elevated CO2 and nitrogen deposition. New Phytol 147:189–200
Turner J, Olson PR (1976) Nitrogen relations in a Douglas-fir plantation. Ann Bot 40:1185–1193
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass C. Soil Biol Biochem 19:703–707
Van Groenigen K, Harris D, Horwath WR, Hartwig UA, Van Kessel C (2002) Linking sequestration of 13C and 15N in aggregates in a pasture soil following 8 years of elevated atmospheric CO2. Glob Chang Biol 8:1094–1108
Van Lagen B (1996) Soil analyses. In: Buurman P, Van Lagen B, Velthorst EJ (eds) Manual for soil and water analyses. Backhuys Publishers, Leiden, The Netherlands, pp 1–120
Verstraete W (1981) Nitrification in agricultural systems. Call for control. In: Clark FE, Roswall T (eds) Terrestrial nitrogen cycles. Processes, ecosystems strategies and management impacts. Ecological Bulletin (Stockholm), pp 565–572
Zak DR, Pregitzer KS, Curtis PS, Teeri JA, Fogel R, Randlett DL (1993) Elevated atmospheric CO2 and feedback between carbon and nitrogen cycles. Plant Soil 151:103–116
Zak DR, Pregitzer KS, King JS, Holmes WE (2000) Elevated atmospheric CO2, fine roots and the response of soil microorganism: a review and hypothesis. New Phytol 147:201–222
Acknowledgements
The authors are grateful to Prof. Giuseppe Scarascia Mugnozza coordinator of EU EUROFACE (EVR1-CT-2002-40027) and MIUR Centre of Excellence “Forests and climate” projects for allowing the use of POP-EuroFACE experimental station.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Barbara Wick.
Rights and permissions
About this article
Cite this article
Lagomarsino, A., Moscatelli, M.C., Hoosbeek, M.R. et al. Assessment of soil nitrogen and phosphorous availability under elevated CO2 and N-fertilization in a short rotation poplar plantation. Plant Soil 308, 131–147 (2008). https://doi.org/10.1007/s11104-008-9614-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-008-9614-4