Abstract
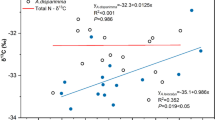
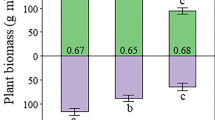
Belowground biomass is a critical factor regulating ecosystem functions of coastal marshes, including soil organic matter (SOM) accumulation and the ability of these systems to keep pace with sea-level rise. Nevertheless, belowground biomass responses to environmental and vegetation changes have been given little emphasis marsh studies. Here we present a method using stable carbon isotopes and color to identify root and rhizomes of Schoenoplectus americanus (Pers.) Volk. ex Schinz and R. Keller (C3) and Spartina patens (Ait.) Muhl. (C4) occurring in C3− and C4-dominated communities in a Chesapeake Bay brackish marsh. The functional significance of the biomass classes we identified is underscored by differences in their chemistry, depth profiles, and variation in biomass and profiles relative to abiotic and biotic factors. C3 rhizomes had the lowest concentrations of cellulose (29.19%) and lignin (14.43%) and the lowest C:N (46.97) and lignin:N (0.16) ratios. We distinguished two types of C3 roots, and of these, the dark red C3 roots had anomalously high C:N (195.35) and lignin:N (1.14) ratios, compared with other root and rhizome classes examined here and with previously published values. The C4-dominated community had significantly greater belowground biomass (4119.1 g m−2) than the C3-dominated community (3256.9 g m−2), due to greater total root biomass and a 3.6-fold higher C3-root:rhizome ratio in the C4-dominated community. C3 rhizomes were distributed significantly shallower in the C4-dominated community, while C3 roots were significantly deeper. Variability in C3 rhizome depth distributions was explained primarily by C4 biomass, and C3 roots were explained primarily by water table height. Our results suggest that belowground biomass in this system is sensitive to slight variations in water table height (across an 8 cm range), and that the reduced overlap between C3 and C4 root profiles in the C4-dominated community may account for the greater total root biomass observed in that community. Given that future elevated atmospheric CO2 and accelerated sea-level rise are likely to increase C3 abundance in Atlantic and Gulf coast marshes, investigations that quantify how patterns of C3 and C4 belowground biomass respond to environmental and biological factors stand to improve our understanding of ecosystem-wide impacts of global changes on coastal wetlands.
Similar content being viewed by others
Abbreviations
- SOM:
-
soil organic matter
- C:
-
carbon
- CO2:
-
carbon dioxide
- N:
-
nitrogen
- δ13C:
-
13C abundance relative to standard Peedee Belemnite
- SERC:
-
Smithsonian Environmental Research Center
References
J D Aber J M Melillo K J Nadelhoffer C A McClaugherty J Pastor (1985) ArticleTitleFine root turnover in forest ecosystems in relation to quantity and form of nitrogen availability – a comparison of 2 methods Oecologia 66 317–321 Occurrence Handle10.1007/BF00378292
W J Arp (1991) Vegetation of a North American Salt Marsh and Elevated Atmospheric Carbon Dioxide University of Amsterdam Amsterdam
W J Arp B G Drake W T Pockman P S Curtis D F Whigham (1993) ArticleTitleInteractions between C3 and C4 salt-marsh plant species during 4 years of exposure to elevated etmospheric CO2 Vegetatio 104 133–143 Occurrence Handle10.1007/BF00048149
A S Ball B G Drake (1997) ArticleTitleShort-term decomposition of litter produced by plants grown in ambient and elevated atmospheric CO2 concentrations Global Change Biol. 3 29–35
F A Bazzaz (1990) ArticleTitleThe responses of natural ecosystems to the rising global CO2 levels Annu. Rev. Ecol. Syst. 21 167–196 Occurrence Handle10.1146/annurev.es.21.110190.001123
R Benner A E Maccubbin R E Hodson (1984) ArticleTitlePreparation, characterization, and microbial-degradation of specifically radiolabelled C-14 lignicelluloses from marine and fresh-water macrophytes Appl. Environ. Microbiol. 47 381–389 Occurrence Handle1:CAS:528:DyaL2cXhtVSitLY%3D Occurrence Handle16346477
M D Bertness A M Ellison (1987) ArticleTitleDeterminants of pattern in a New England salt marsh plant community Ecol. Monogr. 57 129–147
P M Bradley J T Morris (1992) ArticleTitleEffect of salinity on the critical nitrogen concentration of Spartina alterniflora Loisel Aquat. Bot. 43 149–161 Occurrence Handle10.1016/0304-3770(92)90040-P
S D Bridgham S P Faulkner C J Richardson (1991) ArticleTitleSteel rod oxidation as a hydrologic indicator in wetland soils Soil Sci. Soc. of Am. J. 55 856–862
S W Broome I A Mendelssohn K L Mckee (1995) ArticleTitleRelative growth of Spartina patens (Ait) Muhl and Scirpus olneyi Gray occurring in a mixed stand as affected by salinity and flooding depth Wetlands 15 20–30
D M Burdick (1989) ArticleTitleRoot aerenchyma development in Spartina patens in response to flooding Am. J. Bot. 76 777–780
D M Burdick I A Mendelssohn (1990) ArticleTitleRelationship between anatomical and metabolic responses to soil waterlogging in the coastal grass Spartina patens J. Exp. Bot. 41 223–228 Occurrence Handle1:CAS:528:DyaK3cXhslOntL0%3D
D M Burdick I A Mendelssohn K L McKee (1989) ArticleTitleLive standing crop and metabolism of the marsh grass Spartina patens as related to edaphic factors in a brackish, mixed marsh community in Louisiana Estuaries 12 195–204
Y Choi Y Wang Y P Hsieh L Robinson (2001) ArticleTitleVegetation succession and carbon sequestration in a coastal wetland in northwest Florida: Evidence from carbon isotopes Global Biogeochem. Cycles 15 311–319 Occurrence Handle10.1029/2000GB001308 Occurrence Handle1:CAS:528:DC%2BD3MXktlWhsLc%3D
L H Comas D M Eissenstat A N Lakso (2000) ArticleTitleAssessing root death and root system dynamics in a study of grape canopy pruning New Phytol. 147 171–178 Occurrence Handle10.1046/j.1469-8137.2000.00679.x Occurrence Handle1:CAS:528:DC%2BD3cXms1yltLw%3D
H Coops N Geilen G Vandervelde (1994) ArticleTitleDistribution and growth of the helophyte species Phragmites australis and Scirpus lacustris in water depth gradients in relation to wave exposure Aquat. Bot. 48 273–284 Occurrence Handle10.1016/0304-3770(94)90020-5
H Coops G Vandervelde (1996) ArticleTitleEffects of waves on helophyte stands: Mechanical characteristics of stems of Phragmites australis and Scirpus lacustris Aquat. Bot. 53 175–185
P S Curtis B G Drake D F Whigham (1989) ArticleTitleNitrogen and carbon dynamics in C3 and C4 estuarine marsh plants grown under elevated CO2 in situ Oecologia 78 297–301
P S Curtis L M Balduman B G Drake D F Whigham (1990) ArticleTitleElevated atmospheric CO2 effects on belowground processes in C3 and C4 estuarine marsh communities Ecology 71 2001–2006
B G Drake (1992) ArticleTitleA field study of the effects of elevated CO2 on ecosystem processes in a Chesapeake Bay wetland Aust. J. Bot. 40 579–595 Occurrence Handle10.1071/BT9920579 Occurrence Handle1:CAS:528:DyaK3sXht1WitLs%3D
Drake B G, Arp W, Curtis P S, Leadley P W, Sager J and Whigham D 1986 Effects of Elevated CO2 on Chesapeake Bay Wetlands. I. Description of the Study Site. United States Department of Energy, Carbon Dioxide Research Division Report Number 034, Office of Energy Research, Washington, DC
B G Drake G Peresta E Beugeling R Matamala (1996) Long-term elevated CO2 exposure in a Chesapeake Bay wetland: Ecosystem gas exchange, primary production, and tissue nitrogen G W Koch H A Mooney (Eds) Carbon Dioxide and Terrestrial Ecosystems Academic Press San Diego
J R Ehleringer R F Sage L B Flanagan R W Pearcy (1991) ArticleTitleClimate change and the evolution of C4 photosynthesis Trends Ecol. Evol. 6 95–99 Occurrence Handle10.1016/0169-5347(91)90183-X
S M L Ewe L D Sternberg (2003) ArticleTitleSeasonal gas exchange characteristics of Schinus terebinthifolius in a native and disturbed upland community in Everglades National Park, Florida Forest Ecol. Manag. 179 27–36
J L Gallagher F G Plumley (1979) ArticleTitleUnderground biomass profiles and productivity in Atlantic coastal marshes Am. J. Bot. 66 156–161
W S Gordon R B Jackson (2000) ArticleTitleNutrient concentrations in fine roots Ecology 81 275–280
T E Jordan J W Pierce D F Correll (1986) ArticleTitleFlux of particulate matter in the tidal marshes and subtidal shallows of the Rhode River estuary Estuaries 9 310–319
M S Kearney R E Grace J C Stevenson (1988) ArticleTitleMarsh loss in Nanticoke estuary, Chesapeake Bay Geogr. Rev. 78 205–220
U Krauss J W Deacon (1994) ArticleTitleRoot turnover of groundnut (Arachis hypogaea L.) in soil tubes Plant Soil 166 259–270 Occurrence Handle10.1007/BF00008339 Occurrence Handle1:CAS:528:DyaK2MXjsl2jurw%3D
P W Leadley B G Drake (1993) ArticleTitleOpen top chambers for exposing plant canopies to elevated CO2 concentration and for measuring net gas exchange Vegetatio 104 3–15 Occurrence Handle10.1007/BF00048141
G Lin L D L Sternberg (1992) ArticleTitleEffect of growth form, salinity, nutrient and sulfide on photosynthesis, carbon isotope discrimination and growth of red mangrove (Rhizophora-mangle L) Aust. J. Plant. Physiol. 19 509–517 Occurrence Handle1:CAS:528:DyaK3sXjs1Wjug%3D%3D
W J Mitsch J G Gosselink (1993) Wetlands Van Nostrand Reinhold, New York New York 722
J T Morris W B Bowden (1986) ArticleTitleA mechanistic, numerical model of sedimentation, mineralization, and decomposition for marsh sediments Soil Sci. Soc. of Am. J. 50 96–105 Occurrence Handle1:CAS:528:DyaL28Xht1yqsrk%3D
G Naidoo K L Mckee I A Mendelssohn (1992) ArticleTitleAnatomical and metabolic responses to waterlogging and salinity in Spartina alterniflora and S. patens (Poaceae) Am. J. Bot. 79 765–770 Occurrence Handle1:CAS:528:DyaK38Xls1Oqsbk%3D
C E Owensby J M Ham A K Knapp L M Auen (1999) ArticleTitleBiomass production and species composition change in a tallgrass prairie ecosystem after long-term exposure to elevated atmospheric CO2 Global Change Biol. 5 497–506 Occurrence Handle10.1046/j.1365-2486.1999.00245.x
R T Parrondo J G Gosselink C S Hopkinson (1978) ArticleTitleEffects of salinity and drainage on the growth of three salt marsh grasses Bot. Gaz. 139 102–107 Occurrence Handle10.1086/336975 Occurrence Handle1:CAS:528:DyaE1cXktVGhtbo%3D
D Pont J W Day P Hensel E Franquet F Torre P Rioual C Ibanez E Coulet (2002) ArticleTitleResponse scenarios for the deltaic plain of the Rhone in the face of an acceleration in the rate of sea-level rise with special attention to Salicornia-type environments Estuaries 25 337–358
J Pozo R Colino (1992) ArticleTitleDecomposition processes of Spartina maritima in a salt marsh of the Basque Country Hydrobiologia 231 165–175 Occurrence Handle10.1007/BF00018200 Occurrence Handle1:CAS:528:DyaK38XktVKqurs%3D
D P Rasse G Peresta B G Drake (2005) ArticleTitleSeventeen years of elevated CO2 exposure in a Chesapeake Bay Wetland: sustained but contrasting responses of plant growth and CO2 uptake Global Change Biol. 11 369–377
Ross W M, Chabreck R H 1972 Factors affecting the growth and survival of natural and planted stands of Scirpus olneyi. Proceedings of the Annual Conference, Southeastern Association of Game and Fish Commissioners. 26, 178–188
J M Rybczyk J C Callaway J W Day (1998) ArticleTitleA relative elevation model for a subsiding coastal forested wetland receiving wastewater effluent Ecol. Modell. 112 23–44 Occurrence Handle10.1016/S0304-3800(98)00125-2 Occurrence Handle1:CAS:528:DyaK1cXmt1Ojtbs%3D
C J Saunders (2003) Soil Accumulation in a Chesapeake Bay Salt Marsh: Modeling 500 Years of Global Change, Vegetation Change, and Rising Atmospheric CO2 Duke University Durham, North Carolina
D Scavia J C Field D F Boesch R W Buddemeier V Burkett D R Cayan M Fogarty M A Harwell R W Howarth C Mason D J Reed T C Royer A H Sallenger J G Titus (2002) ArticleTitleClimate change impacts on US coastal and marine ecosystems Estuaries 25 149–164
R A Scheffer R Aerts (2000) ArticleTitleRoot decomposition and soil nutrient and carbon cycling in two temperate fen ecosystems Oikos 91 541–549 Occurrence Handle10.1034/j.1600-0706.2000.910316.x
M Schwarzbeck (1982) Competition for belowground space in salt marsh plants Johns Hopkins University Baltimore, Maryland
D M Seliskar (1985) ArticleTitleMorphometric variations of five tidal marsh halophytes along environmental gradients Am. J. Bot. 72 1340–1352
D M Seliskar (1988) ArticleTitleWaterlogging stress and ethylene production in the dune slack plant, Scirpus americanus J. Exp. 39 1639–1648 Occurrence Handle1:CAS:528:DyaL1MXhsFWns70%3D
D M Seliskar (1990) ArticleTitleThe role of waterlogging and sand accretion in modulating the morphology of the dune slack plant Scirpus americanus Can. J. Bot. 68 1780–1787
I Valiela J M Teal S D Allen R Vanetten D Goehringer S Volkmann (1985) ArticleTitleDecomposition in salt-marsh ecosystems – the phases and major factors affecting disappearance of above-ground organic-matter J. Exp. Mar. Biol. Ecol. 89 26–54 Occurrence Handle10.1016/0022-0981(85)90080-2
F J Vernberg (1993) ArticleTitleSalt-marsh processes – a review Environ. Toxicol. Chem. 12 2167–2195
E C Webb I A Mendelssohn (1996) ArticleTitleFactors affecting vegetation dieback of an oligohaline marsh in coastal Louisiana: field manipulation of salinity and submergence Am. J. Bot. 83 1429–1434
D S White B L Howes (1994) ArticleTitleTranslocation, remineralization, and turnover of nitrogen in the roots and rhizomes of Spartina alterniflora (Gramineae) Am. J. Bot. 81 1225–1234 Occurrence Handle1:CAS:528:DyaK2MXitFWjsr8%3D
R K Wieder S T Starr (1998) ArticleTitleQuantitative determination of organic fractions in highly organic, Sphagnum peat soils Comm. Soil Sci. Plant Anal. 29 847–857 Occurrence Handle1:CAS:528:DyaK1cXktVKktbs%3D
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saunders, C.J., Megonigal, J.P. & Reynolds, J.F. Comparison of Belowground Biomass in C3- and C4-Dominated Mixed Communities in a Chesapeake Bay Brackish Marsh. Plant Soil 280, 305–322 (2006). https://doi.org/10.1007/s11104-005-3275-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11104-005-3275-3