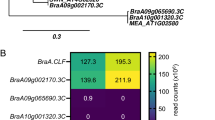
Abstract
Key message
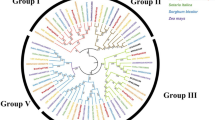
A genome-wide analysis identified 116 NAC genes in rose, including stress-related ones with different expression patterns under drought and salt stress. Silencing of RcNAC091, a member of the ATAF subfamily, decreased dehydration tolerance in rose.
The NAC (NAM, ATAF, and CUC) transcription factors (TFs) are plant-specific proteins that regulate various developmental processes and stress responses. However, knowledge of the NAC TFs in rose (Rosa chinensis), one of the most important horticultural crops, is limited. In this study, 116 NAC genes were identified from the rose genome and classified into 16 subfamilies based on protein phylogenetic analysis. Chromosomal mapping revealed that the RcNAC genes were unevenly distributed on the seven chromosomes of rose. Gene structure and motif analysis identified a total of ten conserved motifs, of which motifs 1–7 were highly conserved and present in most rose NACs, while motifs 8–10 were present only in a few subfamilies. Further study of the stress-related RcNACs based on the transcriptome data showed differences in the expression patterns among the organs, at various floral developmental stages, and under drought and salt stress in rose leaves and roots. The stress-related RcNACs possessed cis-regulatory elements (CREs) categorized into three groups corresponding to plant growth and development, phytohormone response, and abiotic and biotic stress response. Reverse transcription-quantitative real-time PCR (RT-qPCR) analysis of 11 representative RcNACs revealed their differential expression in rose leaves and roots under abscisic acid (ABA), polyethylene glycol (PEG), and sodium chloride (NaCl) treatments. Furthermore, the silencing of RcNAC091 verified its role in positively regulating the dehydration stress response. Overall, the present study provides valuable insights into stress-related RcNACs and paves the way for stress tolerance in rose.







Similar content being viewed by others
Data availability
Enquiries about data availability should be directed to the authors
References
Aron MB, Lu S, Anderson JB, Farideh C, Derbyshire MK, Carol D, Fong JH, Geer LY, Geer RC, Gonzales NR, Gwadz M, Hurwitz DI, Jackson JD, Ke Z, Lanczycki CJ, Lu F, Marchler GH, Mullokandov M, Omelchenko MV, Robertson CL, Song JS, Thanki N, Yamashita RA, Zhang D, Zhang N, Zheng C, Bryant SH (2011) CDD: a conserved domain database for the functional annotation of proteins. Nucleic Acids Res 39:D225–D229. https://doi.org/10.1093/nar/gkq1189
Bailey TL, Boden M, Buske FA, Frith M, Grant CE, Clementi L, Ren J, Li WW, Noble WS (2009) MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res 37:W202–W208. https://doi.org/10.1093/nar/gkp335
Bo X, Misato O, Masatoshi Y, Kiminori T, Mayumi W, Mayuko S, Minoru K, Yoshimi N, Ryosuke S, Hi Y, Takashi M, Tetsuya K, Arata Y, Ko K, Mitsuyasu H, Taku D (2014) Contribution of NAC transcription factors to plant adaptation to land. Sci 343:1505–1508. https://doi.org/10.1126/science.1248417
Cenci A, Guignon V, Roux N, Rouard M (2014) Genomic analysis of NAC transcription factors in banana (Musa acuminata) and definition of NAC orthologous groups for monocots and dicots. Plant Mol Biol Rep 85:63–80. https://doi.org/10.1007/s11103-013-0169-2
Chen C, Chen H, Zhang Y, Thomas H, Frank M, He Y, Xia R (2020) TBtools: an integrative toolkit developed for interactive analyses of big biological data. Mol Plant. https://doi.org/10.1101/289660
Dai F, Zhang C, Jiang X, Kang M, Yin X, Lü P, Zhang X, Zheng Y, Gao J (2012) RhNAC2 and RhEXPA4 are involved in the regulation of dehydration tolerance during the expansion of rose petals. Plant Physiol 160:2064–2082. https://doi.org/10.1104/pp.112.207720
Dai W, Wang M, Gong X, Liu JH (2018) The transcription factor FcWRKY40 of Fortunella crassifolia functions positively in salt tolerance through modulation of ion homeostasis and proline biosynthesis by directly regulating SOS2 and P5CS1 homologs. New Phytol 219:972–989. https://doi.org/10.1111/nph.15240
Dar NA, Amin I, Wani W, Wani SA, Shikari AB, Wani SH, Masoodi KZ (2017) Abscisic acid: a key regulator of abiotic stress tolerance in plants. Plant Gene 11:106–111. https://doi.org/10.1016/j.plgene.2017.07.003
Diao W, John S, Wang S, Liu J, Pan B, Guo G, Ge W, Mohammad D, Ali SH (2018) Genome-wide analyses of the NAC transcription factor gene family in pepper (Capsicum annuum L.): chromosome location, phylogeny, structure, expression patterns, cis-elements in the promoter, and interaction network. Int J Mol Sci 19:1048–1061. https://doi.org/10.3390/ijms19041028
Ding A, Li S, Li W, Hao Q, Wan X, Wang K, Liu Q, Liu Q, Jiang Q (2019) RhNAC31, a novel rose NAC transcription factor, enhances tolerance to multiple abiotic stresses in Arabidopsis. Acta Physiol Plant. https://doi.org/10.1007/s11738-019-2866-1
Dong X, Jiang Y, Yang Y, Xiao Z, Bai X, Gao J, Tan S, Yoonkang H, Hao S, He F (2019) Identification and expression analysis of the NAC gene family in coffea canephora. Agron 9:670. https://doi.org/10.3390/agronomy9110670
Dudhate A, Shinde H, Yu P, Tsugama D, Gupta SK, Liu S, Takano T (2021) Comprehensive analysis of NAC transcription factor family uncovers drought and salinity stress response in pearl millet (Pennisetum glaucum). BMC Genom. https://doi.org/10.21203/rs.3.rs-37445/v3
Fábio TN, Paulo SS, Sandra RC, Jorge HF, De Rosa V, Pompermayer P, Arruda P (2005) SsNAC23, a member of the NAC domain protein family, is associated with cold, herbivory and water stress in sugarcane. Plant Sci 169:93–106. https://doi.org/10.1016/j.plantsci.2005.03.008
Fang Y, You J, Xie K, Xie W, Xiong L (2008) Systematic sequence analysis and identification of tissue-specific or stress-responsive genes of NAC transcription factor family in rice. Mol Genet Genomics 280:547–563. https://doi.org/10.1007/s00438-008-0386-6
Finn RD, Alex B, Jody C, Penelope C, Eberhardt RY, Eddy SR, Andreas H, Kirstie H, Liisa H, Jaina M (2014) Pfam: the protein families database. Nucleic Acids Res 42:D222–D230. https://doi.org/10.1093/nar/gkt1223
Fujita M, Fujita Y, Maruyama K, Seki M, Hiratsu K, Ohme-Takagi M, Tran LP, Yamaguchi-Shinozaki K, Shinozaki K (2004) A dehydration-induced NAC protein, RD26, is involved in a novel ABA-dependent stress-signaling pathway. Plant J 39:863–876. https://doi.org/10.1111/j.1365-313X.2004.02171.x
Gong L, Zhang H, Liu X, Gan X, Nie F, Yang W, Zhang L, Chen Y, Song Y, Zhang H (2020) Ectopic expression of HaNAC1, an ATAF transcription factor from Haloxylon ammodendron, improves growth and drought tolerance in transgenic Arabidopsis. Plant Physiol Bioch 151:535–544. https://doi.org/10.1016/j.plaphy.2020.04.008
Guérin C, Roche J, Allard V, Ravel C, Mouzeya S, Bouzidi MF (2019) Genome-wide analysis, expansion and expression of the NAC family under drought and heat stresses in bread wheat (T. aestivum L.). PLoS One 14:e0213390. https://doi.org/10.1371/journal.pone.0213390
Guo X, Yu C, Luo L, Wan H, Zhen N, Xu T, Tan J, Pan H, Zhang Q (2017) Transcriptome of the floral transition in Rosa chinensis ‘Old Blush.’ BMC Genom 18:199. https://doi.org/10.1186/s12864-017-3584-y
Han Y, Yu J, Zhao T, Cheng T, Zhang Q (2019) Dissecting the genome-wide evolution and function of R2R3-MYB transcription factor family in Rosa chinensis. Genes 10:823. https://doi.org/10.3390/genes10100823
Han D, Du M, Zhou Z, Wang S, Li T, Han J, Xu T, Yang G (2020) An NAC transcription factor gene from Malus baccata, MbNAC29, increases cold and high salinity tolerance in Arabidopsis. In Vitro Cell Dev-Pl 56:588–599. https://doi.org/10.1007/s11627-020-10105-9
Jiang X, Zhang C, Lü P, Jiang G, Liu X, Dai F, Gao J (2014) RhNAC3, a stress-associated nac transcription factor, has a role in dehydration tolerance through regulating osmotic stress-related genes in rose petals. Plant Biotechnol J 12:38–48. https://doi.org/10.1111/pbi.12114
Jin H, Huang F, Cheng H, Song H, Yu D (2013) Overexpression of the GmNAC2 gene, an NAC transcription factor, reduces abiotic stress tolerance in tobacco. Plant Mol Biol Rep 31:435–442. https://doi.org/10.1007/s11105-012-0514-7
Jin J, Tian F, Yang D, Meng Y, Kong L, Luo J, Gao G (2017) PlantTFDB 4.0: toward a central hub for transcription factors and regulatory interactions in plants. Nucleic Acids Res 45:D1040–D1045. https://doi.org/10.1093/nar/gkw1328
Jung S, Ficklin SP, Lee T, Cheng CH, Blenda A, Zheng P, Yu J, Bombarely A, Cho I, Ru S, Evans K, Peace C, Abbott AG, Mueller LA, Olmstead MA, Main D (2014) The genome database for rosaceae (GDR): year 10 update. Nucleic Acids Res 42:D1237–D1244. https://doi.org/10.1093/nar/gkt1012
Kevin LH, Bruno C, Nishadi DS, Gareth M, Wasiu A, James A, Jorge A, Matthieu B, Dan MB, Lahcen C, Manuel C, Marc C, Mikkel C, Carla C, Alayne C, Paul D, Silvie F, Astri G, Nancy G, Laurent G, Parul G, Kim EH, Erin H, Sarah EH, Pankaj J, Sophie HJ, Paul JK, Nick L, Uma M, Thomas M, Mark DM, Ben M, Matthieu M, Guy N, Sushma N, Andrew O, Irene P, Mateus P, Michael P, Helder P, Emily P, Justin P, Marc R, Matthew R, Vasily S, Daniel MS, Joshua S, Marcela KT, Stephen JT, Martin U, Sharon W, Doreen W, Gary W, Andrew DY, Paul F (2020) Ensembl genomes 2020—enabling non-vertebrate genomic research. Nucleic Acids Res 48:D689–D695. https://doi.org/10.1093/nar/gkz890
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Laloum T, Martín G, Duque P (2018) Alternative splicing control of abiotic stress responses. Trends in Plant Sci 23:P140-150. https://doi.org/10.1016/j.tplants.2017.09.019
Le DT, Nishiyama R, Watanabe Y, Mochida K, Yamaguchi-Shinozaki K, Shinozaki K, Tran LP (2011) Genome-wide survey and expression analysis of the plant-specific NAC transcription factor family in soybean during development and dehydration stress. DNA Res 18:263–276. https://doi.org/10.1093/dnares/dsr015
Lescot M, Déhais P, Thijs G, Marchal K, Moreau Y, Van de Peer Y, Rouzé P, Rombauts S (2020) PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res 30:325–327. https://doi.org/10.1093/nar/30.1.325
Li W, Li X, Chao J, Zhang Z, Wang W, Guo Y (2018) NAC family transcription factors in tobacco and their potential role in regulating leaf senescence. SRA. https://doi.org/10.3389/fpls.2018.01900
Li W, Geng Z, Zhang C, Wang K, Jiang X (2021a) Whole-genome characterization of Rosa chinensis AP2/ERF transcription factors and analysis of negative regulator RcDREB2B in Arabidopsis. BMC Genom 22:90. https://doi.org/10.1186/s12864-021-07396-6
Li W, Fu L, Geng Z, Zhao X, Liu Q, Jiang X (2021b) Physiological characteristic changes and full-length transcriptome of rose (Rosa chinensis) roots and leaves in response to drought stress. Plant Cell Physiol 61:2153–2166. https://doi.org/10.1093/pcp/pcaa137
Livak KJ, Schmittgen DT (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lu P, Chen N, Rui A, Su Z, Qi B, Ren F, Chen J, Wang X (2007) A novel drought-inducible gene, ATAF1, encodes a nac family protein that negatively regulates the expression of stress-responsive genes in Arabidopsis. Plant Mol Biol Rep 63:289–305. https://doi.org/10.1007/s11103-006-9089-8
Mehari TG, Xu Y, Magwanga RO, Umer MJ, Shiraku ML, Hou Y (2021) Identification and functional characterization of GhD01g0514 (GhNAC072) transcription factor in response to drought stress tolerance in cotton. Plant Biol 166:361–375. https://doi.org/10.1016/j.plaphy.2021.05.050
Mohanta TK, Yadav D, Khan A, Hashem A, Tabassum B, Khan AL, Abd-Allah EF, Al-Harrasi A (2020) Genomics, molecular and evolutionary perspective of NAC transcription factors. PLoS One 15:e0231425. https://doi.org/10.1371/journal.pone.0231425
Nakashima K, Takasaki H, Mizoi J, Shinozakicd K, Yamaguchi-Shinozakiab K (2012) NAC transcription factors in plant abiotic stress responses. Biochim Biophys Acta Biomembr 1819:97–103. https://doi.org/10.1016/j.bbagrm.2011.10.005
Nicholas KB (1997) GeneDoc: analysis and visualization of genetic variation. Embnew News 4:14
Nuruzzaman M, Manimekalai R, Sharoni AM, Satoh K, Kondoh H, Ooka H, Kikuchi S (2010) Genome-wide analysis of NAC transcription factor family in rice. Gene 465:30–44. https://doi.org/10.1016/j.gene.2010.06.008
Ohnishi T, Sugahara S, Yamada T, Kikuchi K, Yoshiba Y, Hirano HY, Tsutsumi N (2005) OsNAC6, a member of the NAC gene family, is induced by various stresses in rice. Genes Genet Syst 80:135–139. https://doi.org/10.1266/ggs.80.135
Ooka H, Satoh K, Doi K, Nagata T, Otomo Y, Murakami K, Matsubara K, Osato N, Kawai J, Carninci P, Hayashizaki Y, Suzuki K, Kojima K, Takahara Y, Yamamoto K, Kikuchi S (2003) Comprehensive analysis of NAC family genes in Oryza sativa and Arabidopsis thaliana. DNA Res 10:239–247. https://doi.org/10.1093/dnares/10.6.239
Panu A, Manohar J, Konstantin A, Delphine B, Gabor C, Edouard C, Séverine D, Volker F, Arnaud F, Elisabeth G, Aurélien G, Céline H, Vassilios I, Dmitry K, Robin L, Sébastien M, Khaled M, Nicole R, Grégoire R, Ioannis X, Heinz S (2012) ExPASy: SIB bioinformatics resource portal. Nucleic Acids Res 40:W597–W603. https://doi.org/10.1093/nar/gks400
Pei H, Ma N, Tian J, Luo J, Chen J, Li J, Zheng Y, Chen X, Fei Z, Gao J (2013) An NAC transcription factor controls ethylene-regulated cell expansion in flower petals. Plant Physiol 163:775–791. https://doi.org/10.1104/pp.113.223388
Prakash A, Jeffryes M, Bateman A, Finn R (2017) The hmmer web server for protein sequence similarity search. Curr Protoc Bioinformatics 60:3.15.1-3.15.23. https://doi.org/10.1002/cpbi.40
Puranik S, Sahu PP, Srivastava P, Prasad M (2012) NAC proteins: regulation and role in stress tolerance. Trends in Plant Sci 17:369–381. https://doi.org/10.1016/j.tplants.2012.02.004
Shao H, Wang H, Tang X (2015) NAC transcription factors in plant multiple abiotic stress responses: progress and prospects. Front Plant Sci 6:902. https://doi.org/10.3389/fpls.2015.00902
Shiriga K, Sharma R, Kumar K, Yadavc SK, Hossaina F, Thirunavukkarasua N (2014) Genome-wide identification and expression pattern of drought-responsive members of the NAC family in maize. Meta Gene 2:407–417. https://doi.org/10.1016/j.mgene.2014.05.001
Singh KB, Foley RC, Oñate-Sánchez L (2002) Transcription factors in plant defense and stress responses. Curr Opin Plant Biol 5:430–436. https://doi.org/10.1016/S1369-5266(02)00289-3
So HA, Lee JH (2019) NAC transcription factors from soybean (Glycine max L.) differentially regulated by abiotic stress. J Plant Biol 62:147–160. https://doi.org/10.1007/s12374-018-0285-2
Su H, Zhang S, Yuan X, Chen C, Wang X, Hao Y (2013) Genome-wide analysis and identification of stress-responsive genes of the NAM-ATAF1,2-CUC2 transcription factor family in apple. Plant Physiol Bioch 71:11–21. https://doi.org/10.1016/j.plaphy.2013.06.022
Tan J, Wang J, Luo L, Yu C, Xu T, Wu Y, Cheng T, Wang J, Pan H, Zhang Q (2017) Genetic relationships and evolution of old Chinese garden roses based on SSRs and chromosome diversity. Sci Rep 7(1):1–10. https://doi.org/10.1038/s41598-017-15815-6
Tian X, Wang Z, Zhang Q, Ci H, Wang P, Yu L, Jia G (2018) Genome-wide transcriptome analysis of the salt stress tolerance mechanism in Rosa chinensis. PLoS One 13:e0200938. https://doi.org/10.1371/journal.pone.0200938
Wang Y, Tang H, DeBarry JD, Tan X, Li J, Wang X, Lee T, Jin H, Marler B, Guo H, Kissinger JD, Andrew HP (2012) MCScanX: a toolkit for detection and evolutionary analysis of gene synteny and collinearity. Nucleic Acids Res 40:e49. https://doi.org/10.1093/nar/gkr1293
Wang N, Zheng Y, Xin H, Fang L, Li S (2013) Comprehensive analysis of NAC domain transcription factor gene family in Vitis vinifera. Plant Cell Rep 32:61–75. https://doi.org/10.1007/s00299-012-1340-y
Wang B, Guo X, Wang C, Ma J, Niu F, Zhang H, Yang B, Liang W, Han F, Jiang Y (2015) Identification and characterization of plant-specific NAC gene family in canola (Brassica napus L.) reveal novel members involved in cell death. Plant Mol Biol Rep 87:395–411. https://doi.org/10.1007/s11103-015-0286-1
Yang X, Wang X, Ji L, Yi Z, Fu C, Ran J, Hu R, Zhou G (2015) Overexpression of a Miscanthus lutarioriparius NAC gene MlNAC5 confers enhanced drought and cold tolerance in Arabidopsis. Plant Cell Rep 34:943–958. https://doi.org/10.1007/s00299-015-1756-2
Yang Z, Nie G, Feng G, Han J, Huang L, Zhang X (2021) Genome-wide identification, characterization, and expression analysis of the NAC transcription factor family in orchardgrass (Dactylis glomerata L.). BMC Genom 22:178. https://doi.org/10.1186/s12864-021-07485-6
Yoshihiro N, Kazuo N, Zabta KS, Yoh S, Takashi F, Hiroshi A, Mari N, Kazuo S, Kazuko Y (2003) Interaction between two cis-acting elements, ABRE and DRE, in ABA-dependent expression of Arabidopsis rd29A gene in response to dehydration and high-salinity stresses. Plant J 34:137–148. https://doi.org/10.1046/j.1365-313X.2003.01708.x
Acknowledgements
The research was supported by the National Key Research and Development Program of China (No. 2018YFD1000400) and the Innovative Program for Graduate Students of Qingdao Agricultural University (No. QNYCX21085). The authors would like to thank TopEdit (www.topeditsci.com) for linguistic assistance during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
JX conceived and designed the experiments; GL, SL, and FL carried out the experiments; GL and LS conducted the data analysis. JX and GL wrote the manuscript; ZJ and LQ revised the manuscript and contributed the plant material. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Geng, L., Su, L., Fu, L. et al. Genome-wide analysis of the rose (Rosa chinensis) NAC family and characterization of RcNAC091. Plant Mol Biol 108, 605–619 (2022). https://doi.org/10.1007/s11103-022-01250-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-022-01250-3