Abstract
Key message
PSBR1 is a moso bamboo gene negatively regulated by brassinosteroid, which encodes a mitochondrial localized protein. Overexpression of PSBR1 leads to growth inhibition in various growth progresses in Arabidopsis.
Abstract
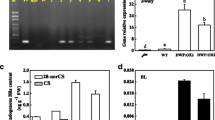
The young shoot of moso bamboo (Phyllostachys edulis) is known as one of the fastest growing plant organs. The roles of phytohormones in the fast-growth of bamboo shoot are not fully understood. Brassinosteroids (BRs) are a group of growth-promoting steroid hormones that play important roles in cell elongation and division. While BR related genes are highly enriched in fast-growing internodes in moso bamboo, the functions of BR in the fast-growth process is not understood at the molecular level. Here, we identified a poaceae specific gene, PSBR1 (Poaceae specific and BR responsive gene 1) from the moso bamboo genome. PSBR1 was highly expressed in the stem and leaves of bamboo seedling, and the elongating nodes of fast-growing bamboo shoot. PSBR1′s expression is increased by BR biosynthesis inhibitor propiconazole but decreased by BR treatment. PSBR1 encodes a novel protein that is localized to the mitochondria in tobacco and bamboo protoplast. The Arabidopsis transgenic plants overexpressing PSBR1 show growth inhibition in both vegetative and reproductive stages. This study suggests that PSBR1 is a BR regulated mitochondrial protein in bamboo, which inhibits plant growth when overexpressed in Arabidopsis.






Similar content being viewed by others
References
Arendsee ZW, Li L, Wurtele ES (2014) Coming of age: orphan genes in plants. Trends Plant Sci 19(11):698–708
Bekh-Ochir D, Shimada S, Yamagami A, Kanda S, Ogawa K, Nakazawa M, Matsui M, Sakuta M, Osada H, Asami T, Nakano T (2013) A novel mitochondrial DnaJ/Hsp40 family protein BIL2 promotes plant growth and resistance against environmental stress in brassinosteroid signaling. Planta 237(6):1509–1525
Bohni PC, Daum G, Schatz G (1983) Import of proteins into mitochondria. Partial purification of a matrix-located protease involved in cleavage of mitochondrial precursor polypeptides. J Biol Chem 258(8):4937–4943
Chien L-F, Wu Y-C, Chen H-P (2011) Mitochondrial energy metabolism in young bamboo rhizomes from Bambusa oldhamii and Phyllostachys edulis during shooting stage. Plant Physiol Biochem 49(4):449–457
Clough SJ, Bent AF (1998) Floral dip: a simplified method for agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16(6):735–743
Clouse SD, Sasse JM (1998) Brassinosteroids: essential regulators of plant growth and development. Annu Rev Plant Biol 49(1):427–451
Cui K, He C-Y, Zhang J-G, Duan A-G, Zeng Y-F (2012) Temporal and spatial profiling of internode elongation-associated protein expression in rapidly growing culms of bamboo. J Proteome Res 11(4):2492–2507
Deng X-G, Zhu T, Zhang D-W, Lin H-H (2015) The alternative respiratory pathway is involved in brassinosteroid-induced environmental stress tolerance in Nicotiana benthamiana. J Exp Bot 66(20):6219–6232
Divi UK, Krishna P (2009) Brassinosteroid: a biotechnological target for enhancing crop yield and stress tolerance. N Biotechnol 26(3–4):131–136
Earley KW, Haag JR, Pontes O, Opper K, Juehne T, Song KM, Pikaard CS (2006) Gateway-compatible vectors for plant functional genomics and proteomics. Plant J 45(4):616–629
Engler C, Youles M, Gruetzner R, Ehnert T-M, Werner S, Jones JDG, Patron NJ, Marillonnet S (2014) A golden gate modular cloning toolbox for plants. ACS Synth Biol 3(11):839–843
Fan C, Ma J, Guo Q, Li X, Wang H, Lu M (2013) Selection of reference genes for quantitative real-time PCR in bamboo (Phyllostachys edulis). PLoS ONE 8:e565732
Feher A, Otvos K, Pasternak TP, Szandtner AP (2008) The involvement of reactive oxygen species (ROS) in the cell cycle activation (G(0)-to-G(1) transition) of plant cells. Plant Signal Behav 3(10):823–826
Fu J (2001) Chinese moso bamboo: its importance. Bamboo 22(5):5–7
Gamuyao R, Nagai K, Ayano M, Mori Y, Minami A, Kojima M, Suzuki T, Sakakibara H, Higashiyama T, Ashikari M (2017) Hormone distribution and transcriptome profiles in bamboo shoots provide insights on bamboo stem emergence and growth. Plant Cell Physiol 58(4):702–716
Gao J, Zhang Y, Zhang C, Qi F, Li X, Mu S, Peng Z (2014) Characterization of the floral transcriptome of Moso bamboo (Phyllostachys edulis) at different flowering developmental stages by transcriptome sequencing and RNA-seq analysis. PLoS ONE 9:e989106
Hartwig T, Corvalan C, Best NB, Budka JS, Zhu J-Y, Choe S, Schulz B (2012) Propiconazole is a specific and accessible brassinosteroid (BR) biosynthesis inhibitor for Arabidopsis and maize. PLoS ONE 7(5):e36625
Hawlitschek G, Schneider H, Schmidt B, Tropschug M, Hartl FU, Neupert W (1988) Mitochondrial protein import: identification of processing peptidase and of PEP, a processing enhancing protein. Cell 53(5):795–806
Isagi Y, Kawahara T, Kamo K, Ito H (1997) Net production and carbon cycling in a bamboo Phyllostachys pubescens stand. Plant Ecol 130(1):41–52
Kohler RH, Zipfel WR, Webb WW, Hanson MR (1997) The green fluorescent protein as a marker to visualize plant mitochondria in vivo. Plant J 11(3):613–621
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33(7):1870–1874
Lee CM, Sedman J, Neupert W, Stuart RA (1999) The DNA helicase, Hmi1p, is transported into mitochondria by a C-terminal cleavable targeting signal. J Biol Chem 274(30):20937–20942
Letunic I, Bork P (2018) 20 years of the SMART protein domain annotation resource. Nucleic Acids Res 46(D1):D493–D496
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408
Nelson BK, Cai X, Nebenfuehr A (2007) A multicolored set of in vivo organelle markers for co-localization studies in Arabidopsis and other plants. Plant J 51(6):1126–1136
Okahisa Y, Yoshimura T, Imamura Y (2006) Seasonal and height-dependent fluctuation of starch and free glucose contents in moso bamboo (Phyllostachys pubescens) and its relation to attack by termites and decay fungi. J Wood Sci 52(5):445–451
Peng Z, Zhang C, Zhang Y, Hu T, Mu S, Li X, Gao J (2013) Transcriptome sequencing and analysis of the fast growing shoots of moso bamboo (Phyllostachys edulis). PLoS ONE 8(11):e78944
Rucktaeschel R, Girzalsky W, Erdmann R (2011) Protein import machineries of peroxisomes. Biochim Biophys Acta 1808(3):892–900
Soreng RJ, Peterson PM, Romaschenko K, Davidse G, Zuloaga FO, Judziewicz EJ, Filgueiras TS, Davis JI, Morrone O (2015) A worldwide phylogenetic classification of the Poaceae (Gramineae). J Syst Evol 53(2):117–137
Sun Y, Fan XY, Cao DM, Tang W, He K, Zhu JY, He JX, Bai MY, Zhu S, Oh E, Patil S, Kim TW, Ji H, Wong WH, Rhee SY, Wang ZY (2010) Integration of brassinosteroid signal transduction with the transcription network for plant growth regulation in Arabidopsis. Dev Cell 19(5):765–777
Thompson MJ, Meudt WJ, Mandava NB, Dutky SR, Lusby WR, Spaulding DW (1982) Synthesis of brassinosteroids and relationship of structure to plant growth-promoting effects. Steroids 39(1):89–105
Tsukagoshi H, Busch W, Benfey PN (2010) Transcriptional regulation of ROS controls transition from proliferation to differentiation in the root. Cell 143(4):606–616
Wang HY, Cui K, He CY, Zeng YF, Liao SX, Zhang JG (2015) Endogenous hormonal equilibrium linked to bamboo culm development. Genet Mol Res 14(3):11312–11323
Wang T, Wang H, Cai D, Gao Y, Zhang H, Wang Y, Lin C, Ma L, Gu L (2017a) Comprehensive profiling of rhizome-associated alternative splicing and alternative polyadenylation in moso bamboo (Phyllostachys edulis). Plant J 91(4):684–699
Wang W, Gu L, Ye S, Zhang H, Cai C, Xiang M, Gao Y, Wang Q, Lin C, Zhu Q (2017b) Genome-wide analysis and transcriptomic profiling of the auxin biosynthesis, transport and signaling family genes in moso bamboo (Phyllostachys heterocycla). BMC Genomics 18:870
Wiedemann N, Pfanner N, Ryan MT (2001) The three modules of ADP/ATP carrier cooperate in receptor recruitment and translocation into mitochondria. EMBO J 20(5):951–960
Zhang Y, Su J, Duan S, Ao Y, Dai J, Liu J, Wang P, Li Y, Liu B, Feng D (2011) A highly efficient rice green tissue protoplast system for transient gene expression and studying light/chloroplast-related processes. Plant methods 7(1):30
Zhang H, Wang H, Zhu Q, Gao Y, Wang H, Zhao L, Wang Y, Xi F, Wang W, Yang Y (2018) Transcriptome characterization of moso bamboo (Phyllostachys edulis) seedlings in response to exogenous gibberellin applications. BMC Plant Biol 18(1):125
Zhao H, Lou Y, Sun H, Li L, Wang L, Dong L, Gao Z (2016) Transcriptome and comparative gene expression analysis of Phyllostachys edulis in response to high light. BMC Plant Biol 16:34
Zhou M-B, Yang P, Gao P-J, Tang D-Q (2011a) Identification of differentially expressed sequence tags in rapidly elongating Phyllostachys pubescens internodes by suppressive subtractive hybridization. Plant Mol Biol Rep 29(1):224–231
Zhou X, Li Q, Chen X, Liu J, Zhang Q, Liu Y, Liu K, Xu J (2011b) The Arabidopsis RETARDED ROOT GROWTH gene encodes a mitochondria-localized protein that is required for cell division in the root meristem. Plant Physiol 157(4):1793–1804
Acknowledgements
We thank Dr. Jia-Ying Zhu for editing the manuscript. This work was supported by a grant from the Natural Science Foundation of Fujian province (NO. 2016J01089) and FAFU-International Collaborative Program (KXb16005A) to W.W., the National Natural Science Foundation of China grant (NO. 31500226) to X.Y., the China Postdoctoral Science Foundation (2018M642551) to ZZ.Z., and Program for scientific and technological innovation team for universities of Fujian province (No. 118/KLA18069A) to Q.Z.. The funding body had no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Author information
Authors and Affiliations
Contributions
WW and ZYW designed the research program. ZG, ZZ, XY, KY and YC prepared materials. ZG performed research. ZG, ZZ, ZYW, KS, ZZZ, QZ and WW analyzed data. ZG, ZZ and WW wrote the drafting of manuscript. All authors have read, revised and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11103_2020_975_MOESM1_ESM.tif
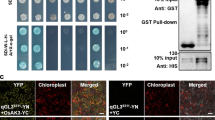
Supplemental Figure 1: Whole cell image of PSBR1 subcellular localization in tobacco, Arabidopsis and bamboo protoplast. a-l Subcellular localization of PSBR1 in Nicotiana benthamiana cells. a PSBR1-YFP signals. b DAPI stain signals. c Peroxisome maker mCherry-PTS1 signals. d Chlorophyll auto-fluorescence signals. e BF, bright-field images. f Merged of (a) and (b). g Merged of (a) and (c). h Merged of (a) and (d). i PSBR1-YFP signals. j Mitochondrial maker CoxIV-mCherry signals. k Merged of (i) and (j). l BF. Bar = 20 μm. m-p Subcellular localization of PSBR1 in Arabidopsis. m PSBR1-YFP signals. n Mito tracker orange signals. o Merged of (m) and (n). p BF. Bar = 20 μm. q-t Subcellular localization of PSBR1 in a protoplast of Moso bamboo. q PSBR1-YFP signals. r Mito tracker orange signals. s Merged of (q) and (r). t BF. Bar = 5 μm (TIF 10530 kb)
11103_2020_975_MOESM2_ESM.tif
Supplemental Figure 2: Whole cell image of subcellular localization of dissected PSBR1 fragments. a Diagram of N- and C- terminal fragments of PSBR1 for YFP fusion. b, f, j and n YFP signals. c, g, k and o Mitochondrial maker CoxIV-mCherry signals. d, h, l and p Merged images of mCherry and YFP signals. e, i, m and q BF. Bar = 20 μm (TIF 13792 kb)
Rights and permissions
About this article
Cite this article
Guo, Z., Zhang, Z., Yang, X. et al. PSBR1, encoding a mitochondrial protein, is regulated by brassinosteroid in moso bamboo (Phyllostachys edulis). Plant Mol Biol 103, 63–74 (2020). https://doi.org/10.1007/s11103-020-00975-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-020-00975-3