Abstract
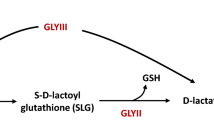
Methylglyoxal (MG) is a key signaling molecule resulting from glycolysis and other metabolic pathways. During abiotic stress, MG levels accumulate to toxic levels in affected cells. However, MG is routinely detoxified through the action of DJ1/PARK7/Hsp31 proteins that are highly conserved across kingdoms and mutations in such genes are associated with neurodegenerative diseases. Here, we report for the first time that, similar to abiotic stresses, MG levels increase during biotic stresses in plants, likely contributing to enhanced susceptibility to a wide range of stresses. We show that overexpression of yeast Heat shock protein 31 (Hsp31), a DJ-1 homolog with robust MG detoxifying capabilities, confers dual biotic and abiotic stress tolerance in model plant Nicotiana tabacum. Strikingly, overexpression of Hsp31 in tobacco imparts robust stress tolerance against diverse biotic stress inducers such as viruses, bacteria and fungi, in addition to tolerance against a range of abiotic stress inducers. During stress, Hsp31 was targeted to mitochondria and induced expression of key stress-related genes. These results indicate that Hsp31 is a novel attractive tool to engineer plants against both biotic and abiotic stresses.







Similar content being viewed by others
References
Abdallah J, Mihoub M, Gautier V, Richarme G (2016) The DJ-1 superfamily members YhbO and YajL from Escherichia coli repair proteins from glycation by methylglyoxal and glyoxal. Biochem Biophys Res Commun 470:282–286. doi:10.1016/j.bbrc.2016.01.068
Ahuja I, de Vos RCH, Bones AM, Hall RD (2010) Plant molecular stress responses face climate change. Trends Plant Sci 15:664–674. doi:10.1016/j.tplants.2010.08.002
Ali W, Isner JC, Isayenkov SV et al (2012) Heterologous expression of the yeast arsenite efflux system ACR3 improves Arabidopsis thaliana tolerance to arsenic stress. New Phytol 194:716–723. doi:10.1111/j.1469-8137.2012.04092.x
Allaman I, Bélanger M, Magistretti PJ (2015) Methylglyoxal, the dark side of glycolysis. Front Neurosci. doi:10.3389/fnins.2015.00023
Allocati N, Federici L, Masulli M, Di Ilio C (2009) Glutathione transferases in bacteria. FEBS J 276:58–75. doi:10.1111/j.1742-4658.2008.06743.x
Aslam K, Hazbun TR (2016) Hsp31, a member of the DJ-1 superfamily, is a multitasking stress responder with chaperone activity. Prion 10:103–111. doi:10.1080/19336896.2016.1141858
Bankapalli K, Saladi S, Awadia SS et al (2015) Robust glyoxalase activity of Hsp31, a ThiJ/DJ-1/PfpI family member protein, is critical for oxidative stress resistance in Saccharomyces cerevisiae. J Biol Chem 290:26491–26507. doi:10.1074/jbc.M115.673624
Baulcombe D (2004) RNA silencing in plants. Nature 431:356–363. doi:10.1079/IVP2004619
Bevan M (1984) Binary Agrobacterium vectors for plant transformation. Nucleic Acids Res 12:8711–8721. doi:10.1093/nar/12.22.8711
Bohnert HJ, Gong Q, Li P, Ma S (2006) Unraveling abiotic stress tolerance mechanisms-getting genomics going. Curr Opin Plant Biol 9:180–188. doi:10.1016/j.pbi.2006.01.003
Boyland E, Chasseaud LF (1967) Enzyme-catalysed conjugations of glutathione with unsaturated compounds. Biochem J 104:95–102
Canet-Avilés RM, Wilson MA, Miller DW et al (2004) The Parkinson’s disease protein DJ-1 is neuroprotective due to cysteine-sulfinic acid-driven mitochondrial localization. Proc Natl Acad Sci USA 101:9103–9108. doi:10.1073/pnas.0402959101
Choi J, Sullards MC, Olzmann JA et al (2006) Oxidative damage of DJ-1 is linked to sporadic Parkinson and Alzheimer diseases. J Biol Chem 281:10816–10824. doi:10.1074/jbc.M509079200
Conrath U, Beckers GJM, Flors V, et al (2006) Priming: getting ready for battle. Mol Pant-Microbe Interact 19:1062–1071. doi:10.1094/MPMI-19-1062
Daudi A, O’Brien JA (2012) Detection of hydrogen peroxide by DAB staining in Arabidopsis leaves. Bio-protocol 2:e263. doi:10.1007/BF00139728.5. http://www.bio-protocol.org/e263
Davie CA (2008) A review of Parkinson’s disease. Br Med Bull 86:109–127. doi:10.1093/bmb/ldn013
de las Mercedes Dana M, Pintor-Toro JA, Cubero B (2006) Transgenic tobacco plants overexpressing chitinases of fungal origin show enhanced resistance to biotic and abiotic stress agents. Plant Physiol 142:722–730. doi:10.1104/pp.106.086140
Dixon DP, Cummins I, Cole DJ, Edwards R (1998) Glutathione-mediated detoxification systems in plants. Curr Opin Plant Biol 1:258–266. doi:10.1007/s00299-002-0545-x
Eswaran N, Parameswaran S, Sathram B et al (2010) Yeast functional screen to identify genetic determinants capable of conferring abiotic stress tolerance in Jatropha curcas. BMC Biotechnol 10:23. doi:10.1186/1472-6750-10-23
Furtado Macedo A (2012) Abiotic stress responses in plants. Springer, Berlin. doi:10.1007/978-1-4614-0634-1
Gill SS, Tuteja N (2010) Polyamines and abiotic stress tolerance in plants. Plant Signal Behav 5:26–33. doi:10.4161/psb.5.1.10291
Gill SS, Anjum NA, Hasanuzzaman M et al (2013) Glutathione and glutathione reductase: a boon in disguise for plant abiotic stress defense operations. Plant Physiol Biochem 70:204–212. doi:10.1016/j.plaphy.2013.05.032
Goyal RK, Hancock REW, Mattoo AK, Misra S (2013) Expression of an engineered heterologous antimicrobial peptide in potato alters plant development and mitigates normal abiotic and biotic responses. PLoS ONE. doi:10.1371/journal.pone.0077505
Griffiths H, Parry MAJ, Hsiao T (2002) Plant responses to water stress. Annu Rev Plant Physiol 89:801–802
Hao L-Y, Giasson BI, Bonini NM (2010) DJ-1 is critical for mitochondrial function and rescues PINK1 loss of function. Proc Natl Acad Sci USA 107:9747–9752. doi:10.1073/pnas.0911175107
Hasim S, Hussin NA, Alomar F et al (2014) A glutathione-independent glyoxalase of the DJ-1 superfamily plays an important role in managing metabolically generated methylglyoxal in candida albicans. J Biol Chem 289:1662–1674. doi:10.1074/jbc.M113.505784
Hossain MA, Piyatida P, da Silva JAT, Fujita M (2012) Molecular mechanism of heavy metal toxicity and tolerance in plants: central role of glutathione in detoxification of reactive oxygen species and methylglyoxal and in heavy metal chelation. J Bot 2012:1–37. doi:10.1155/2012/872875
Hussain SS, Ali M, Ahmad M, Siddique KHM (2011) Polyamines: natural and engineered abiotic and biotic stress tolerance in plants. Biotechnol Adv 29:300–311. doi:10.1016/j.biotechadv.2011.01.003
Junn E, Jang WH, Zhao X et al (2009) Mitochondrial localization of DJ-1 leads to enhanced neuroprotection. J Neurosci Res 87:123–129. doi:10.1002/jnr.21831
Kasai H, Iwamoto-Tanaka N, Fukada S (1998) DNA modifications by the mutagen glyoxal: adduction to G and C, deamination of C and GC and GA cross-linking. Carcinogenesis 19:1459–1465. doi:10.1093/carcin/19.8.1459
Kaur C, Singla-Pareek SL, Sopory SK (2014) Glyoxalase and Methylglyoxal as Biomarkers for Plant Stress Tolerance. Crit Rev Plant Sci 33:429–456. doi:10.1080/07352689.2014.904147
Kaur C, Kushwaha HR, Mustafiz A et al (2015) Analysis of global gene expression profile of rice in response to methylglyoxal indicates its possible role as a stress signal molecule. Front Plant Sci 6:682. doi:10.3389/fpls.2015.00682
Lee JY, Song J, Kwon K et al (2012) Human DJ-1 and its homologs are novel glyoxalases. Hum Mol Genet 21:3215–3225. doi:10.1093/hmg/dds155
Lin J, Nazarenus TJ, Frey JL et al (2011) A plant DJ-1 homolog is essential for Arabidopsis thaliana chloroplast development. PLoS ONE. doi:10.1371/journal.pone.0023731
Luo M, Liu X, Singh P, et al. (2012) Chromatin modifications and remodeling in plant abiotic stress responses. Biochim Biophys Acta 1819:129–136. doi:10.1016/j.bbagrm.2011.06.008
May MJ, Vernoux T, Leaver C et al (1998) Glutathione homeostasis in plants: implications for environmental sensing and plant development. J Exp Bot 49:649–667. doi:10.1093/jxb/49.321.649
Morcos M, Du X, Pfisterer F et al (2008) Glyoxalase-1 prevents mitochondrial protein modification and enhances lifespan in Caenorhabditis elegans. Aging Cell 7:260–269. doi:10.1111/j.1474-9726.2008.00371.x
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. doi:10.1111/j.1399-3054.1962.tb08052.x
Nakabayashi R, Yonekura-Sakakibara K, Urano K et al (2014) Enhancement of oxidative and drought tolerance in Arabidopsis by overaccumulation of antioxidant flavonoids. Plant J 77:367–379. doi:10.1111/tpj.12388
Nakahara Y, Sawabe S, Kainuma K et al (2015) Yeast functional screen to identify genes conferring salt stress tolerance in Salicornia europaea. Front Plant Sci 6:920. doi:10.3389/fpls.2015.00920
Nakaminami K, Matsui A, Shinozaki K, Seki M (2012) RNA regulation in plant abiotic stress responses. Biochim Biophys Acta 1819:149–153. doi:10.1016/j.bbagrm.2011.07.015
Niki E, Yoshida Y, Saito Y, Noguchi N (2005) Lipid peroxidation: mechanisms, inhibition, and biological effects. Biochem Biophys Res Commun 338:668–676. doi:10.1016/j.bbrc.2005.08.072
Obata T, Fernie AR (2012) The use of metabolomics to dissect plant responses to abiotic stresses. Cell Mol Life Sci 69:3225–3243. doi:10.1007/s00018-012-1091-5
Padilla-Chacón D, Cordoba E, Olivera T et al (2010) Heterologous expression of yeast Hxt2 in arabidopsis thaliana alters sugar uptake, carbon metabolism and gene expression leading to glucose tolerance of germinating seedlings. Plant Mol Biol 72:631–641. doi:10.1007/s11103-010-9602-y
Pareek A, Sopory SK, Bohnert HJ (2010) Abiotic stress adaptation in plants. Physiol Mol Genom Found. doi:10.1007/978-90-481-3112-9
Peleg Z, Blumwald E (2011) Hormone balance and abiotic stress tolerance in crop plants. Curr Opin Plant Biol 14:290–295. doi:10.1016/j.pbi.2011.02.001
Raza H (2011) Dual localization of glutathione S-transferase in the cytosol and mitochondria: Implications in oxidative stress, toxicity and disease. FEBS J 278:4243–4251. doi:10.1111/j.1742-4658.2011.08358.x
Rhee SG, Yang K-S, Kang SW et al (2005) Controlled elimination of intracellular H2O2: regulation of peroxiredoxin, catalase, and glutathione peroxidase via post-translational modification. Antioxid Redox Signal 7:619–626. doi:10.1089/ars.2005.7.619
Roy SJ, Tucker EJ, Tester M (2011) Genetic analysis of abiotic stress tolerance in crops. Curr Opin Plant Biol 14:232–239. doi:10.1016/j.pbi.2011.03.002
Shangari N, O’Brien PJ (2004) The cytotoxic mechanism of glyoxal involves oxidative stress. Biochem Pharmacol 68:1433–1442. doi:10.1016/j.bcp.2004.06.013
Shivaprasad PV, Thillaichidambaram P, Balaji V, Veluthambi K (2006) Expression of full-length and truncated Rep genes from Mungbean yellow mosaic virus-Vigna inhibits viral replication in transgenic tobacco. Virus Genes 33:365–374. doi:10.1007/s11262-006-0077-5
Shivaprasad PV, Dunn RM, Santos BA et al (2011) Extraordinary transgressive phenotypes of hybrid tomato are influenced by epigenetics and small silencing RNAs. EMBO J 31:257–266. doi:10.1038/emboj.2011.458
Subedi KP, Choi D, Kim I et al (2011) Hsp31 of Escherichia coli K-12 is glyoxalase III. Mol Microbiol 81:926–936. doi:10.1111/j.1365-2958.2011.07736.x
Szabados L, Kovács H, Zilberstein A, Bouchereau A (2011) Plants in extreme environments: importance of protective compounds in stress tolerance. Adv Bot Res. doi:10.1016/B978-0-12-387692-8.00004-7
Szalai G, Kellos T, Galiba G, Kocsy G (2009) Glutathione as an antioxidant and regulatory molecule in plants under abiotic stress conditions. J Plant Growth Regul 28:66–80. doi:10.1007/s00344-008-9075-2
Taira T, Saito Y, Niki T et al (2004) DJ-1 has a role in antioxidative stress to prevent cell death. EMBO Rep 5:213–218. doi:10.1038/sj.embor.7400074
Tardieu F, Tuberosa R (2010) Dissection and modelling of abiotic stress tolerance in plants. Curr Opin Plant Biol 13:206–212. doi:10.1016/j.pbi.2009.12.012
Thornalley PJ (1990) The glyoxalase system: new developments towards functional characterization of a metabolic pathway fundamental to biological life. Biochem J 269:1–11
Thornalley PJ (1996) Pharmacology of methylglyoxal: formation, modification of proteins and nucleic acids, and enzymatic detoxification: a role in pathogenesis and antiproliferative chemotherapy. Gen Pharmacol 27:565–573. doi:10.1016/0306-3623(95)02054-3
Thornalley PJ (2008) Protein and nucleotide damage by glyoxal and methylglyoxal in physiological systems–role in ageing and disease. Drug Metabol Drug Interact 23:125–150. doi:10.1515/DMDI.2008.23.1-2.125
Thornalley PJ, Langborg A, Minhas HS (1999) Formation of glyoxal, methylglyoxal and 3-deoxyglucosone in the glycation of proteins by glucose. Biochem J 344:109–116. doi:10.1042/bj3440109
Treutter D (2005) Significance of flavonoids in plant resistance and enhancement of their biosynthesis. Plant Biol 7:581–591. doi:10.1055/s-2005-873009
Tsai CJ, Aslam K, Drendel HM et al (2015) Hsp31 is a stress response chaperone that intervenes in the protein misfolding process. J Biol Chem 290:24816–24834. doi:10.1074/jbc.M115.678367
Urano K, Kurihara Y, Seki M, Shinozaki K (2010) “Omics” analyses of regulatory networks in plant abiotic stress responses. Curr Opin Plant Biol 13:132–138. doi:10.1016/j.pbi.2009.12.006
Veena RVS, Sopory SK (1999) Glyoxalase I from Brassica juncea: molecular cloning, regulation and its over-expression confer tolerance in transgenic tobacco under stress. Plant J 17:385–395. doi:10.1046/j.1365-313X.1999.00390.x
Wang X, Petrie TG, Liu Y et al (2012) Parkinson’s disease-associated DJ-1 mutations impair mitochondrial dynamics and cause mitochondrial dysfunction. J Neurochem 121:830–839. doi:10.1111/j.1471-4159.2012.07734.x
Wei Y, Ringe D, Wilson MA, Ondrechen MJ (2007) Identification of functional subclasses in the DJ-1 superfamily proteins. PLoS Comput Biol 3:0120–0126. doi:10.1371/journal.pcbi.0030010
Willekens H, Chamnongpol S, Davey M et al (1997) Catalase is a sink for H2O2 and is indispensable for stress defence in C3 plants. EMBO J 16:4806–4816. doi:10.1093/emboj/16.16.4806
Wilson MA (2011) The role of cysteine oxidation in DJ-1 function and dysfunction. Antioxid Redox Signal 15:111–122. doi:10.1089/ars.2010.3481
Xu XM, Møller SG (2010) ROS removal by DJ-1: Arabidopsis as a new model to understand Parkinson’s Disease. Plant Signal Behav 5:1034–1036. doi:10.4161/psb.5.8.12298
Xu XM, Lin H, Maple J et al (2010) The Arabidopsis DJ-1a protein confers stress protection through cytosolic SOD activation. J Cell Sci 123:1644–1651. doi:10.1242/jcs.063222
Yadav SK, Singla-Pareek SL, Ray M et al (2005a) Methylglyoxal levels in plants under salinity stress are dependent on glyoxalase I and glutathione. Biochem Biophys Res Commun 337:61–67. doi:10.1016/j.bbrc.2005.08.263
Yadav SK, Singla-Pareek SL, Reddy MK, Sopory SK (2005b) Transgenic tobacco plants overexpressing glyoxalase enzymes resist an increase in methylglyoxal and maintain higher reduced glutathione levels under salinity stress. FEBS Lett 579:6265–6271. doi:10.1016/j.febslet.2005.10.006
Zhou W, Freed CR (2005) DJ-1 up-regulates glutathione synthesis during oxidative stress and inhibits A53T alpha-synuclein toxicity. J Biol Chem 280:43150–43158. doi:10.1074/jbc.M507124200
Zhou W, Zhu M, Wilson MA et al (2006) The oxidation state of DJ-1 regulates its chaperone activity toward α-synuclein. J Mol Biol 356:1036–1048. doi:10.1016/j.jmb.2005.12.030
Acknowledgements
The authors acknowledge access to Imaging, greenhouse and sequencing facilities from their respective institutions. Thanks to Prof. K. Veluthambi for pBIN19 vector, viral clones and Agrobacterium strain LBA4404 (pSB1), Dr. Radhika Venkatesan for P. syringae DC3000, Prof. Janardhana for Alternaria Spp., Swetha Chenna for help in structure prediction and N. D. Sunitha for comments.
Author contributions
PVS and PD designed all experiments, discussed results and wrote the manuscript. MP performed most of the experiments. KB designed constructs and performed microscopy.
Funding
PVS acknowledges support from Ramanujan Fellowship (SR/S2/RJN-109/2012; Department of Science and Technology, Government of India). PVS lab is supported by NCBS-TIFR core funding and a grant (BT/PR12394/AGIII/103/891/2014) from Department of Biotechnology, Government of India. PDS acknowledges support from Swarnajayanti Fellowship (DST/SJF/LS-01/2011–2012), DBT-IISc partnership program (DBT/BF/PR/INS/2011-12/IISc) and UGC-CAS SAP-II program (UGC LT. No. F. 5-2/2012. SAP-II). KB acknowledges research fellowship from CSIR.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Melvin, P., Bankapalli, K., D’Silva, P. et al. Methylglyoxal detoxification by a DJ-1 family protein provides dual abiotic and biotic stress tolerance in transgenic plants. Plant Mol Biol 94, 381–397 (2017). https://doi.org/10.1007/s11103-017-0613-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-017-0613-9