Abstract
Purpose
To study the baseline characteristics predicting hypogonadotropic hypogonadism (HH) persistence in men with macroprolactinoma that achieved prolactin normalization.
Design
Retrospective cohort study.
Methods
Male patients diagnosed with macroprolactinoma and HH that received cabergoline treatment with subsequent prolactin normalization were included: men that achieved eugonadism, and men that remained hypogonadal. Patient’s demographic, clinical and biochemical parameters, sellar imaging, and visual fields tests were obtained. Univariate and multivariate models were used to identify predictors of HH persistence.
Results
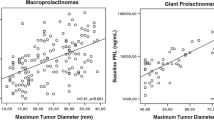
Fifty-eight male patients (age 49.2 ± 12.6 years) with a median baseline prolactin of 1154 ng/mL (IQR 478–2763 ng/mL) and adenoma (maximal) diameter of 25.9 ± 14.8 mm were followed for a median of 5.6 years (IQR 3.0–10.7). Twelve men (21%) suffered from HH persistence at the end of follow-up and 46 men achieved eugonadism. Forty-two out of 46 men (91%) accomplished eugonadism within the first year following prolactin normalization. In a multivariate logistic regression model, hypopituitarism (OR 10.1; 95% CI 1.10–101.94), visual field defect (OR 9.9; 95% CI 1.07–92.33), and low baseline testosterone levels (OR 0.5; 95% CI 0.29–0.93) were independent predictors of HH persistence.
Conclusion
In our cohort of men with macroprolactinoma that reached prolactin normalization with cabergoline treatment, 21% had HH persistence. Pituitary hormone deficiency, visual field defects, and low baseline testosterone levels were independently associated with HH persistence. 91% of men achieved eugonadism within the first year following prolactin normalization. These findings may support informed clinical decision-making regarding the initiation of testosterone replacement in men with macroprolactinomas.


Similar content being viewed by others
References
Fernandez A, Karavitaki N, Wass JA (2010) Prevalence of pituitary adenomas: a community-based, cross-sectional study in Banbury (Oxfordshire, UK). Clin Endocrinol (Oxford) 72:377–382. https://doi.org/10.1111/j.1365-2265.2009.03667.x
Mindermann T, Wilson CB (1994) Age-related and gender-related occurrence of pituitary adenomas. Clin Endocrinol (Oxford) 41:359–364. https://doi.org/10.1111/j.1365-2265.1994.tb02557.x
Delgrange E, Trouillas J, Maiter D, Donckier J, Tourniaire J (1997) Sex-related difference in the growth of prolactinomas: a clinical and proliferation marker study. J Clin Endocrinol Metab 82:2102–2107. https://doi.org/10.1210/jcem.82.7.4088
Calle-Rodrigue RD, Giannini C, Scheithauer BW et al (1998) Prolactinomas in male and female patients: a comparative clinicopathologic study. Mayo Clin Proc 73:1046–1052. https://doi.org/10.4065/73.11.1046
Colao A, Vitale G, Cappabianca P, Briganti F, Ciccarelli A, De Rosa M, Zarrilli S, Lombardi G (2004) Outcome of cabergoline treatment in men with prolactinoma: effects of a 24-month treatment on prolactin levels, tumor mass, recovery of pituitary function, and semen analysis. J Clin Endocrinol Metab 89:1704–1711. https://doi.org/10.1210/jc.2003-030979
Pinzone JJ, Katznelson L, Danila DC, Pauler DK, Miller CS, Klibanski A (2000) Primary medical therapy of micro- and macroprolactinomas in men. J Clin Endocrinol Metab 85:3053–3057. https://doi.org/10.1210/jcem.85.9.6798
Rudman Y, Duskin-Bitan H, Manisterski Y, Pertzov B, Akirov A, Masri-Iraqi H, Shimon I (2021) Long-term response to cabergoline and multi-modal treatment in men with macroprolactinoma: does size really matter? Clin Endocrinol (Oxford) 95:606–617. https://doi.org/10.1111/cen.14541
Sibal L, Ugwu P, Kendall-Taylor P, Ball SG, James RA, Pearce SH, Hall K, Quinton R (2002) Medical therapy of macroprolactinomas in males: I. Prevalence of hypopituitarism at diagnosis: II. Proportion of cases exhibiting recovery of pituitary function. Pituitary 5:243–246. https://doi.org/10.1023/a:1025377816769
Tirosh A, Benbassat C, Lifshitz A, Shimon I (2015) Hypopituitarism patterns and prevalence among men with macroprolactinomas. Pituitary 18:108–115. https://doi.org/10.1007/s11102-014-0563-z
De Rosa M, Ciccarelli A, Zarrilli S, Guerra E, Gaccione M, Di Sarno A, Lombardi G, Colao A (2006) The treatment with cabergoline for 24 month normalizes the quality of seminal fluid in hyperprolactinaemic males. Clin Endocrinol (Oxford) 64:307–313. https://doi.org/10.1111/j.1365-2265.2006.02461.x
Sehemby M, Lila AR, Sarathi V, Shah R, Sankhe S, Jaiswal SK, Ramteke-Jadhav S, Patil V, Shah N, Bandgar T (2020) Predictors of chronic LH-testosterone axis suppression in male macroprolactinomas with normoprolactinemia on cabergoline. J Clin Endocrinol Metab 105:dgaa650. https://doi.org/10.1210/clinem/dgaa650
Shimon I, Benbassat C, Tzvetov G, Grozinsky-Glasberg S (2011) Anemia in a cohort of men with macroprolactinomas: increase in hemoglobin levels follows prolactin suppression. Pituitary 14:11–15. https://doi.org/10.1007/s11102-010-0251-6
Moult PJ, Rees LH, Besser GM (1982) Pulsatile gonadotrophin secretion in hyperprolactinaemic amenorrhoea an the response to bromocriptine therapy. Clin Endocrinol (Oxford) 16:153–162. https://doi.org/10.1111/j.1365-2265.1982.tb03159.x
Espinosa E, Sosa E, Mendoza V, Ramírez C, Melgar V, Mercado M (2016) Giant prolactinomas: are they really different from ordinary macroprolactinomas? Endocrine 52:652–659. https://doi.org/10.1007/s12020-015-0791-7
Melmed S, Casanueva FF, Hoffman AR et al (2011) Diagnosis and treatment of hyperprolactinemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2011(96):273–288. https://doi.org/10.1210/jc.2010-1692
Colao A, Savastano S (2011) Medical treatment of prolactinomas. Nat Rev Endocrinol 7:267–278. https://doi.org/10.1038/nrendo.2011.37
Karavitaki N, Dobrescu R, Byrne JV, Grossman AB, Wass JA (2013) Does hypopituitarism recover when macroprolactinomas are treated with cabergoline? Clin Endocrinol (Oxford) 79:217–223. https://doi.org/10.1111/cen.12124
Walia R, Bhansali A, Dutta P, Khandelwal N, Sialy R, Bhadada S (2011) Recovery pattern of hypothalamo-pituitary-testicular axis in patients with macroprolactinomas after treatment with cabergoline. Indian J Med Res 134:314–319
Karavitaki N, Thanabalasingham G, Shore HC et al (2006) Do the limits of serum prolactin in disconnection hyperprolactinaemia need re-definition? A study of 226 patients with histologically verified non-functioning pituitary macroadenoma. Clin Endocrinol (Oxford) 65:524–529. https://doi.org/10.1111/j.1365-2265.2006.02627.x
Voica M, Tetlay M, Thompson DV, Hasan F (2021) Recovery of male hypogonadism following successful treatment of prolactinoma: the experience of an integrated health network. J Endocr Soc 5(Suppl 1):A632. https://doi.org/10.1210/jendso/bvab048.1289
Iglesias P, Arcano K, Berrocal VR, Bernal C, Villabona C, Díez JJ (2018) Giant prolactinoma in men: clinical features and therapeutic outcomes. Horm Metab Res 50:791–796. https://doi.org/10.1055/a-0752-0741
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to study conception and design. YR, IS performed a literature search. YR, HDB, HMI, IS contributed to data collection. YR, IS contributed to data analysis and synthesis. YR, IS drafted the first version of the manuscript. All authors contributed to writing and critically reviewing the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rudman, Y., Duskin-Bitan, H., Masri-Iraqi, H. et al. Predicting hypogonadotropic hypogonadism persistence in male macroprolactinoma. Pituitary 25, 882–890 (2022). https://doi.org/10.1007/s11102-022-01259-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-022-01259-3