Abstract
Purpose
Giant prolactinoma (GP) in childhood and adolescence is a rare entity with scarce literature. We aimed to describe clinical features, biochemistry, radiology, genetics, management, and outcome in pediatric (≤ 20 years) GP.
Methods
Retrospective record review of 18 pediatric GP patients from our center and systematic review including these and 77 from the literature (total cohort: 95).
Results
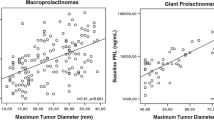
GP constituted 20% of our pediatric prolactinoma cohort. In the total cohort (age: 15.4 ± 3.5 years), the majority (77, 82.8%) were males. Mass effect symptoms (88.6%), and pubertal delay/arrest in males (82.1%) were frequent. Median basal prolactin was 8649 (3246–17,532) ng/ml and the maximum tumor dimension was 5.5 ± 1.5 cm. MEN1 and AIP mutations were noted in 7 (21.9%) and 6 (18.8%) patients, respectively. Males with central hypogonadism had baseline bi-testicular volume of 20.2 ± 8.4 cc, lower LH than FSH (−2.04 ± 0.9 vs. −0.7 ± 1.6 SDS, p = 0.0075), and mostly, normal inhibin B. Majority (49/76, 64.5%) received dopamine agonist (DA) as first-line treatment with additional therapy in 35% (17/49). DA monotherapy arm had less frequent central hypothyroidism (42.9% vs 87.1%, p = 0.002) and central adrenal insufficiency (7.1% vs 66.7%, p = 0.0003) than multimodal therapy. A smaller tumor dimension (4.7 vs. 5.7 cm, p = 0.04) was associated with normoprolactinemia on DA monotherapy and AIP mutations (33.3% vs. nil, p = 0.02) with multimodal therapy.
Conclusion
GP is characterized by male predominance with frequent delay/arrest of puberty (82%), but relative sparing of the FSH-inhibin B axis in boys. DA monotherapy may be preferred as the first-line therapy in pediatric GP.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during this study are available on request from the corresponding author on reasonable request.
References
Chanson P, Maiter D (2019) The epidemiology, diagnosis and treatment of prolactinomas: the old and the new. Best Pract Res Clin Endocrinol Metab 33:101290. https://doi.org/10.1016/j.beem.2019.101290
Hoffmann A, Adelmann S, Lohle K et al (2018) Pediatric prolactinoma: initial presentation, treatment, and long-term prognosis. Eur J Pediatr 177:125–132. https://doi.org/10.1007/s00431-017-3042-5
Arya VB, Aylwin SJB, Hulse T et al (2021) Prolactinoma in childhood and adolescence-Tumour size at presentation predicts management strategy: single centre series and a systematic review and meta-analysis. Clin Endocrinol (Oxf) 94:413–423. https://doi.org/10.1111/cen.14394
Shimon I (2019) Giant prolactinomas. Neuroendocrinology 109:51–56. https://doi.org/10.1159/000495184
Alikasifoglu A, Celik NB, Ozon ZA et al (2021) Management of prolactinomas in children and adolescents; which factors define the response to treatment? Pituitary. https://doi.org/10.1007/s11102-021-01184-x
Moraes AB, dos Silva CM, S, Vieira Neto L, Gadelha MR, (2013) Giant prolactinomas: the therapeutic approach. Clin Endocrinol (Oxf) 79:447–456. https://doi.org/10.1111/cen.12242
Di Chiro G, Nelson KB (1962) The volume of the sella turcica. Am J Roentgenol Radium Ther Nucl Med 87:989–1008
Khadilkar VV, Khadilkar AV (2015) Revised Indian Academy of Pediatrics 2015 growth charts for height, weight and body mass index for 5–18-year-old Indian children. Indian J Endocrinol Metab 19:470–476. https://doi.org/10.4103/2230-8210.159028
Fanelli F, Baronio F, Ortolano R et al (2018) Normative basal values of hormones and proteins of gonadal and adrenal functions from birth to adulthood. Sex Dev 12:50–94. https://doi.org/10.1159/000486840
Peña AS, Witchel SF, Hoeger KM et al (2020) Adolescent polycystic ovary syndrome according to the international evidence-based guideline. BMC Med 18:72. https://doi.org/10.1186/s12916-020-01516-x
Khare S, Lila AR, Patt H et al (2016) Gender differences in macroprolactinomas: a single centre experience. Endocr Connect 5:20–27. https://doi.org/10.1530/EC-15-0105
Maiter D, Delgrange E (2014) Therapy of endocrine disease: the challenges in managing giant prolactinomas. Eur J Endocrinol 170:R213-227. https://doi.org/10.1530/EJE-14-0013
Salenave S, Ancelle D, Bahougne T et al (2015) Macroprolactinomas in children and adolescents: factors associated with the response to treatment in 77 patients. J Clin Endocrinol Metab 100:1177–1186. https://doi.org/10.1210/jc.2014-3670
Delgrange E, Trouillas J, Maiter D et al (1997) Sex-related difference in the growth of prolactinomas: a clinical and proliferation marker study. J Clin Endocrinol Metab 82:2102–2107. https://doi.org/10.1210/jcem.82.7.4088
Delgrange E, Raverot G, Bex M et al (2014) Giant prolactinomas in women. Eur J Endocrinol 170:31–38. https://doi.org/10.1530/EJE-13-0503
Howlett TA, Wass JA, Grossman A et al (1989) Prolactinomas presenting as primary amenorrhoea and delayed or arrested puberty: response to medical therapy. Clin Endocrinol (Oxf) 30:131–140. https://doi.org/10.1111/j.1365-2265.1989.tb03734.x
Yang A, Cho SY, Park H et al (2020) Clinical, hormonal, and neuroradiological characteristics and therapeutic outcomes of prolactinomas in children and adolescents at a single center. Front Endocrinol (Lausanne) 11:527. https://doi.org/10.3389/fendo.2020.00527
Acharya SV, Gopal RA, Bandgar TR et al (2009) Clinical profile and long term follow up of children and adolescents with prolactinomas. Pituitary 12:186–189. https://doi.org/10.1007/s11102-008-0149-8
Kim Y-M, Seo GH, Kim Y-M et al (2018) Broad clinical spectrum and diverse outcomes of prolactinoma with pediatric onset: medication-resistant and recurrent cases. Endocr J 65:307–315. https://doi.org/10.1507/endocrj.EJ17-0268
Ben-Jonathan N, Hugo E (2015) Prolactin (PRL) in adipose tissue: regulation and functions. Adv Exp Med Biol 846:1–35. https://doi.org/10.1007/978-3-319-12114-7_1
Augustine RA, Knowles PJ, Khant Aung Z et al (2019) Impaired hypothalamic leptin sensitivity in pseudopregnant rats treated with chronic prolactin to mimic pregnancy. J Neuroendocrinol 31:e12702. https://doi.org/10.1111/jne.12702
Raverot G, Wierinckx A, Dantony E et al (2010) Prognostic factors in prolactin pituitary tumors: clinical, histological, and molecular data from a series of 94 patients with a long postoperative follow-up. J Clin Endocrinol Metab 95:1708–1716. https://doi.org/10.1210/jc.2009-1191
Melmed S, Casanueva FF, Hoffman AR et al (2011) Diagnosis and treatment of hyperprolactinemia: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 96:273–288. https://doi.org/10.1210/jc.2010-1692
Espinosa E, Sosa E, Mendoza V et al (2016) Giant prolactinomas: are they really different from ordinary macroprolactinomas? Endocrine 52:652–659. https://doi.org/10.1007/s12020-015-0791-7
Tichomirowa MA, Barlier A, Daly AF et al (2011) High prevalence of AIP gene mutations following focused screening in young patients with sporadic pituitary macroadenomas. Eur J Endocrinol 165:509–515. https://doi.org/10.1530/EJE-11-0304
van den Broek MFM, van Nesselrooij BPM, Verrijn Stuart AA et al (2019) Clinical relevance of genetic analysis in patients with pituitary adenomas: a systematic review. Front Endocrinol (Lausanne) 10:837. https://doi.org/10.3389/fendo.2019.00837
Martínez de LaPiscina I, Portillo Najera N, Rica I et al (2021) Clinical and genetic characteristics in patients under 30 years with sporadic pituitary adenomas. Eur J Endocrinol 185:485–496. https://doi.org/10.1530/EJE-21-0075
Daly AF, Tichomirowa MA, Petrossians P et al (2010) Clinical characteristics and therapeutic responses in patients with germ-line AIP mutations and pituitary adenomas: an international collaborative study. J Clin Endocrinol Metab 95:E373-383. https://doi.org/10.1210/jc.2009-2556
de Laat JM, Dekkers OM, Pieterman CRC et al (2015) Long-term natural course of pituitary tumors in patients with MEN1: results from the DutchMEN1 study group (DMSG). J Clin Endocrinol Metab 100:3288–3296. https://doi.org/10.1210/JC.2015-2015
Sehemby M, Lila AR, Sarathi V et al (2020) Predictors of chronic LH-testosterone axis suppression in male macroprolactinomas with normoprolactinemia on cabergoline. J Clin Endocrinol Metab 105:650. https://doi.org/10.1210/clinem/dgaa650
Carter JN, Tyson JE, Tolis G et al (1978) Prolactin-screening tumors and hypogonadism in 22 men. N Engl J Med 299:847–852. https://doi.org/10.1056/NEJM197810192991602
Bernard V, Young J, Chanson P, Binart N (2015) New insights in prolactin: pathological implications. Nat Rev Endocrinol 11:265–275. https://doi.org/10.1038/nrendo.2015.36
Rastrelli G, Corona G, Mannucci E, Maggi M (2014) Factors affecting spermatogenesis upon gonadotropin-replacement therapy: a meta-analytic study. Andrology 2:794–808. https://doi.org/10.1111/andr.262
Shah R, Patil V, Sarathi V et al (2021) Prior testosterone replacement therapy may impact spermatogenic response to combined gonadotropin therapy in severe congenital hypogonadotropic hypogonadism. Pituitary 24:326–333. https://doi.org/10.1007/s11102-020-01111-6
Hamidi O, Van Gompel J, Gruber L et al (2019) Management and outcomes of giant prolactinoma: a series of 71 patients. Endocr Pract 25:340–352. https://doi.org/10.4158/EP-2018-0392
Suliman SGI, Gurlek A, Byrne JV et al (2007) Nonsurgical cerebrospinal fluid rhinorrhea in invasive macroprolactinoma: incidence, radiological, and clinicopathological features. J Clin Endocrinol Metab 92:3829–3835. https://doi.org/10.1210/jc.2007-0373
Acknowledgements
The authors thank Dr. Neelam Jaguste and Dr. Aparna Kamble for their assistance in conducting the research.
Funding
This research did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sector.
Author information
Authors and Affiliations
Contributions
SK., VS. and ARL. were involved in the conception, data collection, analysis, interpretation, and drafting of the article. MS., SSM and MK. were involved in the data collection, analysis, interpretation, and critical revision of the article. SS. had done the radiological analysis. VAP., NS., and TB. all played a role in the conception, design, and interpretation of data and also critically reviewed and approved the final version of the article.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that no conflict of interest could be perceived as prejudicing the impartiality of the research reported.
Ethical approval
The study was started after approval by the Institutional Ethics Committee (EC/OA-142/2020), Seth GS Medical College and KEM Hospital. Individual patient identity was kept confidential and coded prior to analysis.
Consent of participate
Considering retrospective nature of analysis, waiver for consent was obtained from the Ethics committee.
Consent for publication
All the the co-authors have consented for publication of the study results.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kumar, S., Sarathi, V., Lila, A.R. et al. Giant prolactinoma in children and adolescents: a single-center experience and systematic review. Pituitary 25, 819–830 (2022). https://doi.org/10.1007/s11102-022-01250-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-022-01250-y