Abstract
Purpose
The authors review their treatment experience and summarize clinical outcomes for patients with hypophysitis over the past 15 years.
Methods
A retrospective analysis was conducted on patients with lymphocytic, granulomatous or IgG4-related hypophysitis treated from 1997 to 2014 at a single academic center. Patients’ medical records were reviewed and binary logistic regression analysis was used to assess whether various clinical parameters were associated with improved outcomes including endocrine function, radiographic appearance and disease recurrence.
Results
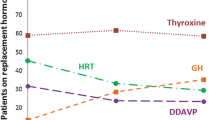
Twenty-one patients (13 women and 8 men) were identified with a median diagnosis age of 37.4 years. All but two patients (90 %) were diagnosed histopathologically and the remaining two were diagnosed clinically with lymphocytic hypophysitis. 16 patients (76 %) had lymphocytic hypophysitis, 3 (14 %) had granulomatous hypophysitis, 1 (5 %) had IgG4-related hypophysitis and 1 (5 %) had mixed lymphocytic-granulomatous. Patients presented with various symptoms of expanding sellar mass with most common signs including headache (57 %), polyuria/polydipsia (52 %), vision changes (52 %) and amenorrhea or decreased libido (48 %). Pre-treatment endocrine evaluation revealed that 12 (57 %) patients had complete anterior hypopituitarism, 11 patients (52 %) had diabetes insipidus, ten patients (48 %) had mild hyperprolactinemia and three patients (14 %) had isolated endocrine axis deficiencies with partial gland function. We observed a broad diversity in pre-treatment imaging with common findings including uniform contrast enhancement (62 %), thickened infundibulum (57 %) and loss of hypophysis bright spot on T1 imaging (43 %). Patients were treated with steroids and hormone supplementation as needed. 16 patients (76 %) had recorded post-treatment MRI scans which revealed that half had radiographic improvement and half had stable or worsened post-treatment imaging. Only female gender was found to significantly predict improved odds of post-steroid radiographic improvement. For post-treatment endocrine evaluation, six patients (29 %) did not have an evaluation on record, four patients (19 %) had some improvement in at least one axis, seven patients (33 %) had stable but non-worsened endocrine function and four patients (19 %) had worsened endocrine function post-steroids.
Conclusions
Hypophysitis is an increasingly recognized diagnosis that can present with a broad array of radiographic and clinical features. Surgical biopsy can be helpful to make definitive diagnosis and may guide treatment decision-making.




Similar content being viewed by others
References
Caturegli P, Newschaffer C, Olivi A et al (2005) Autoimmune hypophysitis. Endocr Rev 26:599–614
Caturegli P, Lupi I, Landek-Salgado M et al (2008) Pituitary autoimmunity: 30 years later. Autoimmun Rev 7:631–637
Leporati P, Landek-Salgado MA, Lupi I et al (2011) IgG4-related hypophysitis: a new addition to the hypophysitis spectrum. J Clin Endocrinol Metab 96:1971–1980
Rivera J-A (2006) Lymphocytic hypophysitis: disease spectrum and approach to diagnosis and therapy. Pituitary 9:35–45
Khare S, Jagtap VS, Budyal SR et al (2013) Primary (autoimmune) hypophysitis: a single centre experience. Pituitary
Falorni A, Minarelli V, Bartoloni E et al (2014) Diagnosis and classification of autoimmune hypophysitis. Autoimmun Rev 13:412–416
Goudie RB, Pinkerton PH (1962) Anterior hypophysitis and Hashimoto’s disease in a young woman. J Pathol Bacteriol 83:584–585
Shi J, Zhang J, Wu Q et al (2009) Granulomatous hypophysitis: two case reports and literature review. J Zhejiang Univ Sci B 10:552–558
Hamnvik O-PR, Laury AR, Laws ER, Kaiser UB (2010) Lymphocytic hypophysitis with diabetes insipidus in a young man. Nat Rev Endocrinol 6:464–470
Bellastella A, Bizzarro A, Coronella C et al (2003) Lymphocytic hypophysitis: a rare or underestimated disease? Eur J Endocrinol Eur Fed Endocr Soc 149:363–376
De Bellis A, Sinisi AA, Conte M et al (2007) Antipituitary antibodies against gonadotropin-secreting cells in adult male patients with apparently idiopathic hypogonadotropic hypogonadism. J Clin Endocrinol Metab 92:604–607
De Bellis A, Dello Iacovo A, Bellastella G et al (2014) Characterization of pituitary cells targeted by antipituitary antibodies in patients with isolated autoimmune diseases without pituitary insufficiency may help to foresee the kind of future hypopituitarism. Pituitary 17:457–463
Torino F, Barnabei A, De Vecchis L et al (2012) Hypophysitis induced by monoclonal antibodies to cytotoxic T lymphocyte antigen 4: challenges from a new cause of a rare disease. Oncologist 17:525–535
Shimatsu A, Oki Y, Fujisawa I, Sano T (2009) Pituitary and stalk lesions (infundibulo-hypophysitis) associated with immunoglobulin G4-related systemic disease: an emerging clinical entity. Endocr J 56:1033–1041
Van der Vliet HJJ, Perenboom RM (2004) Multiple pseudotumors in IgG4-associated multifocal systemic fibrosis. Ann Intern Med 141:896–897
Wong S, Lam WY, Wong WK, Lee KC (2007) Hypophysitis presented as inflammatory pseudotumor in immunoglobulin G4-related systemic disease. Hum Pathol 38:1720–1723
Bando H, Iguchi G, Fukuoka H et al (2014) The prevalence of IgG4-related hypophysitis in 170 consecutive patients with hypopituitarism and/or central diabetes insipidus and review of the literature. Eur J Endocrinol Eur Fed Endocr Soc 170:161–172
Honegger J, Fahlbusch R, Bornemann A et al (1997) Lymphocytic and granulomatous hypophysitis: experience with nine cases. Neurosurgery 40:713–722. Discussion 722–723
Leung GKK, Lopes M-BS, Thorner MO et al (2004) Primary hypophysitis: a single-center experience in 16 cases. J Neurosurg 101:262–271
Gutenberg A, Hans V, Puchner MJA et al (2006) Primary hypophysitis: clinical-pathological correlations. Eur J Endocrinol Eur Fed Endocr Soc 155:101–107
Menon SK, Sarathi V, Bandgar TR et al (2009) Autoimmune hypophysitis: a single centre experience. Singap Med J 50:1080–1084
Gutenberg A, Larsen J, Lupi I et al (2009) A radiologic Score to distinguish autoimmune hypophysitis from nonsecreting pituitary adenoma preoperatively. Am J Neuroradiol 30:1766–1772
Leporati P, Landek-Salgado MA, Lupi I et al (2011) IgG4-related hypophysitis: a new addition to the hypophysitis spectrum. J Clin Endocrinol Metab 96:1971–1980
Umehara H, Okazaki K, Masaki Y et al (2012) Comprehensive diagnostic criteria for IgG4-related disease (IgG4-RD), 2011. Mod Rheumatol Jpn Rheum Assoc 22:21–30
Asa SL, Bilbao JM, Kovacs K et al (1981) Lymphocytic hypophysitis of pregnancy resulting in hypopituitarism: a distinct clinicopathologic entity. Ann Intern Med 95:166–171
Landek-Salgado MA, Gutenberg A, Lupi I et al (2010) Pregnancy, postpartum autoimmune thyroiditis, and autoimmune hypophysitis: intimate relationships. Autoimmun Rev 9:153–157
Sautner D, Saeger W, Lüdecke DK et al (1995) Hypophysitis in surgical and autoptical specimens. Acta Neuropathol (Berl) 90:637–644
Fehn M, Sommer C, Ludecke DK et al (1998) Lymphocytic hypophysitis: light and electron microscopic findings and correlation to clinical appearance. Endocr Pathol 9:71–78
Buxton N, Robertson I (2001) Lymphocytic and granulocytic hypophysitis: a single centre experience. Br J Neurosurg 15:242–245. Discussion 245–246
Yamamoto M, Takahashi H, Ohara M et al (2006) A case of Mikulicz’s disease (IgG4-related plasmacytic disease) complicated by autoimmune hypophysitis. Scand J Rheumatol 35:410–411
Zen Y, Kitagawa S, Minato H et al (2005) IgG4-positive plasma cells in inflammatory pseudotumor (plasma cell granuloma) of the lung. Hum Pathol 36:710–717
Fujiu K, Sakuma H, Miyamoto H, Yamaguchi B (2010) Immunoglobulin G4-related inflammatory pseudotumor of the lung. Gen Thorac Cardiovasc Surg 58:144–148
Nishikawa G, Nakamura K, Yamada Y et al (2011) Inflammatory pseudotumors of the kidney and the lung presenting as immunoglobulin G4-related disease: a case report. J Med Case Rep 5:480
Albini CH, MacGillivray MH, Fisher JE et al (1988) Triad of hypopituitarism, granulomatous hypophysitis, and ruptured Rathke’s cleft cyst. Neurosurgery 22:133–136
Wearne MJ, Barber PC, Johnson AP (1995) Symptomatic Rathke’s cleft cyst with hypophysitis. Br J Neurosurg 9:799–803
Roncaroli F, Bacci A, Frank G, Calbucci F (1998) Granulomatous hypophysitis caused by a ruptured intrasellar Rathke’s cleft cyst: report of a case and review of the literature. Neurosurgery 43:146–149
Hama S, Arita K, Tominaga A et al (1999) Symptomatic Rathke’s cleft cyst coexisting with central diabetes insipidus and hypophysitis: case report. Endocr J 46:187–192
Daikokuya H, Inoue Y, Nemoto Y et al (2000) Rathke’s cleft cyst associated with hypophysitis: MRI. Neuroradiology 42:532–534
Nishikawa T, Takahashi JA, Shimatsu A, Hashimoto N (2007) Hypophysitis caused by Rathke’s cleft cyst case report. Neurol Med Chir (Tokyo) 47:136–139
Komatsu F, Tsugu H, Komatsu M et al (2010) Clinicopathological characteristics in patients presenting with acute onset of symptoms caused by Rathke’s cleft cysts. Acta Neurochir (Wien) 152:1673–1678
Myers JL, Kurtin PJ, Katzenstein AL et al (1995) Lymphomatoid granulomatosis. Evidence of immunophenotypic diversity and relationship to Epstein–Barr virus infection. Am J Surg Pathol 19:1300–1312
Jaffe ES, Wilson WH (1997) Lymphomatoid granulomatosis: pathogenesis, pathology and clinical implications. Cancer Surv 30:233–248
Roschewski M, Wilson WH (2012) Lymphomatoid granulomatosis. Cancer J Sudbury Mass 18:469–474
Tagliavini E, Rossi G, Valli R et al (2013) Lymphomatoid granulomatosis: a practical review for pathologists dealing with this rare pulmonary lymphoproliferative process. Pathologica 105:111–116
Katzenstein AL, Carrington CB, Liebow AA (1979) Lymphomatoid granulomatosis: a clinicopathologic study of 152 cases. Cancer 43:360–373
Patsalides AD, Atac G, Hedge U et al (2005) Lymphomatoid granulomatosis: abnormalities of the brain at MR imaging. Radiology 237:265–273
Bushunow PW, Casas V, Duggan DB (1996) Lyphomatoid granulomatosis causing central diabetes insipidus: case report and review of the literature. Cancer Invest 14:112–119
Carmichael JD (2012) Update on the diagnosis and management of hypophysitis. Curr Opin Endocrinol Diabetes Obes 19:314–321
Lecube A, Francisco G, Rodríguez D et al (2003) Lymphocytic hypophysitis successfully treated with azathioprine: first case report. J Neurol Neurosurg Psychiatry 74:1581–1583
Curtò L, Torre ML, Cotta OR et al (2010) Lymphocytic hypophysitis: differential diagnosis and effects of high-dose pulse steroids, followed by azathioprine, on the pituitary mass and endocrine abnormalities—report of a case and literature review. Sci World J 10:126–134
Lu Z, Yang G, Ba J et al (2011) Spontaneous pregnancy in a patient with a relapse of lymphocytic hypophysitis successfully treated with azathioprine and glucocorticoids. Endocrine 39:296–297
Papanastasiou L, Pappa T, Tsiavos V et al (2011) Azathioprine as an alternative treatment in primary hypophysitis. Pituitary 14:16–22
Yang G-Q, Lu Z-H, Gu W-J et al (2011) Recurrent autoimmune hypophysitis successfully treated with glucocorticoids plus azathioprine: a report of three cases. Endocr J 58:675–683
Caputo C, Bazargan A, McKelvie PA et al (2014) Hypophysitis due to IgG4-related disease responding to treatment with azathioprine: an alternative to corticosteroid therapy. Pituitary 17:251–256
Schreckinger M, Francis T, Rajah G et al (2012) Novel strategy to treat a case of recurrent lymphocytic hypophysitis using rituximab. J Neurosurg 116:1318–1323
Selch MT, DeSalles AAF, Kelly DF et al (2003) Stereotactic radiotherapy for the treatment of lymphocytic hypophysitis: report of two cases. J Neurosurg 99:591–596
Ray DK, Yen CP, Vance ML et al (2010) Gamma knife surgery for lymphocytic hypophysitis. J Neurosurg 112:118–121
Howlett TA, Levy MJ, Robertson IJ (2010) How reliably can autoimmune hypophysitis be diagnosed without pituitary biopsy. Clin Endocrinol (Oxf) 73:18–21
Swearingen B, Barker FG 2nd, Katznelson L et al (1998) Long-term mortality after transsphenoidal surgery and adjunctive therapy for acromegaly. J Clin Endocrinol Metab 83:3419–3426
Jahangiri A, Wagner JR, Pekmezci M et al (2013) A comprehensive long-term retrospective analysis of silent corticotrophic adenomas vs hormone-negative adenomas. Neurosurgery 73:8–17. Discussion 17–18
Jahangiri A, Wagner J, Han SW et al (2014) Morbidity of repeat transsphenoidal surgery assessed in more than 1000 operations. J Neurosurg 121:67–74
Acknowledgments
We thank Jeffrey Wagner, BS for his assistance with identification of cases.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Imber, B.S., Lee, H.S., Kunwar, S. et al. Hypophysitis: a single-center case series. Pituitary 18, 630–641 (2015). https://doi.org/10.1007/s11102-014-0622-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-014-0622-5