Abstract
Background
The management of giant prolactinomas remains a major challenge, despite dopamine agonists being the first line of treatment, owing to its efficacy to normalize prolactin levels and reduce tumor volume. The aim of this study is to characterize the therapeutic aspects, manifestations and outcomes of 16 cases of giant prolactinomas admitted at a single tertiary center in Riyadh, Saudi Arabia.
Methods
Retrospective data collection involving 16 Saudi patients diagnosed with giant prolactinoma at the Pituitary Clinic in King Fahad Medical City, Riyadh, Saudi Arabia between January 2006 and July 2012.
Results
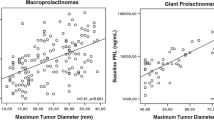
A total of 16 patients (ten males; six females) with age of diagnosis between 21 and 55 years (mean 34.9 years) were included in the analysis. The most common presenting features include headache, visual defects and sexual dysfunction. Baseline mean serum prolactin level were extremely high for both sexes which eventually decreased by as much as 97 % after cabergoline treatment. Serum prolactin concentrations completely normalized in six patients and significantly decreased in five patients 3–5 times that of normal range. Tumor volume also decreased by an average of 86 % for males and 87 % for females. Two patients had no tumor size change with cabergoline and required surgery.
Conclusion
Findings indicate that cabergoline provides dramatic clinical improvements with excellent safety profile. Cabergoline should therefore be considered as the primary therapy for giant prolactinomas.
Similar content being viewed by others
References
Ciccarelli A, Daly AF, Beckers A (2005) The epidemiology of prolactinomas. Pituitary 8(1):3–6
Schaller B (2005) Gender-related differences in prolactinomas. A clinicopathological study. Neuro Endocrinol Lett 26(2):152–159
Shrivastava RK, Arginteanu MS, King WA, Post KD (2002) Giant prolactinomas: clinical management and long-term follow up. J Neurosurg 97(2):299–306
Murphy FY, Vesely DL, Jordan RM, Flanigan S, Kohler PO (1987) Giant invasive prolactinomas. Am J Med 83(5):995–1002
Corsello SM, Ubertini G, Altomare M, Lovicu RM, Migneco MG, Rota CA, Colosimo C (2003) Giant prolactinomas in men: efficacy of cabergoline treatment. Clin Endocrinol (Oxf) 58(5):662–670
Gillam MP et al (2006) Advances in the treatment of prolactinomas. Endocr Rev 27(5):485–534
Cho EH, Lee SA, Chung JY, Koh EH, Cho YH, Kim JH, Kim CJ, Kim MS (2009) Efficacy and safety of cabergoline as first line treatment for invasive giant prolactinoma. J Korean Med Sci 24(5):874–878
Hofle G, Gasser R, Mohsenipour I, Finkenstedt G (1998) Surgery combined with dopamine agonists versus dopamine agonists alone in long-term treatment of macroprolactinoma: a retrospective study. Exp Clin Endocrinol Diabetes 106(3):211–216
Pinzone JJ, Katznelson L, Danila DC, Pauler DK, Miller CS, Klibanski A (2000) Primary medical therapy of micro- and macroprolactinomas in men. J Clin Endocrinol Metab 85(9):3053–3057
Berezin M, Shimon I, Hadani M (1995) Prolactinoma in 53 men: clinical characteristics and modes of treatment (male prolactinoma). J Endocrinol Invest 18(6):436–441
Maiter D, Delgrange E (2014) The challenges in managing giant prolactinomas. Eur J Endocrinol 170(6):R213–R227
Acharya SV, Gopal RA, Menon PS, Bandgar TR, Shah NS (2010) Giant prolactinoma and effectiveness of medical management. Endocr Pract 16(1):42–46
Hulting AL, Muhr C, Lundberg PO, Werner S (1985) Prolactinomas in men: clinical characteristics and the effect of bromocriptine treatment. Acta Med Scand 217(1):101–109
Wu ZB, Yu CJ, Su ZP, Zhuge QC, Wu JS, Zheng WM (2006) Bromocriptine treatment of invasive giant prolactinomas involving the cavernous sinus: results of a long-term follow up. J Neurosurg 104(1):54–61
Colao A, Vitale G, Cappabianca P, Briganti F, Ciccarelli A, De Rosa M, Zarrilli S, Lombardi G (2004) Outcome of cabergoline treatment in men with prolactinoma: effects of a 24-month treatment on prolactin levels, tumor mass, recovery of pituitary function, and semen analysis. J Clin Endocrinol Metab 89(4):1704–1711
Shimon I, Benbassat C, Hadani M (2007) Effectiveness of long-term cabergoline treatment for giant prolactinoma: study of 12 men. Eur J Endocrinol 156(2):225–231
Minniti G, Jaffrain-Rea ML, Santoro A, Esposito V, Ferrante L, Delfini R, Cantore G (2002) Giant prolactinomas presenting as skull base tumors. Surg Neurol 57(2):99–103 (discussion 103–104)
Acharya SV, Gopal RA, Menon PS, Bandgar TR, Shah NS (2010) Giant prolactinoma and effectiveness of medical management. Endocr Pract 16(1):42–46
Molitch ME (2003) Dopamine resistance of prolactinomas. Pituitary 6(1):19–27
Symon L, Jakubowski J, Kendall B (1979) Surgical treatment of giant pituitary adenomas. J Neurol Neurosurg Psychiatry 42(11):973–982
Pia HW, Grote E, Hildebrandt G (1985) Giant pituitary adenomas. Neurosurg Rev 8(3–4):207–220
Vroonen L, Jaffrain-Rea ML, Petrossians P, Tamagno G, Chanson P, Vilar L, Borson-Chazot F, Naves LA, Brue T, Gatta B, Delemer B, Ciccarelli E, Beck-Peccoz P, Caron P, Daly AF, Beckers A (2012) Prolactinomas resistant to standard doses of cabergoline: a multicenter study of 92 patients. Eur J Endocrinol 167(5):651–662
Odin P, Oehlwein C, Storch A, Polzer U, Werner G, Renner R, Shing M, Ludolph A, Schuler P (2006) Efficacy and safety of high-dose cabergoline in Parkinson’s disease. Acta Neurol Scand 113(1):18–24
Moraes AB, Silva CM, Vieira Neto L, Gadelha MR (2013) Giant prolactinomas: the therapeutic approach. Clin Endocrinol (Oxf) 79(4):447–456
Acknowledgments
This study has been supported by a small grant from the College of Medicine, King Fahad Medical City, Riyadh, Saudi Arabia.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Almalki, M.H., Buhary, B., Alzahrani, S. et al. Giant prolactinomas: clinical manifestations and outcomes of 16 Arab cases. Pituitary 18, 405–409 (2015). https://doi.org/10.1007/s11102-014-0588-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-014-0588-3