Abstract
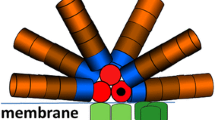
Ultrafast time resolved emission spectra were measured in whole cells of a PSI-deficient mutant of Synechocystis sp. PCC 6803 at room temperature and at 77K to study excitation energy transfer and trapping. By means of a target analysis it was estimated that the terminal emitter of the phycobilisome, termed allophycocyanin 680, transfers its energy with a rate of (20 ps)–1 to PSII. This is faster than the intraphycobilisome energy transfer rates between a rod and a core cylinder, or between the core cylinders.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Change history
28 March 2018
The original article has been corrected. The article is published with Open Access but was missing Open Access information. This has been added.
Abbreviations
- APC:
-
allophycocyanin
- DAS:
-
decay-associated spectrum
- ΔPSI:
-
PSI-deficient mutant of Synechocystis sp. PCC 6803
- EAS:
-
evolution-associated spectrum
- ET:
-
electron transfer
- EET:
-
excitation energy transfer
- FWHM:
-
full width at half maximum
- IRF:
-
instrument response function
- PB:
-
phycobilisome
- PC:
-
phycocyanin
- rms:
-
root mean square
- RP:
-
radical pair
- SAS:
-
species-associated spectrum
- SNR:
-
signal to noise ratio
- SS:
-
steady-state spectra
- SVD:
-
singular value decomposition
- TRES:
-
time resolved emission spectrum
- WT:
-
wild type
References
Acuña A.M., Kaňa R., Gwizdala M. et al: A method to decompose spectral changes in Synechocystis PCC 6803 during light-induced state transitions.–Photosynth. Res. 130: 237–249, 2016a.
Acuña A.M., Snellenburg J.J., Gwizdala M. et al.: Resolving the contribution of the uncoupled phycobilisomes to cyanobacterial pulse-amplitude modulated (PAM) fluorometry signals.–Photosynth. Res. 127: 91–102, 2016b.
Adir N.: Elucidation of the molecular structures of components of the phycobilisome: reconstructing a giant.–Photosynth. Res. 85: 15–32, 2005.
Arteni A.A., Ajlani G., Boekema E.J.: Structural organisation of phycobilisomes from Synechocystis sp strain PCC6803 and their interaction with the membrane.–Biochim. Biophys. Acta 1787: 272–279, 2009.
Gillbro T., Sandstrom A., Sundstrom V. et al.: Picosecond study of energy-transfer kinetics in phycobilisomes of Synechococcus 6301 and the mutant AN 112.–Biochim. Biophys. Acta 808: 52–65, 1985.
Glazer A.N.: Phycobilisome - a macromolecular complex optimized for light energy-transfer.–Biochim. Biophys. Acta 768: 29–51, 1984.
Govindjee, Shevela D., Björn L.O.: Evolution of the Z-scheme of photosynthesis: a perspective.–Photosynth. Res. 133: 5–15, 2017.
Holzwarth A.R.: Data analysis of time-resolved measurements.–In: Amesz J., Hoff A.J. (ed.): Biophysical Techniques in Photosynthesis. Pp. 75–92. Kluwer, Dordrecht 1996.
Jallet D., Gwizdala M., Kirilovsky D.: ApcD, ApcF and ApcE are not required for the Orange Carotenoid Protein related phycobilisome fluorescence quenching in the cyanobacterium Synechocystis PCC 6803.–Biochim. Biophys. Acta 1817: 1418–1427, 2012.
Liu H., Zhang H., Niedzwiedzki D.M. et al.: Phycobilisomes Supply excitations to both photosystems in a megacomplex in cyanobacteria.–Science 342: 1104–1107, 2013.
Maksimov E.G., Kuzminov F.I., Konyuhov I.V. et al.: Photosystem 2 effective fluorescence cross-section of cyanobacterium Synechocystis sp. PCC6803 and its mutants.–J. Photoch. Photobio. B 104: 285–291, 2011.
Mirkovic T., Ostroumov E.E., Anna J.M. et al.: Light absorption and energy transfer in the antenna complexes of photosynthetic organisms.–Chem. Rev. 117: 249–293, 2017.
Sandström A., Gillbro T., Sundström V. et al.: Picosecond study of energy-transfer within 18-S particles of AN-112 (a mutant of Synechococcus 6301) phycobilisomes.–BBABioenergetics 933: 54–64, 1988.
Scott M., McCollum C., Vasil’ev S. et al.: Mechanism of the down regulation of photosynthesis by blue light in the cyanobacterium Synechocystis sp PCC 6803.–Biochemistry 45: 8952–8958, 2006.
Shen G., Boussiba S., Vermaas W.F.: Synechocystis sp PCC 6803 strains lacking photosystem I and phycobilisome function.–Plant Cell 5: 1853–1863, 1993.
Snellenburg J.J., Dekker J.P., van Grondelle R. et al.: Functional compartmental modeling of the photosystems in the thylakoid membrane at 77 K.–J. Phys. Chem. B 117: 11363–11371, 2013.
Snellenburg J.J., Wlodarczyk L.M., Dekker J.P. et al: A model for the 77 K excited state dynamics in Chlamydomonas reinhardtii in state 1 and state 2.–Biochim. Biophys. Acta 1858: 64–72, 2017.
Tian L., Farooq S., van Amerongen H.: Probing the picosecond kinetics of the photosystem II core complex in vivo.–Phys. Chem. Chem. Phys. 15: 3146–3154, 201
Tian L., Gwizdala M., van Stokkum I.H.M. et al.: Picosecond kinetics of light harvesting and photoprotective quenching in wild-type and mutant phycobilisomes isolated from the cyanobacterium Synechocystis PCC 6803.–Biophys. J. 102:1692–1700, 2012.
Tian L., van Stokkum I.H.M., Koehorst R.B.M. et al.: Site, rate, and mechanism of photoprotective quenching in cyanobacteria.–J. Am. Chem. Soc. 133: 18304–18311, 2011.
Tian L., van Stokkum I.H.M., Koehorst R.B.M. et al.: Light harvesting and blue-green light induced non-photochemical quenching in two different c-phycocyanin mutants of Synechocystis PCC 6803.–J. Phys. Chem. B 117: 11000–11006, 2013b.
van Grondelle R., Dekker J.P., Gillbro T. et al.: Energy transfer and trapping in photosynthesis.–BBA-Bioenergetics 1187: 1–65, 1994.
van Stokkum I.H.M., Gwizdala M., Tian L. et al.: A functional compartmental model of the Synechocystis PCC 6803 phycobilisome.–DOI: 10.1007/s11120-017-0424-5, 2017.
van Stokkum I.H.M., Larsen D.S., van Grondelle R.: Global and target analysis of time-resolved spectra.–BBA-Bioenergetics 1657: 82–104, 2004.
van Stokkum I.H.M., van Oort B., van Mourik F. et al.: (Sub)-Picosecond spectral evolution of fluorescence studied with a synchroscan streak-camera system and target analysis.–In: Aartsma T.J., Matysik J. (ed.): Biophysical Techniques in Photosynthesis. Pp. 223–240. Springer, Dordrecht 2008.
Watanabe M., Ikeuchi M.: Phycobilisome: architecture of a lightharvesting supercomplex.–Photosynth. Res. 116: 265–276, 2013.
Wlodarczyk L.M., Dinc E., Croce R. et al.: Excitation energy transfer in Chlamydomonas reinhardtii deficient in the PSI core or the PSII core under conditions mimicking state transitions.–Biochim. Biophys. Acta 1857: 625–633, 2016.
Xie J., Zhao J.-Q., Peng C.: Analysis of the disk-to-disk energy transfer processes in C-phycocyanin complexes by computer simulation technique.–Photosynthetica 40: 251–257, 2002
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgments: The ΔPSI mutant which was originally constructed by Wim Vermaas (Arizona State University, Tempe, Arizona, USA) was kindly provided to us by Christiane Funk (Umeå University, Sweden). This research was performed as part of the BioSolar Cells research program, sponsored by the Dutch Ministry of Economic Affairs. This work is part of the research program of the Netherlands Organization for Scientific Research (NWO, previously FOM). IHMvS and RvG acknowledge financial support of the European Research Council [Advanced Grant proposal 267333 (PHOTPROT) to RvG].
This article is published with open access at link.springer.com
A correction to this article is available at https://doi.org/10.1007/s11099-018-0797-5
Electronic supplementary material
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Acuña, A.M., Van Alphen, P., Van Grondelle, R. et al. The phycobilisome terminal emitter transfers its energy with a rate of (20 ps)–1 to photosystem II. Photosynthetica 56, 265–274 (2018). https://doi.org/10.1007/s11099-018-0779-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-018-0779-7