Abstract
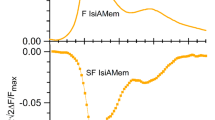
Energy transfer of aromatic amino acids in photosystem 2 (PS2) core antenna complexes CP43 and CP47 was studied using absorption spectroscopy, fluorescence spectroscopy, and the 0.35 nm crystal structure of PS2 core complex. The energy of tyrosines (Tyrs) was not effectively transferred to tryptophans (Trps) in CP43 and CP47. The fluorescence emission spectrum of CP43 and CP47 by excitation at 280 nm should be a superposition of the Tyr and Trp fluorescence emission spectra. The aromatic amino acids in CP43 and CP47 could transfer their energy to chlorophyll (Chl) a molecules by the Dexter mechanism and the Föster mechanism, and the energy transfer efficiency in CP47 was much higher than that in CP43. In CP47 the Föster mechanism must be the dominant energy transfer mechanism between aromatic amino acids and Chl a molecules, whereas in CP43 the Dexter mechanism must be the dominant one. Hence solar ultraviolet radiation brings not only damages but also benefits to plants.
Similar content being viewed by others
Abbreviations
- Chl:
-
chlorophyll
- GuHCl:
-
guanidine hydrochloride
- LHC2:
-
light-harvesting complex 2
- PS:
-
photosystem
- RC:
-
reaction centre
- Trp:
-
tryptophan
- Tyr:
-
tyrosine
- UV:
-
ultraviolet
- β-Car:
-
β-carotene
References
Alfonso, M., Montoya, G., Cases, R., Rodríguez, R., Picorel, R.: Core antenna complexes, CP43 and CP47, of higher plant photosystem II. Spectral properties, pigment stoichiometry, and amino acid composition.-Biochemistry 33: 10494–10500, 1994.
Barber, J., Morris, E., Büchel, C.: Revealing the structure of the photosystem II chlorophyll binding proteins, CP43 and CP47.-Biochim. biophys. Acta 1459: 239–247, 2000.
Barrow, C.J., Yasuda, A., Kenny, P.T.M., Zagorski, M.G.: Solution conformations and aggregational properties of synthetic amyloid β-peptides of Alzheimer disease analysis of circular dichroism spectra.-J. mol. Biol. 225: 1075–1093, 1992.
Bian, W., Wong, R., Sun, S., Zhou, Q.: Study on unfolding process of neo-trichosanthin in the presence of GdnHCl.-Spectrosc. spectr. Anal. 20: 492–494, 2000.
Bricker, T.M.: The structure and function of CPa-1 and CPa-2 in photosystem II.-Photosynth. Res. 24: 1–13, 1990.
Bricker, T.M., Frankel, L.K.: The structure and function of CP47 and CP43 in Photosystem II.-Photosynth. Res. 72: 131–146, 2002.
Burstein, E.A., Vedenkina, N.S., Ivkova, M.N.: Fluorescence and the location of tryptophan residues in protein molecules.-Photochem. Photobiol. 18: 263–279, 1973.
De Weerd, F.L., van Stokkum, I.H.M., van Amerongen, H., Dekker, J.P., van Grondelle, R.: Pathways for energy transfer in the core light-harvesting complexes CP43 and CP47 of photosystem II.-Biophys. J. 82: 1586–1597, 2002.
Dubey, V.K., Jagannadham, M.V.: Differences in the unfolding of procerain induced by pH, guanidine hydrochloride, urea, and temperature.-Biochemistry 42: 12287–12297, 2003.
Eftink, M.R., Shastry, M.C.R.: Fluorescence methods for studying kinetics of protein-folding reactions.-Methods Enzymol. 278: 258–286, 1997.
Ferreira, K.N., Iverson, T.M., Maghlaoui, K., Barber, J., So Iwata: Architecture of the photosynthetic oxygen-evolving center.-Science 303: 1831–1838, 2004.
Föster, T.: Delocalized excitation and excitation transfer.-In: Sinanoğlu, O. (ed.): Modern Quantum Chemistry. Part III. Pp. 93–137. Academic Press, New York 1965.
Ghanotakis, D.F., De Paula, J.C., Demetriou, D.M., Bowlby, N.R., Petersen, J., Babcock, G.T., Yocum, D.F.: Isolation and characterization of the 47 kDa protein and the D1–D2-cytochrome b-559 complex.-Biochim. biophys. Acta 974: 44–53, 1989.
Groot, M.L., Peterman, E.J., van Stokkum, I.H., Dekker, J.P., van Grondelle, R.: Triplet and fluorescing states of the CP47 antenna complex of photosystem II studied as a function of temperature.-Biophys. J. 68: 281–290, 1995.
Guo, S.K., Tang, C.Q., Yang, Z.L., Li, L.B., Kuang, T.Y., Gong, Y.D., Zhao, N.M.: Effects of acid and alkali on the light absorption, energy transfer and protein secondary structures of core antenna subunits CP43 and CP47 of Photosystem II.-Photochem. Photobiol. 79: 291–296, 2004.
Henderson, R., Baldwin, J.M., Ceska, T.A., Zemlin, F., Beckmann, E., Downing, K.H.: Model for the structure of bacteriorhodopsin based on high-resolution electron cryomicroscopy.-J. mol. Biol. 213: 899–929, 1990.
Isaev-Ivanov, V.V., Kozlov, M.G., Baitin, D.M., Masui, R., Kuramitsu, S., Lanzov, V.A.: Fluorescence and excitation Escherichia coli recA protein spectra analyzed separately for Tyr and tryptophan residues.-Arch. Biochem. Biophys. 376: 124–140, 2000.
Kalbin, G., Li, S.S., Olsman, H., Pettersson, M., Engwall, M., Åke Strid: Effects of UV-B in biological and chemical systems: Equipment for wavelength dependence determination.-J. biochem. biophys. Methods 65: 1–12, 2005.
Kalisky, O., Feitelson, J., Ottolenghi, M.: Photochemistry and fluorescence of bacteriorhodopsin excited in its 280-nm absorption band.-Biochemistry 20: 205–209, 1981.
Kelly, S.M., Price, N.C.: The application of circular dichroism to studies of protein folding and unfolding.-Biochim. biophys. Acta 1338: 161–165, 1997.
Kuwabara, T., Murata, N.: Inactivation of photosynthetic oxygen evolution and concomitant release of three polypeptides in the photosystem II particles of spinach chloroplasts.-Plant Cell Physiol. 23: 533–539, 1982.
Lakowicz, J.R.: Principles of Fluorescence Spectroscopy.-Pp. 342–381. Plenum Press, New York-London 1983.
Nozaki, Y.: The preparation of guanidine hydrochloride.-Methods Enzymol. 26: 43–51, 1972.
Oikawa, K., Lieberman, D.M., Reithmeier, R.A.F.: Conformation and stability of the anion transport protein of human erythrocyte membranes.-Biochemistry 24: 2843–2848, 1985.
Pace, C.N.: Determination and analysis of urea and guanidine hydrochloride denaturation curves.-Methods Enzymol. 131: 266–280, 1986.
Shan, J.X., Wang, J.S., Li, L.B., Zhao, N.M., Kuang, T.Y.: The excitation energy transfer between β-Car and Chl a molecules in PSII core antenna complexes CP43 and CP47.-Chin. Sci. Bull. 45: 1579–1583, 2000.
Shan, J.X., Wang, J.S., Ruan, X., Li, L.B., Gong, Y.D., Zhao, N.M., Kuang, T.Y.: Changes of absorption spectra during heat-induced denaturation of photosystem II core antenna complexes CP43 and CP47: revealing the binding states of chlorophyll molecules in these two complexes.-Biochim. biophys. Acta 1504: 396–408, 2001.
Shutova, T., Deikus, G., Irrgang, K.D., Klimov, V.V., Renger, G.: Origin and properties of fluorescence emission from the extrinsic 33 kDa manganese stabilizing protein of higher plant water oxidizing complex.-Biochim. biophys. Acta 1504: 371–378, 2001.
Tanaka, N., Kajimoto, S., Mitani, D., Kunugi, S.: Effects of guanidine hydrochloride and high pressure on subsite flexibility of β-amylase.-Biochim. biophys. Acta 1596: 318–325, 2002.
Van Grondelle, R., Dekker, J.P., Gillbro, T., Sundstrom, V.: Energy transfer and trapping in photosynthesis.-Biochim. biophys. Acta 1187: 1–65, 1994.
Wang, J.S., Shan, J.X., Xu, Q., Ruan, X., Gong, Y.D., Kuang, T.Y., Zhao, N.M.: Spectroscopic study of trypsin, heat and Triton X-100-induced denaturation of the chlorophyll-binding protein CP43.-J. Photochem. Photobiol. 58: 136–142, 2000.
Wang, J.S., Shan, J.X., Xu, Q., Ruan, X., Gong, Y.D., Kuang, T.Y., Zhao, N.M.: Light-and heat-induced denaturation of photosystem II core-antenna complexes CP43 and CP47.-J. Photochem. Photobiol. 50: 189–196, 1999.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Qu, YG., Qin, XC., Wang, WF. et al. Energy transfer of aromatic amino acids in photosystem 2 core antenna complexes CP43 and CP47. Photosynthetica 45, 266–271 (2007). https://doi.org/10.1007/s11099-007-0043-z
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11099-007-0043-z