Abstract
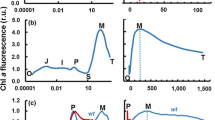
Fluorescence parameters obtained during steady-state electron transport are frequently used to evaluate photosynthetic efficiency of plants. We studied the behaviour of those parameters as a function of irradiance-adapted fluorescence yields FS and F'M. Applied simulations showed that photochemical quenching evaluated by qP is greatly influenced by the steady-state fluorescence level (FS), and that its evolution is not complementary to non-photochemical quenching (qN). On the other hand, the relative photochemical and non-photochemical quenching coefficients (qP(rel) and qN(rel)) proposed by Buschmann (1995) represent better the balance between the energy dissipation pathways. However, these relative parameters are also non-linearly related when the FS level is varied. We investigated the application of a new parameter, the relative unquenched fluorescence (UQF(rel)) which takes into account the fraction of non-quenched fluorescence yield (FS), which is related to closed photosystem 2 reaction centres not participating in electron transport. By using computer simulations and real in vivo measurements, we found that this new parameter is complementary to qP(rel) and qN(rel), which may facilitate the use of PAM fluorescence as diagnostic tool in environmental studies.
Similar content being viewed by others
Abbreviations
- Chl:
-
chlorophyll
- F0 :
-
minimum fluorescence yield in dark-adapted state
- F'0 :
-
minimum fluorescence yield in light-adapted state
- FM :
-
maximum fluorescence yield in dark-adapted state
- F'M :
-
maximum fluorescence yield in light-adapted state
- FS :
-
steady-state fluorescence level in light-adapted state
- LHC:
-
light-harvesting complex
- PAM:
-
Pulse-Amplitude-Modulated
- PS:
-
photosystem
- qN and qN(rel) :
-
non-photochemical and relative non-photochemical quenching
- qP and qP(rel) :
-
photochemical and relative photochemical quenching
- RCs:
-
reaction centres
- UQF(rel) :
-
relative unquenched fluorescence
- Φ'M :
-
effective photosystem 2 quantum yield
References
Barón, M., Arellano, J.B., López Gorgé, J.: Copper and photosystem II: A controversial relationship. — Physiol. Plant. 94: 174–180, 1995.
Bilger, W., Björkman, O.: Role of the xanthophyll cycle in photoprotection elucidated by measurements of light-induced absorbance changes, fluorescence and photosynthesis in leaves of Hedera canariensis. — Photosynth. Res. 25: 173–185, 1990.
Björkman, O., Demmig, B.: Photon yield of O2 evolution and chlorophyll fluorescence characteristics at 77 K among vascular plants of diverse origins. — Planta 170: 489–504, 1987.
Bolhàr-Nordenkampf, H.R., Long, S.P., Baker, N.R., Öquist, G., Schreiber, U., Lechner, E.G.: Chlorophyll fluorescence as a probe of the photosynthetic competence of leaves in the field: a review of current instrumentation. — Funct. Ecol. 3: 497–514, 1989.
Brack, W., Frank, H.: Chlorophyll a fluorescence: A tool for the investigation of toxic effects in the photosynthetic apparatus. — Ecotoxicol. environ. Safety 40: 34–41, 1998.
Buschmann, C.: Variation of the quenching of chlorophyll fluorescence under different intensities of the actinic light in wild-type plants of tobacco and in an Aurea mutant deficient of light-harvesting-complex. — J. Plant Physiol. 145: 245–252, 1995.
Buschmann, C.: Photochemical and non-photochemical quenching coefficients of the chlorophyll fluorescence: comparison of variation and limits. — Photosynthetica 37: 217–224, 1999.
Demmig-Adams, B., Adams, W.W., III, Barker, D.H., Logan, B.A., Bowling, D.R., Verhoeven, A.S.: Using chlorophyll fluorescence to assess the fraction of absorbed light allocated to thermal dissipation of excess excitation. — Physiol. Plant. 98: 253–264, 1996.
El Jay, A., Ducruet, J.-M., Duval, J.-C., Pelletier, J.P.: A high-sensitivity chlorophyll fluorescence assay for monitoring herbicide inhibition of photosystem II in the chlorophyte Selenastrum capricornutum: Comparison with effect on cell growth. — Arch. Hydrobiol. 140: 273–286, 1997.
Flameling, I.A., Kromkamp, J.: Light dependence of quantum yields for PSII charge separation and oxygen evolution in eucaryotic algae. — Limnol. Oceanogr. 43: 284–297, 1998.
Frankart, C., Eullaffroy, P., Vernet, G.: Comparative effects of four herbicides on non-photochemical fluorescence quenching in Lemna minor. — Environ. exp. Bot. 49: 159–168, 2003.
Genty, B., Briantais, J.-M., Baker, N.R.: The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. — Biochim. biophys. Acta 990: 87–92, 1989.
Georgieva, K., Yordanov, I.: Temperature dependence of photochemical and non-photochemical fluorescence quenching in intact pea leaves. — J. Plant Physiol. 144: 754–759, 1994.
Heinze, I., Dau, H., Senger, H.: The relation between the photochemical yield and variable fluorescence of photosystem II in the green alga Scenedesmus obliquus. — J. Photochem. Photobiol. B 32: 89–95, 1996.
Joliot, A., Joliot, P.: Étude cinétique de la réaction photochimique libérant l'oxygène au cours de la photosynthèse. — Compt. rend. Séances Acad. Sci. Paris 258: 4622–4625, 1964.
Juneau, P., Dewez, D., Matsui, S., Kim, S.-G., Popovic, R.: Evaluation of different algal species sensitivity to mercury and metolachlor by PAM-fluorometry. — Chemosphere 45: 589–598, 2001.
Juneau, P., El Berdey, A., Popovic, R.: PAM-fluorometry in the determination of the sensitivity of Chlorella vulgaris, Selenatrum capricornutum and Chlamydomonas reinhardtii to copper. — Arch. environ. Contamin. Toxicol. 42: 155–164, 2002.
Juneau, P., Lawrence, J.E., Suttle, C.A., Harrison, P.J.: Effects of viral infection on photosynthetic processes in the bloom-forming alga Heterosigma akashiwo. — Aquat. Microb. Ecol. 31: 9–17, 2003.
Juneau, P., Popovic, R.: Evidence for the rapid phytotoxicity and environmental stress evaluation using the PAM fluorometric method: Importance and future application. — Ecotoxicology 8: 449–455, 1999.
Kramer, D.M., Johnson, G., Kiirats, O., Edwards, G.E.: New fluorescence parameters for determination of QA redox state and excitation energy fluxes. — Photosynth. Res. 79: 209–218, 2004.
Krause, G.H., Jahns, P.: Pulse amplitude modulated chlorophyll fluorometry and its application in plant science. — In: Green, B.R., Parson, W.W. (ed.): Light-harvesting Antennas in Photosynthesis. Pp. 373–399. Kluwer Academic Publ., Dordrecht 2003.
Krause, G.H., Weis, E.: Chlorophyll fluorescence as a tool in plant physiology. II. Interpretation of fluorescence signals. — Photosynth. Res. 5: 139–157, 1984.
Krause, G.H., Weis, E.: Chlorophyll fluorescence and photosynthesis: The basics. — Annu. Rev. Plant Physiol. Plant mol. Biol. 42: 313–349, 1991.
Lavergne, J., Trissl, H.-W.: Theory of fluorescence induction in photosystem II: Derivation of analytical expressions in a model including exciton-radical-pair equilibrium and restricted energy transfer between photosynthetic units. — Biophys. J. 68: 2474–2492, 1995.
Lazàr, D.: Chlorophyll a fluorescence induction. — Biochim. biophys. Acta 1412: 1–28, 1999.
Lichtenthaler, H.K., Rinderle, U.: The role of chlorophyll fluorescence in the detection of stress conditions in plants. — CRC crit. Rev. anal. Chem. 19: S29–S85, 1988.
Lippemeier, S., Frampton, D.M.F., Blackburn, S.I., Geier, S.C., Negri, A.P.: Influence of phosphorus limitation on toxicity and photosynthesis of Alexandrium minutum (dinophyceae) monitored by in-line detection of variable chlorophyll fluorescence. — J. Phycol. 38: 320–331, 2003.
Lippemeier, S., Hartig, P., Colijn, F.: Direct impact of silicate on the photosynthetic performance of the diatom Thalassiosira weissflogii assessed by on-and off-line PAM fluorescence measurements. — J. Plankton Res. 21: 269–283, 1999.
Naessens, M., Leclerc, J.C., Tran-Minh, C.: Fiber optic biosensor using Chlorella vulgaris for determination of toxic compounds. — Ecotoxicol. environ. Safety 46: 181–185, 2000.
Roháçek, K.: Chlorophyll fluorescence parameters: the definitions, photosynthetic meaning, and mutual relationships. — Photosynthetica 40: 13–29, 2002.
Schreiber, U., Bilger, W., Neubauer, C.: Chlorophyll fluorescence as a nonintrusive indicator for rapid assessment of in vivo photosynthesis. — In: Schulze, E.D., Caldwell, M.M. (ed.): Ecophysiology of Photosynthesis. Pp. 49–70. Springer-Verlag, Berlin 1994.
Schreiber, U., Schliwa, U., Bilger, W.: Continuous recording of photochemical and non-photochemical chlorophyll fluorescence quenching with a new type of modulation fluorometer. — Photosynth. Res. 10: 51–62, 1986.
USEPA: Algal assay procedure bottle test. — United States Environmental Protection Agency, Corvallis 1971.
Weis, E., Berry, J.A.: Quantum efficiency of Photosystem II in relation to ‘energy’-dependent quenching of chlorophyll fluorescence. — Biochim. biophys. Acta 894: 198–208, 1987.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Juneau, P., Green, B.R. & Harrison, P.J. Simulation of Pulse-Amplitude-Modulated (PAM) fluorescence: Limitations of some PAM-parameters in studying environmental stress effects. Photosynthetica 43, 75–83 (2005). https://doi.org/10.1007/s11099-005-5083-7
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11099-005-5083-7