Abstract
Backgound
Conbercept and ranibizumab have been used to treat retinopathy of prematurity (ROP). However, the clinical effectiveness of conbercept and ranibizumab remains controversial.
Aim
This meta-analysis aimed to compare the effectiveness of conbercept and ranibizumab for treating ROP.
Method
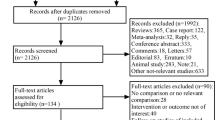
We systematically searched Pubmed, Web of Science, Embase, the Cochrane Library, Ovid, Scopus, China National Knowledge Infrastructure, Wanfang Database, CQVIP, Duxiu Database, SinoMed, and X-MOL to screen relevant studies up to November 2022. Retrospective cohort studies and randomized controlled trials (RCTs) evaluating the effectiveness of conbercept and ranibizumab in treating patients with ROP were selected. The outcomes assessed were the rates of primary cure, ROP recurrence, and retreatment. Statistical analysis was performed using Stata.
Results
Seven studies (n = 989) were selected in the meta-analysis. There were 303 cases (594 eyes) treated with conbercept and 686 patients (1,318 eyes) treated with ranibizumab. Three studies reported the primary cure rate. Compared to ranibizumab, conbercept had a significantly higher primary cure rate (odds ratio [OR] 1.91, 95% confidence interval [CI] 1.05–3.49, P < 0.05). Five studies reported the rate of ROP recurrence, and there were no significant differences between conbercept and ranibizumab (OR 0.62, 95% CI 0.28–1.38, P > 0.05). Three studies reported the rate of retreatment, and the rates were not significantly different between conbercept and ranibizumab (OR 0.78, 95% CI 0.21–2.93, P > 0.05).
Conclusion
Conbercept had a higher rate of primary cure in ROP patients. More RCTs are needed to compare the efficacy of conbercept and ranibizumab in treating ROP.


Similar content being viewed by others
References
Vinekar A, Sinha S, Mohan A, et al. Role of birth order in the development of retinopathy of prematurity in multiple gestation: a study of 1040 Asian Indian premature infants. SN Compr Clin Med. 2022;4(55):5–7.
Du M, Chen T, Yi Su, et al. Research progress on the risk factors of retinopathy of prematurity. Eye Sci. 2020;35(4):249–54.
Justiniano-Becerra YG, Ramirez-Solis F, Aguilar-Morales K, et al. Retinopathy of prematurity: 5 years and 715 patients, an epidemiological analysis. Invest Ophthalmol Vis Sci. 2022;63(7):4201–F0261.
Sankar MJ, Sankar J, Chandra P. Anti-vascular endothelial growth factor (VEGF) drugs for treatment of retinopathy of prematurity. Cochrane Database Syst Rev. 2018;1(1):CD009734.
Jin E, Yin H, Wang Z, et al. Comparison of visual function and fundus structure outcomes in long-term follow-up of retinopathy of prematurity undergoing anti-vascular endothelia growth factor agents and laser treatment. Chin J Ocul Fundus Dis. 2022;38(7):546–50.
Qi X, Chao Z, Xiao J, et al. Anti-VEGF and laser therapy for type 1 retinopathy of prematurity: a Meta analysis. Int Eye Sci. 2021;21(2):285–90.
Kenneth TE, Kertes PJ. Ranibizumab in neovascular age-related macular degeneration. Clin Interv Aging. 2006;1(4):451–66.
Lu X, Sun X. Profile of conbercept in the treatment of neovascular age-related macular degeneration. Drug Des Devel Ther. 2015;22(9):2311–20.
Soll RF. Treatment of retinopathy of prematurity: moving forward with uncertainty. JAMA. 2022;328(4):332–3.
Chen X, Lu H, Wang T. Progress of the application of conbercept in ocular neovascular diseases. IES. 2022;22(8):1361–4.
Pediatric Retinology Group of Ophthalmology Professional Committee of Cross-Strait Medical and Health Exchange Association, Fundus Disease Group of Chinese Medical Association Ophthalmology Branch. Expert consensus on intravitreal injection of anti-vascular endothelial growth factor drugs for retinopathy of prematurity. Chin J Ocular Fundus Dis. 2021;37(11):836–40.
Cao R, Li Y, Liu W, et al. Meta-analysis of comparison of IVB and IVR used for retinopathy of prematurity. IES. 2021;21(3):487–92.
Shi R, Yang L, Qi Y, et al. Comparison of conbercept with ranbizumab for the treatment efficiency of macular edema: a meta-analysis. IES. 2017;17(12):2242–7.
Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25(9):603–5.
Ai F, Hu K, Shi J, et al. Quality assessment of cohort studies literature on Chinese smoking by using Newcastle-Ottawa-Scale. Chin J Disease Cont Prevent. 2021;25(6):722–9.
Jadad AR, Moore RA, Carroll D, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17(1):1–12.
Li M, Wang L, Zhang Z, et al. Comparison of the effectiveness of conbercept versus rRanibizumab in the treatment of type 1 retinopathy of prematurity. IES. 2021;21(5):866–71.
Cheng Y, Zhu X, Linghu D, et al. Comparison of the effectiveness of conbercept and ranibizumab treatment for retinopathy of prematurity. Acta Ophthalmol. 2020;98(8):e1004–8.
Jin E, Yin H, Li X, et al. Short-term outcomes after intravitreal injections of conbercept verus ranibizumab for the treatment of retinopathy of prematurity. Retina. 2018;38(8):1595–604.
Zheng B. Anti-vascular endothelial growth factor in the treatment of retinopathy of prematurity. J Fujian Med Univ. 2020:1–18.
Wang Z, Li Q, Yang X, et al. Comparison of the treatment outcomes for retinopathy of prematurity with an intravitreal injection of conbercept or ranibizumab. Chin J Optometry Ophthalmol Visual Sci. 2021;23(11):858–63.
Yang D. Epidemiological study of retinopathy of prematurity in dalian and short term efficacy of Anti-VEGF therapy. J Dalian Med Univ. 2019:1–34.
Wu Z, Zhao J, Lam W, et al. Comparison of clinical outcomes of conbercept versus ranibizumab treatment for retinopathy of prematurity: a multicentral prospective randomised controlled trial. Br J Ophthalmol. 2022;106(7):975–9.
Liu W, Li Y, Cao R, et al. A systematic review and meta-analysis to compare the efficacy of conbercept with ranibizumab in patients with macular edema secondary to retinal vein occlusion. Medicine (Baltimore). 2020;99(21): e20222.
Bai F, Ma X, Yan Y, et al. Comparison of the efficacy of ranibizumab and conbercept in the treatment of age-related macular degeneration. Chin J Gerontol. 2020;40(04):826–8.
Liu J, Gong J. Effects of ranibizumab and compaqip on BCVA, PLT and CMT in branch vein occlusion combined with macular edema. J Guangxi Univ(Natural Science). 2020;45(2):458–63.
Zhang M, Yu D, Yang C, et al. The pharmacology study of a new recombinant human VEGF receptor-fc fusion protein on experimental choroidal neovascularization. Pharm Res. 2009;26(1):204–10.
Stephens RJ, Dettmer MR, Roberts BW, et al. Practice patterns and outcomes associated with early sedation depth in mechanically ventilated patients: a systematic review and meta-analysis. Crit Care Med. 2018;46(3):471–9.
Alipanah N, Jarlsberg L, Miller C, et al. Adherence interventions and outcomes of tuberculosis treatment: a systematic review and meta-analysis of trials and observational studies. PLoS Med. 2018;15(7): e1002595.
Suárez-Iglesias D, Miller KJ, Seijo-Martínez M, et al. Benefits of Pilates in Parkinson’s Disease: A Systematic Review and Meta-Analysis. Medicina. 2019;55(8):47.
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jiang, S., Li, X., Fu, M. et al. Comparison of clinical effectiveness of conbercept and ranibizumab for treating retinopathy of prematurity: a meta-analysis. Int J Clin Pharm 45, 1317–1325 (2023). https://doi.org/10.1007/s11096-023-01584-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11096-023-01584-y