Abstract
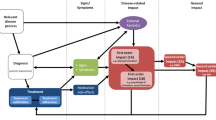
Cognitive pharmacy trials seek to identify interventions that benefit patients. The potential benefits of an intervention are primarily evaluated by outcome measures. The question then is: What is the optimal outcome measure? Unfortunately, the question remains unsolved. Several factors must be taken into consideration when conducting outcome research—particularly within cognitive pharmacy trials. The interventions are often complex and non-specific, and seek to improve symptom control, optimise the use of medications and reduce medication-related risks. “Hard” endpoints, such as mortality and hospital admissions, may not be the optimal outcome measures, since cognitive pharmacy interventions are unlikely to result in changes in these measures. Instead, adverse drug events or “soft” endpoints, such as quality of life, drug-related problems and patient satisfaction may be appropriate choices of outcome measures. Finally, it is not only outcome measures that may pose a challenge when conducting outcome research; other essential components include study design, type of intervention, the patient population, etc.
Similar content being viewed by others
References
Wong ICK. RESPECT Team. Randomised controlled trials (RCTs) to evaluate complex healthcare interventions—a case study. Pharm World Sci. 2004;26(5):247–52.
Krska J, Rowe PH. Outcome measures: a sensitive approach. Int J Pharm Pract. 2010;18:125–7.
Graabaek T, Kjeldsen LJ. Medication reviews by clinical pharmacists at hospitals lead to improved patient outcomes: a systematic review. Basic Clin Pharmacol Toxicol. 2013;112(6):359–73.
Emmerton L, Marriot J, Bessell T, et al. Pharmacists and prescribing rights: review of international developments. J Pharm Pharm Sci. 2005;8:217–25.
Frenk J. Obituary of avedis donabedian. Bull World Health Organ. 2000;70(12):1475.
Donabedian A. The quality of care: how can it be assessed? JAMA. 1988;121(11):1145–50.
Clarke M. Standardising outcomes for clinical trials and systematic reviews. Trials. 2007;8:39.
Craig P, Dieppe P, Macintyre S, Michie S, Nazareth I, Petticrew M. Developing and evaluating complex interventions: new guidance. 2008: http://www.bmj.com/content/337/bmj.a1655.
Bowling A. Research methods in health: investigating health and health services. 2nd ed. Buckingham: Open University Press; 2002.
Aronson JK. Biomarkers and surrogate endpoints. Br J Clin Pharmacol. 2005;59(5):491–4.
Cohn JN. Introduction to surrogate markers. Circulation. 2004;109(25 Suppl 1):IV20–1.
Wieczorek A, Rys P, Skrzekowska-Baran I, Malecki M. The role of surrogate endpoints in the evaluation of efficacy and safety of therapeutic interventions in diabetes mellitus. Rev Diabet Stud. 2008;5(3):128–35.
Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health-survery (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30:473–83.
Pickard AS, Johnson JA, Farris KB. The impact of pharmacist interventions on health-related quality of life. Ann Pharmacother. 1999;33(11):1167–72.
Jepsen P, Johnsen SP, Gillman MW, Sørensen HT. Interpretation of observational studies. Heart. 2004;90:956–60.
Wiffen P. No evidence or evidence of no effect. Eur J Hosp Pharm. 2014;21(2):71.
Krass I. Quasi experimental designs in pharmacist intervention research. IJCP 2016:1–8.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Rights and permissions
About this article
Cite this article
Kjeldsen, L.J., Nielsen, T.R.H. & Olesen, C. The challenges of outcome research. Int J Clin Pharm 38, 705–708 (2016). https://doi.org/10.1007/s11096-016-0293-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11096-016-0293-6