Abstract
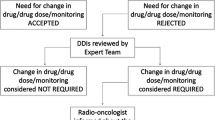
Objective of the study To identify the prevalence of potential drug–drug interactions between hospital pharmacy dispensed anti-cancer agents and community pharmacy dispensed drugs. Setting A retrospective cohort study was conducted on the haematology/oncology department of the internal medicine ward in a large teaching hospital in Amsterdam, the Netherlands. Method Prescription data from the last 100 patients treated with anti-cancer agents were obtained from Paracelsus, the chemotherapy prescribing system in the hospital. The community pharmacy dispensed drugs of these patients were obtained by using OZIS, a system that allows regionally linked pharmacies to call up active medication on any patient. Both medication lists were manually screened for potential drug–drug interactions by using several information sources on interactions, e.g. Pubmed, the Flockhart P450 table, Micromedex and Dutch reference books. Main outcome measure Prevalence of potential drug–drug interactions between anti-cancer agents provided by the hospital pharmacy and drugs dispensed by the community pharmacy. Results Ninety-one patients were included in the study. A total of 31 potential drug–drug interactions were found in 16 patients, of which 15 interactions were clinically relevant and would have required an intervention. Of these interactions 1 had a level of severity ≥D, meaning the potential drug–drug interaction could lead to long lasting or permanent damage, or even death. The majority of the interactions requiring an intervention (67%) had a considerable level of evidence (≥2) and were based on well-documented case reports or controlled interaction studies. Most of the potential drug–drug interactions involved the antiretroviral drugs (40%), proton pump inhibitors (20%) and antibiotics (20%). The anti-cancer drug most involved in the drug–drug interactions is methotrexate (33%). Conclusion This study reveals a high prevalence of potential drug–drug interactions between anti-cancer agents provided by the hospital pharmacy and drugs dispensed by the community pharmacy. It shows us there is need for an optimal medication surveillance mechanism to detect potential drug–drug interactions between these two groups of medication, especially because of the high toxicity of anticancer drugs and thus the severe consequences these interactions can have for the patient.
Similar content being viewed by others
References
Riechelmann RP, Tannock IF, Wang L, Saad ED, Taback NA, Krzyzanowska MK. Potential drug interactions and duplicate prescriptions among cancer patients. J Natl Cancer Inst. 2007;99(8):592–600.
Jaehde U, Liekweg A, Simons S, Westfeld M. Minimising treatment-associated risks in systemic cancer therapy. Pharm World Sci. 2008;30:161–8.
Paracelsus© Version 1.9. 2008. Farmaco-Logica, Onnen, The Netherlands.
Stoop AP, Bal R, Berg M. OZIS and the politics of safety: Using ICT to create a regionally accessible patient medication record. Int J Med Inf. 2007;76:229–35.
G-standaard: Central Dutch Database. The Hague; Winap c2008 http://kennisbank.knmp.nl/index.asp.
Micromedex® Healthcare Series. Greenwood Village, Colo; Thomson Reuters (Healthcare) Inc. http://www.thomsonhc.com/home/dispatch.
Pubmed. Bethesda, MD; US National Library of Medicine. http://www.ncbi.nlm.nih.gov/pubmed/.
Flockhart DA. Drug interactions: cytochrome P450 drug interaction table. Indiana University School of Medicine (2007). http://medicine.iupui.edu/clinpharm/ddis/table.asp. Accessed 8 June 2009.
Koninklijke Nederlandse Maatschappij ter bevordering der Pharmacie, Geneesmiddel Informatie Centrum. Informatorium Medicamentorum. The Hague: KNMP;2009 [Dutch].
de Gier JJ. Commentaren medicatiebewaking 2008/2009. Houten: Health Base; 2008. [Dutch].
College voor zorgverzekeringen. Farmacotherapeutisch Kompas 2009. Houten: Bohn Stafleu Van Loghum; 2009. [Dutch].
Uttamsingh V, Lu C, Miwa G, Gan L. Relative contributions of the five major human cytochromes P450, 1A2, 2C9, 2C19, 2D6, and 3A4, to the hepatic metabolism of the proteasome inhibitor bortezomib. Drug Metab Dispos. 2005;33(11):1723–8.
Zhou S. Drugs behave as substrates, inhibitors and inducers of human cytochrome P450 3A4. Curr Drug Metab. 2008;9:310–22.
Hiv-druginteractions. Liverpool: the University of Liverpool; c1999–2010 [cited: 2009 June 7]. http://www.hiv-druginteractions.org/.
Vaccher E, Spina M, di Gennaro G, Talamini R, Nasti G, Schioppa O, et al. Concomitant cyclophosphamide, doxorubicin, vincristine, and prednisone chemotherapy plus highly active antiretroviral therapy in patients with human immunodeficiency virus-related, non-hodgkin lymphoma. Cancer. 2001;91(1):155–63.
Toffoli G, Corona G, Cattarossi G, Boiocchi M, di Gennaro G, Tirelli U, et al. Effect of highly active antiretroviral therapy (HAART) on pharmacokinetics and pharmacodynamics of doxorubicin in patients with HIV-associated non-Hodgkin’s lymphoma. Ann Oncol. 2004;15:1805–9.
Candussio L, Decorti G, Crivellato E, Granzotto M, Rosati A, Giraldi T, et al. Toxicologic and pharmacokinetic study of low doses of verapamil combined with doxorubicin. Life Sci. 2002;71:3109–19.
Antoniou T, Tseng AL. Interactions between antiretrovirals and antineoplastic drug therapy. Clin Pharmacokinet. 2005;44(2):111–45.
Windle R, MacPherson S, Bell PRF. Neutropenia associated with metronidazole. Br Med J. 1979;2:1219.
Bardakji Z, Jolivet J, Langelier Y, Besner J, Ayoub J. 5-Fluorouracil-metronidazole combination therapy in metastatic colorectal cancer. Cancer Chemother Pharmacol. 1986;18:140–4.
Ferrazzini G, Klein J, Sulh H, Chung D, Griesbrecht E, Koren G. Interaction between trimethoprim-sulfamethoxazole and methotrexate in children with leukaemia. J Pediatr. 1990;117(5):823–6.
Steuer A, Gumpel JM. Methotrexate and trimethoprim: a fatal interaction. Br J Rheumatol. 1998;37:105–6.
Ng HWK, MacFarlane AW, Graham RM, Verbov JL. Near fatal drug interactions with methotrexate given for psoriasis. Br Med J (Clin Res Ed). 1987;295(6601):752–3.
Suzuki K, Doki K, Homma M, Tamaki H, Hori S, Ohtani H, et al. Co-administration of proton pump inhibitors delays elimination of plasma methotrexate in high-dose methotrexate therapy. Br J Clin Pharmacol. 2008;67(1):44–9.
Joerger M, Huitema ADR, van den Bongerd HJGD, Baas P, Schornagel JH, Schellens JHM, Beijnen JH. Determinants of the elimination of methotrexate and 7-hydroxy-methotrexate following high-dose infusional therapy to cancer patients. Br J Clin Pharmacol. 2005;62(1):71–80.
Tröger U, Stötzel B, Martens-Lobenhoffer J, Gollnick H, Meyer FP. Severe myalgia from an interaction between treatments with pantoprazole and methotrexate. Br Med J. 2002;324:1497.
Bauters TGM, Verlooy J, Robays H, Laureys G. Interactions between methotrexate and omeprazole in an adolescent with leukemia: a case report. Pharm World Sci. 2008;30:316–8.
Reid T, Yuen A, Catolico M, Carlson RW. Impact of omeprazole on the plasma clearance of methotrexate. Cancer Chemother Pharmacol. 1993;33:82–4.
Appelo DA, Berger-De Jong IEJ, Janssen MJA. Effect van preoperatieve farmacotherapeutische opnamegesprekken op de kwaliteit van het medicatieoverzicht op een orthopedische afdeling. Pharm Weekbl. 2008;3:8–13. [Dutch].
IKCnet. Amsterdam: Dutch cancer registration authority; c2009. http://www.ikcnet.nl/page.php?id=114. Accessed 12 June 2009 [Dutch].
Funding
None.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Voll, M.L., Yap, K.D., Terpstra, W.E. et al. Potential drug–drug interactions between anti-cancer agents and community pharmacy dispensed drugs. Pharm World Sci 32, 575–580 (2010). https://doi.org/10.1007/s11096-010-9410-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11096-010-9410-0