Abstract
Purpose
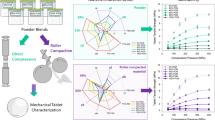
Using a high level of mannitol as a diluent in oral formulations can potentially result in tablet defects (e.g., chipping, cracking) during compression. This work aims to scrutinize the linkage between the mechanical properties and material attributes of mannitol and also uncover how variations between vendors and lots can lead to significant changes in the compaction performance of tablet formulations containing mannitol.
Methods
The mechanical properties (Poisson’s ratio, fracture energy) and mechanical performance (ejection force, pressure transmission ratio, residual radial die-wall stress, and tensile strength) of mannitol compacts were assessed on a compaction simulator for four lots of mannitol from two different vendors. The variation of material attributes of each lot, including particle size distribution (PSD), crystal form, primary crystal size and morphology, specific surface area (SSA), powder flow, and moisture absorption were investigated.
Results
The variability of material attributes in mannitol lots, especially primary crystal size and SSA, can result in significant changes in mechanical properties and mechanical performance such as ejection force and residual radial die-wall stresses, which potentially led to chipping during compression.
Conclusion
The study elucidated the linkage between fundamental material attributes and mechanical properties of mannitol, highlighting their impact on tablet defects and compaction performance in compression. A comprehensive understanding of the variability in mannitol properties between vendors and lots is crucial for successful formulation development, particularly when high percentages of mannitol are included as a brittle excipient.









Similar content being viewed by others
Notes
Details of the Flodex measurement can be found from SUPPLEMENTARY INFORMATION-Experimental section.
* Details of the DVS measurement can be found from SUPPLEMENTARY INFORMATION-Experimental section.
References
Paul S, Chang SY, Dun J, Sun WJ, Wang K, Tajarobi P, et al. Comparative analyses of flow and compaction properties of diverse mannitol and lactose grades. Int J Pharm. 2018;546(1–2):39–49. https://doi.org/10.1016/j.ijpharm.2018.04.058.
Khorsheed B, Gabbott I, Reynolds GK, Taylor SC, Roberts RJ, Salman AD. Twin-screw granulation: Understanding the mechanical properties from powder to tablets. Powder Technol. 2019;341:12. https://doi.org/10.1016/j.powtec.2018.05.013.
Zarbock S, Magnuson B, Hoskins L. Lactose: the hidden culprit in medication intolerance? Orthopedics. 2007;30(8):7. https://doi.org/10.3928/01477447-20070801-10.
Bharate SS, Bharateb SB, Bajajc AN. Incompatibilities of pharmaceutical excipients with active pharmaceutical ingredients: a comprehensive review. J Excipients Food Chem. 2010;1(3):24.
Palugan L, Moutaharrik S, Cirilli M, Gelainb A, Maroni A, Melocchi A, Zema L, Foppoli A, Cerea M. Evaluation of different types of mannitol for dry granulation by roller compaction. J Drug Delivery Sci Technol. 2022;75:103619–21. https://doi.org/10.1016/j.jddst.2022.103619.
Boetzel R, Schlingemann J, Hickert S, Korn C, Kocks G, Luck B, et al. A nitrite excipient database: a useful tool to support N-Nitrosamine risk assessments for drug products. J Pharm Sci. 2022;112(6):1615–24. https://doi.org/10.1016/j.xphs.2022.04.016.
Moser J, Ashworth IW, Harris L, Hillier MC, Nanda KK, Scrivens G. N-Nitrosamine formation in pharmaceutical solid drug products: experimental observations. J Pharm Sci. 2023;112(5):1255–67. https://doi.org/10.1016/j.xphs.2023.01.027.
Rana AHKS. Manufacturing defects of tablets -a review. J Drug Deliv Ther. 2013;3(6):7. https://doi.org/10.22270/jddt.v3i6.722.
Yu D, Nie H. Evaluation of Alternative Metallic Stearates as Lubricants in Pharmaceutical Tablet Formulation. AAPS PharmSciTech. 2022;23(6):200. https://doi.org/10.1208/s12249-022-02338-1.
Tanabeab S, Nakagawa H, Watanabe T, Minamia H, Ando S, Urbanetz NA, Scherließ R. Selection of a round convex tablet shape that mitigates the risk of chipping and capping based on systematic evaluation by utilizing multivariate analysis. Eur J Pharm Sci. 2018;120:10. https://doi.org/10.1016/j.ejps.2018.05.008.
Nie H, Klinzing G, Xu W. A comparative study of applying backscattering and transmission Raman spectroscopy to quantify solid-state form conversion in pharmaceutical tablets. Int J Pharm. 2022;617:121608. https://doi.org/10.1016/j.ijpharm.2022.121608.
Park H, Nie H, Dhiman A, Tomar V, Zhou QT. Understanding Dynamics of Polymorphic Conversion during the Tableting Process Using In Situ Mechanical Raman Spectroscopy. Mol Pharm. 2020;17(8):10. https://doi.org/10.1021/acs.molpharmaceut.0c00460.
Paul S, Tajarobi P, Boissier C, Sun CC. Tableting performance of various mannitol and lactose grades assessed by compaction simulation and chemometrical analysis. Int J Pharm. 2019;566:24–31. https://doi.org/10.1016/j.ijpharm.2019.05.030.
Wagner CM, Pein M, Breitkreutz J. Roll compaction of mannitol: compactability study of crystalline and spray-dried grades. Int J Pharm. 2013;453(2):416–22. https://doi.org/10.1016/j.ijpharm.2013.05.024.
Mareczek L, Riehl C, Harms M, Reichl S. Understanding the Multidimensional Effects of Polymorphism, Particle Size and Processing for D-Mannitol Powders. Pharmaceutics. 2022;14(10). https://doi.org/10.3390/pharmaceutics14102128.
Yoshinari T, Forbes RT, York P, Kawashima Y. The improved compaction properties of mannitol after a moisture-indued polymorphic transition. Int J Pharm. 2003;258:121–31. https://doi.org/10.1016/S0378-5173(03)00157-1.
Vanhoorne V, Almey R, De Beer T, Vervaet C. Delta-mannitol to enable continuous twin-screw granulation of a highly dosed, poorly compactable formulation. Int J Pharm. 2020;583:119374–10. https://doi.org/10.1016/j.ijpharm.2020.119374.
Nie HXW, Ren J, Taylor L, Marsac PJ, John CT, Byrn SR. Impact of Metallic Stearates on Disproportionation of Hydrochloride Salts of Weak Bases in Solid-State Formulations. Mol Pharm. 2016;13(10):12. https://doi.org/10.1021/acs.molpharmaceut.6b00630.
Nie H, Liu Z, Marks BC, Taylor LS, Byrn SR, Marsac PJ. Analytical approaches to investigate salt disproportionation in tablet matrices by Raman spectroscopy and Raman mapping. J Pharm Biomed Anal. 2016;118:328–37. https://doi.org/10.1016/j.jpba.2015.10.024.
Procopio AT, Zavaliangos A, Cunningham JC. Analysis of the diametrical compression test and the applicability to plastically deforming materials. J Mater Sci. 2003;38:3629–39. https://doi.org/10.1023/A:1025681432260.
Cunningham JC, Sinka IC, Zavaliangos A. Analysis of tablet compaction. I. Characterization of mechanical behavior of powder and powder/tooling friction. J Pharm Sci. 2004;93(8):18. https://doi.org/10.1002/jps.20110.
Uzondu B, Leung LY, Mao C, Yang C-Y. A mechanistic study on tablet ejection force and its sensitivity to lubrication for pharmaceutical powders. Int J Pharm. 2018;543(1–2):11.
Koner JS, Rajabi-Siahboomi A, Bowen J, Perrie Y, Kirby D, Mohammeda AR. A Holistic Multi Evidence Approach to Study the Fragmentation Behaviour of Crystalline Mannitol. Sci Rep. 2015;5. https://doi.org/10.1038/srep16352.
Meynard J, Amado-Becker F, Tchoreloff P, Mazel V. On the complexity of predicting tablet capping. Int J Pharm. 2022;623:121949. https://doi.org/10.1016/j.ijpharm.2022.121949.
Koerner M. Triaxial compaction of metal powder. Powder Metall Int. 1971;3(4):3.
Garner S, Ruiz E, Strong J, Zavaliangos A. Mechanisms of crack formation in die compacted powders during unloading and ejection: An experimental and modeling comparison between standard straight and tapered dies. Powder Technol. 2014;264:114–27. https://doi.org/10.1016/j.powtec.2014.04.086.
Cabiscol R, Shi H, Wünsch I, Magnanimo V, Finke JH, Luding S, et al. Effect of particle size on powder compaction and tablet strength using limestone. Adv Powder Technol. 2020;31(3):1280–9. https://doi.org/10.1016/j.apt.2019.12.033.
Fan A, Pallerla S, Carlson G, Ladipo D, Dukich J, Capella R, et al. Effect of particle size distribution and flow property of powder blend on tablet weight variation. Am Pharm Rev. 2005;8:73–8.
Kudo Y, Yasuda M, Matsusaka S. Effect of particle size distribution on flowability of granulated lactose. Adv Powder Technol. 2020;31(1):121–7. https://doi.org/10.1016/j.apt.2019.10.004.
Xiao P, Guo Y, Wang J, Liu H, Zhang J, Wang Y, et al. The effect of granules characters on mechanical properties of press-coated tablets: A comparative study. Int J Pharm. 2022;624:121986. https://doi.org/10.1016/j.ijpharm.2022.121986.
Charoo NA. Critical Excipient Attributes Relevant to Solid Dosage Formulation Manufacturing. J Pharm Innov. 2020;15(1):163–81. https://doi.org/10.1007/s12247-019-09372-w.
Penha F, Gopalan A, Meijlink J, Ibis-Ozdemir F, Eral H. Selective crystallization of D-Mannitol polymorphs using surfactant self-assembly. Crystal Growth Des. 2021;21:3928–35. https://doi.org/10.1021/acs.cgd.1c00243.
Yang Y, Liu J, Hu A, Nie T, Cheng Z, Liu W. A critical review on engineering of D-Mannitol crystals: properties, applications, and polymorphic control. Crystals. 2022;12(8). https://doi.org/10.3390/cryst12081080.
Thakral S, Sonje J, Munjal B, Bhatnagar B, Suryanarayanan R. Mannitol as an Excipient for Lyophilized Injectable Formulations. J Pharm Sci. 2023;112(1):19–35. https://doi.org/10.1016/j.xphs.2022.08.029.
Cares-Pacheco MG, Vaca-Medina G, Calvet R, Espitalier F, Letourneau JJ, Rouilly A, et al. Physicochemical characterization of d-mannitol polymorphs: The challenging surface energy determination by inverse gas chromatography in the infinite dilution region. Int J Pharm. 2014;475(1):69–81. https://doi.org/10.1016/j.ijpharm.2014.08.029.
Yoshinari T, Forbes RT, York P, Kawashima Y. The improved compaction properties of mannitol after a moisture-induced polymorphic transition. Int J Pharm. 2003;258(1–2):11. https://doi.org/10.1016/S0378-5173(03)00157-1.
Xie Y, Cao W, Krishnan S, Lin H, Cauchon N. Characterization of Mannitol Polymorphic Forms in Lyophilized Protein Formulations Using a Multivariate Curve Resolution (MCR)-Based Raman Spectroscopic Method. Pharm Res. 2008;25(10):2292–301. https://doi.org/10.1007/s11095-008-9624-1.
Sun CC. Mechanical properties. In: Li T, Mattei A, editors. Pharmaceutical crystals: science and engineering. John Wiley & Sons, Inc.; 2018. p. 273–96.
Wünsch I, Finke JH, John E, Juhnke M, Kwade A. The influence of particle size on the application of compression and compaction models for tableting. Int J Pharm. 2021;599:120424. https://doi.org/10.1016/j.ijpharm.2021.120424.
Kosugi A, Leong KH, Urata E, Hayashi Y, Kumada S, Okada K, et al. Effect of Different Direct Compaction Grades of Mannitol on the Storage Stability of Tablet Properties Investigated Using a Kohonen Self-Organizing Map and Elastic Net Regression Model. Pharmaceutics. 2020;12(9). https://doi.org/10.3390/pharmaceutics12090886
Acknowledgements
We acknowledge Dr. Sebastian Escotet for his contribution in conducting the mannitol PSD adjustment experiments and Dr. Meng Li for helpful scientific discussions. The authors would also like to acknowledge Fan Zhang-Plasket for her contribution in conducting several material characterization experiments and data collection.
Author information
Authors and Affiliations
Contributions
Haichen Nie: Conceptualization, conducting experiment, data collection and analysis, manuscript drafting, manuscript review & editing; Jiaying Liu: conducting experiment, interpretation of data, manuscript drafting, review and editing; Gerard R. Klinzing, conducting experiment, interpretation of data, manuscript review and editing.
Corresponding author
Ethics declarations
Conflict of Interest
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, J., Klinzing, G.R. & Nie, H. Effect of Material Properties and Variability of Mannitol on Tablet Formulation Development. Pharm Res 40, 2071–2085 (2023). https://doi.org/10.1007/s11095-023-03577-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-023-03577-y