Abstract
Objectives
Results from previous ex-vivo continuous renal replacement therapy (CRRT) models have successfully demonstrated similar extraction coefficients (EC) identified from in-vivo clinical trials. The objectives of this study are to develop an ex-vivo in-vivo correlation (EVIVC) model to predict drug clearance for commonly used antiepileptics and to evaluate similarity in drug extraction across different CRRT modalities to extrapolate dosing recommendations.
Methods
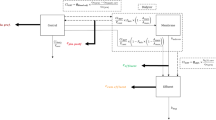
Levetiracetam, lacosamide, and phenytoin CRRT clearance was evaluated using the Prismaflex CRRT system and M150 hemodiafilters using an albumin containing normal saline (ALB-NS) vehicle with 3 different albumin concentrations (2 g/dL, 3 g/dL, and 4 g/dL) and a human plasma vehicle at 3 different effluent flow rates (1 L/hr, 2 L/hr, and 3 L/hr). Blood and effluent/dialysate concentrations were collected after circuit priming. ECs were calculated for each drug, modality, vehicle, and experimental arm combination.
Results
The calculated average EC for levetiracetam and lacosamide was approximated to the fraction unbound from plasma protein. Human plasma and ALB-NS vehicles demonstrated adequate prediction of in-vivo CRRT clearance. Geometric mean ratios indicated similarity in extraction coefficients when comparing between hemofiltration and hemodiafiltration modalities and between filtration and dialysis modalities at effluent flow rates ≤ 2L/hr. Evaluation of phenytoin provided inconsistent findings with regards to extraction coefficient similarity across different CRRT modalities.
Conclusion
The findings indicate that an ex-vivo study can be used as a surrogate to predict in-vivo levetiracetam and lacosamide clearance in patients receiving CRRT.





Similar content being viewed by others
Data Availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
References
Tolwani A. Continuous renal-replacement therapy for acute kidney injury. N Engl J Med. 2012;367:2505–14.
Tandukar S, Palevsky PM. Continuous renal replacement therapy: who, when, why, and how. Chest. 2019;155(3):626–38.
Schefold JC, Haehling SV, Pschowski R, Bender TO, Berkmann C, Briegel S, et al. The effect of continuous versus intermittent renal replacement therapy on the outcome of critically ill patients with acute renal failure (CONVINT): a prospective randomized controlled trial. Crit Care. 2014;18:R11.
Vinsonneau C, Camus C, Combes A, Beauregard MA, Klouche K, Boulain T, et al. Continuous venovenous haemodiafiltration versus intermittent haemodialysis for acute renal failure patients with multiorgan dysfunction syndrome: a mulicentre randomized trial. Lancet. 2006;368(9533):379–85.
Mehta RL, Mcdonald B, Gabbai FB, Pahl M, Pascual M, Farkas A, et al. A randomized clinical trial of continuous versus intermittent dialysis for acute renal failure. Kidney Int. 2001;60:1154–63.
Augustine JJ, Sandy D, Seifert TH, Paganini EP. A randomized controlled trial comparing intermittent with continuous dialysis in patients with ARF. Am J Kid Dis. 2004;44:1000–7.
Goldstein SL, Nolin TD. Lack of drug dosing guidelines for critically ill patients receiving continuous renal replacement therapy. Clin Pharamcol Ther. 2014;96(2):159–61.
Nolin T, Aronoff GR, Fissell WH, Jain L, Madabushi R, Reynold K, et al. Pharmacokinetic assessment in patients receiving Continuous RRT: perspectives from the kidney health initiative. Clin J Am Soc Nephrol. 2015;10:159–64.
Churchwell MD. Use of an in-vitro model of renal replacement therapy systems to estimate extracorporeal drug removal. J Clin Pharmacol. 2012;52:35S-44S.
Kalaria SN, Armahizer M, McCarthy P, Badjatia N, Gobburu JV, Gopalakrishnan M. A practice-based, clinical pharmacokinetic study to inform levetiracetam dosing in critically ill patients undergoing continuous venovenous hemofiltration (PADRE-01). Clin Trans Sci. 2020;13(5):950–9.
Churchwell MD. Use of an in-vitro model of renal replacement therapy systems to estimate extracorporeal drug removal. J Clin Pharmacol. 2012;52:35S-44S.
Purohit PK, Elkomy MH, Frymoyer A, Sutherland SM, Drover D, Hammer GB, et al. Antimicrobial disposition during pediatric continuous renal replacement therapy using an ex-vivo model. Crit Care Med. 2019;47(9):e767–73.
Pasko DA, Chruchwell MD, Salama NN, Mueller BA. Longitudinal hemodiafilter performance in modeled continuous renal replacement therapy. Blood Purif. 2011;32(2):82–8.
Parakininkas D, Greenbaum LA. Comparison of solute clearance in three modes of continuous renal replacement therapy. Pediatr Crit Care Med. 2004;5(3):269–74.
Isla A, Gascon AR, Maynar J, Arzuagga A, Toral D, Pedraz JL. Cefepime and continuous renal replacement therapy (CRRT): in vitro permeability of two CRRT membranes and pharmacokinetics in four critically ill patients. Clin Ther. 2005;27(5):599–608.
Lewis SJ, Switaj LA, Mueller BA. Tedizolid adsorption and transmembrane clearance during in-vitro continuous renal replacement therapy. Blood Purif. 2015;40(1):66–71.
Biagi M, Butler D, Tan X, Qasimeh S, Tejani K, Patel S, et al. Pharmackinetics and dialytic clearance of isavuconazole during in vitro and in vivo continuous renal replacement therapy. Antrimicrob Agents Chemother. 2019. https://doi.org/10.1128/AAC0.1085-19.
Kesner JM, Yardman-Frank JM, Mercier RC, Wong CS, Walker SE, Argyres DP, et al. Trimethoprim and sulfamethoxazole transmembrane clearance during modeled continuous renal replacement therapy. Blood Purif. 2014;38(3–4):195–202.
Shen J, Burgess DJ. In vitro-In vivo correlation for complex non-oral drug products: where do we stand? J Control Release. 2015;219:644–51.
Kalaria SN, Armahizer M, McCarthy P, Badjatia N, Gobburu JV, Gopalakrishnan M. A prospective, real-world, clinical pharmacokinetic study to inform lacosamide dosing in critically ill patients undergoing continuous venovenous haemofiltration (PADRE-02). Br J Clin Pharmacol. 2021;87(11):4375–85.
Kalaria SN, Dahmane E, Armahizer M, McCarthy P, Gopalakrishnan M. Development and validation of a HPLC-UV assay for quantification of levetiracetam concentrations in critically ill patients undergoing continuous renal replacement therapy. Biomed Chromatogr. 2018;32:4257–65.
Hulko M, Haug Ulrike, Gauss J, Boschetti-de-Fierro A, Beck W, and Krause B. Requirements and pitfalls of dialyzer sieving coefficients comparisons. Artif Organs. 2018;42(12):1164–73.
Oltrogge KM, Peppard WJ, Saleh M, Regner KR, Herrmann DJ. Phenytoin removal by continuous venovenous hemomfitlration. Ann Pharmacother. 2013;47(9):1218–22.
Chappell K, Kimmons LA, Haller JT, Canada RB, He H, Hudson JQ. Levetiracetam pharmacokinetics in critically ill patients undergoing renal replacement therapy. Crit Care. 2021;61:216–20.
Sigler MH, Teehan BP. Solute transport in continuous hemodialysis: a new treatment for acute renal failure. Kidney Int. 1987;32:562–71.
Relton S, Greenberg A, Palevsky P. Dialysate and blood flow dependence of diffusive solute clearance during CVVHD. ASAIO J. 1992;38:691–6.
Bonnardeaux A, Richette V, Ouimet D, Geadah D, Habel F, Cardinal J. Solute clearance with high dialysate flow rates and glucose absorption from the dialysate in continuous arteriovenous hemodialysis. Am J Kidney Dis. 1992;19:31–8.
Brunet S, Leblanc M, Geadah M, Parent D, Courteau S, and Cardinal J. Diffusive and convective solute clearance during continuous renal replacement therapy at various dialysate and ultrafiltration flow rates. Am J Kidney Dis. 1999l34:486–92.
Nicholson JP, Wolmarans MR, Park GR. The role of albumin in critical illness. Br J Anaesth. 2000;85(4):599–610.
Smith Sa and Waters NJ. Pharmacokinetic and pharmacodynamic considerations for drugs binding to alpha-1-acid glycoprotein. Pharm Res. 2018;36(2):30:1–19.
Churchwell MD, Pasko DA, Mueller BA. Daptomycin clearance during modeled continuous renal replacement therapy. Blood Purif. 2006;24:548–54.
Lewis SJ, Switaj LA, Mueller BA. Tedizolid adsorption and transmembrane clearance during in vitro continuous renal replacement therapy. Blood Purif. 2015;40:66–71.
Stevenson JM, Patel JH, Churchwell MD, VIlay AM, Depestel DD, Sorgel F, et al. Ertapenem clearance during modeled continuous renal replacement therapy. Int J Artif Organs. 2008;31(12):1027–34.
Jang SM, Hough G, Mueller BA. Ex vivo rezafungin adsorption and clearance during continuous renal replacement therapy. Blood Purif. 2018;46:214–9.
US Department of Health and Human Services Food and Drug Administration, & Center for Drug Evaluation and Research. Guidance for Industry Statistical approaches to establishing bioequivalence. 2010. Retrieved from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/statistical-approaches-establishing-bioequivalence.
Farrar JE, Mueller SW, Stevens V, Kiser TH, Taleb S, Reynolds PM. Correlation of antimicrobial fraction unbound and sieving coefficient in critically ill patients on continuous renal replacement therapy: a systematic review. J Antimicrob Chemother. 2022;77:310–9.
Gopler TA, Marx MA. Drug dosing adjustments during continuous renal replacement therapies. Kidney Int. 1998;66:S165–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
All authors declared no competing interests for this work.
Primary Investigator
The authors confirm that the PI for this paper is Dr. Mathangi Gopalakrishnan.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kalaria, S.N., Armahizer, M., McCarthy, P. et al. Development and Use of an Ex-Vivo In-Vivo Correlation to Predict Antiepileptic Drug Clearance in Patients Undergoing Continuous Renal Replacement Therapy. Pharm Res 39, 827–836 (2022). https://doi.org/10.1007/s11095-022-03287-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-022-03287-x