Abstract
Purpose
The aim of this work was to evaluate the use of short durations of externally applied heat with chemical penetration enhancers to improve delivery of isotretinoin to the skin and in particular via the follicular route.
Methods
A range of chemical penetration enhancers were screened for their ability to improve isotretinoin delivery into human skin with heat using infinite dose, Franz cell experiments conducted in a water bath at a higher temperature to simulate heated conditions. Following this a prototype external heating system was developed that provided short durations of heat and its ability to improve delivery of finite doses into the skin and hair follicles was assessed.
Results
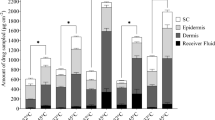
The magnitude of the effect of heat on drug delivery was influenced by the choice of vehicle with changes in isotretinoin flux across skin ranging from not statistically significant to 25 fold increases with heat in the infinite dose studies. The prototype heating system provided significant increases in the total delivery of isotretinoin into the skin from an optimised vehicle. Drug distribution in the skin revealed significant increases in isotretinoin delivery to the hair follicles, and deeper skin layers, but not to the stratum corneum, providing strong evidence that the enhancement in delivery occurred mainly via the hair follicles.
Conclusion
These data indicate that the use of short durations of heat combined with chemical penetration enhancers offers a valuable strategy for improving the delivery of drugs such as isotretinoin to the skin via the hair follicles.

Schematic illustration of the sodium thiosulphate heating system on a Franz diffusion cell and the subsequent impact of a short burst of heat on the delivery of isotretinoin into human skin.






Similar content being viewed by others
References
Jensen BK, McGann LA, Kachevsky V, Franz TJ. The negligible systemic availability of retinoids with multiple and excessive topical application of isotretinoin 0.05% gel (Isotrex) in patients with acne vulgaris. J Am Acad Dermatol. 1991;24:425–8.
Oliveira G, Leverett JC, Emamzadeh M, Lane ME. The effects of heat on skin barrier function and in vivo dermal absorption. Int J Pharm. 2014;464:145–51.
Pattnaik S, Swain K, Rao JV, Varun T, Mallick S. Temperature influencing permeation pattern of alfuzosin: an investigation using DoE. Medicina. 2015;51:253–61.
Wood DG, Brown MB, Jones SA. Understanding heat facilitated drug transport across human epidermis. Eur J Pharm Biopharm. 2012;81:642–9.
Yun J, Lee DH, Im JS, Kim H-I. Improvement in transdermal drug delivery performance by graphite oxide/temperature-responsive hydrogel composites with micro heater. Mater Sci Eng C Mater Biol Appl. 2012;32:1564–70.
Prodduturi S, Sadrieh N, Wokovich AM, Doub WH, Westenberger BJ, Buhse L. Transdermal delivery of fentanyl from matrix and reservoir systems: effect of heat and compromised skin. J Pharm Sci. 2010;99:2357–66.
Akomeah F, Nazir T, Martin GP, Brown MB. Effect of heat on the percutaneous absorption and skin retention of three model penetrants. Eur J Pharm Sci. 2004;21:337–45.
Klemsdal T, Gjesdal K, Bredesen J-E. Heating and cooling of the nitroglycerin patch application area modify the plasma level of nitroglycerin. Eur J Clin Pharmacol. 1992;43:625–8.
Burton JL. The physical properties of sebum in acne vulgaris. Clin Sci. 1970;39:757–67.
Saar BG, Contreras-Rojas LR, Xie XS, Guy RH. Imaging drug delivery to skin with stimulated Raman scattering microscopy. Mol Pharm. 2011;8:969–75.
Caserta F, Brown MB, McAuley WJ. The use of heat and chemical penetration enhancers to increase the follicular delivery of erythromycin to the skin. Eur J Pharm Sci. 2019;132:55–62.
Zhang Q, Murawsky M, LaCount T, Hao J, Kasting GB, Newman B, et al. Characterization of temperature profiles in skin and transdermal delivery system when exposed to temperature gradients in vivo and in vitro. Pharm Res. 2017;34:1491–504.
OECD, Guidance document for the conduct of skin absorption studies, in, 2004.
Moser K, Kriwet K, Naik A, Kalia YN, Guy RH. Passive skin penetration enhancement and its quantification in vitro. Eur J Pharm Biopharm. 2001;52:103–12.
Higuchi T. Physical chemical analysis of percutaneous absorption process from creams and ointments. J Soc Cosmet Chem. 1960;11:70–82.
Crank J. The diffusion equations. In: Crank J, editor. The mathematics of diffusion. Oxford: Clarendon Press; 1975. p. 1–10.
Cornwell P, Barry B. Sesquiterpene components of volatile oils as skin penetration enhancers for the hydrophilic permeant 5-fluorouracil. J Pharm Pharmacol. 1994;46:261–9.
Otberg N, Patzelt A, Rasulev U, Hagemeister T, Linscheid M, Sinkgraven R, et al. The role of hair follicles in the percutaneous absorption of caffeine. Br J Clin Pharmacol. 2008;65:488–92.
Liu X, Grice JE, Lademann J, Otberg N, Trauer S, Patzelt A, et al. Hair follicles contribute significantly to penetration through human skin only at times soon after application as a solvent deposited solid in man. Br J Clin Pharmacol. 2011;72:768–74.
Lehman PA, Slattery JT, Franz TJ. Percutaneous absorption of retinoids: influence of vehicle, light exposure, and dose. J Invest Dermatol. 1988;91:56–61.
McAuley WJ, Caserta F. Film-forming and heated systems. In: Donnelly RF, Singh TRR, editors. Novel delivery Systems for Transdermal and Intradermal Drug Delivery. Chichester: John Wiley & Sons, ltd; 2015. p. 97–124.
M.B. Brown, A.C. Williams, Chemical modulation of Topical & Transdermal permeation, in: the art and science of dermal formulation development, CRC Press, 2019, pp. 45–73.
Eichner A, Stahlberg S, Sonnenberger S, Lange S, Dobner B, Ostermann A, et al. Influence of the penetration enhancer isopropyl myristate on stratum corneum lipid model membranes revealed by neutron diffraction and 2H NMR experiments. Biochim Biophys Acta. 2017;1859:745–55.
Twist JN, Zatz JL. Membrane–solvent–solute interaction in a model permeation system. J Pharm Sci. 1988;77:536–40.
Park J-H, Lee J-W, Kim Y-C, Prausnitz MR. The effect of heat on skin permeability. Int J Pharm. 2008;359:94–103.
McAuley WJ, Oliveira G, Mohammed D, Beezer AE, Hadgraft J, Lane ME. Thermodynamic considerations of solvent/enhancer uptake into a model membrane. Int J Pharm. 2010;396:134–9.
Williams AC. Transdermal and topical drug delivery from theory to clinical practice. London: Pharmaceutical Press; 2003.
Moritz AR, Henriques FC. Studies of thermal injury: II. The relative importance of time and surface temperature in the causation of cutaneous burns. Am J Pathol. 1947;23:695–720.
Lawrence J, Bull J. Thermal conditions which cause skin burns. Eng Med. 1976;5:61–3.
Wood D, Brown M, Jones S, Murnane D. Characterization of latent heat-releasing phase change materials for dermal therapies. J Phys Chem. 2011;115:8369–75.
Teodorescu F, Queniat G, Foulon C, Lecoeur M, Barras A, Boulahneche S, et al. Transdermal skin patch based on reduced graphene oxide: a new approach for photothermal triggered permeation of ondansetron across porcine skin. J Control Release. 2017;245:137–46.
Motwani MR, Rhein LD, Zatz JL. Influence of vehicles on the phase transitions of model sebum. J Cosmet Sci. 2002;53:35–42.
Motwani MR, Rhein LD, Zatz JL. Deposition of salicylic acid into hamster sebaceous glands. J Cosmet Sci. 2004;55:519–31.
Butcher EO, Coonin A. The physical properties of human sebum. J Invest Dermatol. 1949;12:249–54.
Patzelt A, Richter H, Buettemeyer R, Huber HJR, Blume-Peytavi U, Sterry W, et al. Differential stripping demonstrates a significant reduction of the hair follicle reservoir in vitro compared to in vivo. Eur J Pharm Biopharm. 2008;70:234–8.
Szabo G. The regional anatomy of the human integument with special reference to the distribution of hair follicles, sweat glands and melanocytes. Philos Trans R Soc Lond Ser B Biol Sci. 1967;252:447–85.
ACKNOWLEDGMENTS AND DISCLOSURES
The authors would like to thank MedPharm Ltd. (UK) for funding this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Farah, H.A., Brown, M.B. & McAuley, W.J. Heat Enhanced Follicular Delivery of Isotretinoin to the Skin. Pharm Res 36, 124 (2019). https://doi.org/10.1007/s11095-019-2659-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11095-019-2659-7