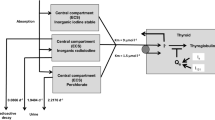
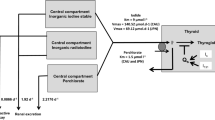
Abstract
Purpose
To propose a new and effective dose regimen for stable potassium iodide (KI) repeated prophylaxis in case of prolonged exposure to radioactive iodine.
Methods
The pharmacokinetics of iodine was determined in rats by compartmental analyses after intravenous and oral administrations of the optimal dose of 1 mg/kg KI, which was previously selected in a dose-effect study. The thyroid protection against iodine-125 incorporation was followed during 24 h after a single oral dosing of KI. A repeated KI prophylaxis was modeled using initial estimates of iodine pharmacokinetic parameters.
Results
A dose regimen consisting in administrations of 1 mg/kg daily for 8 days was selected and studied. Plasma iodine concentrations predicted by simulation were verified by experimental data and varied after the third dose of KI between 174 and 1190 μg/l. The inhibition study of iodine-125 binding in the thyroid as a function of the time showed that the protection effect of KI could be correlated to stable iodine plasma concentrations. Hence, a theoretical decrease in iodine-125 thyroid uptake from 63 to 88% could be achieved in a 24 h-interval between two KI doses.
Conclusion
Given the satisfactory levels of thyroid protection, this dose regimen could be envisaged in order to extent KI indications for repeated prophylaxis.






Similar content being viewed by others
References
Mahoney MC, Lawvere S, Falkner KL, Averkin YI, Ostapenko VA, Michalek AM, et al. Thyroid cancer incidence trends in Belarus: examining the impact of Chernobyl. Int J Epidemiol. 2004;33:1025–33.
Shakhtarin VV, Tsyb AF, Stepanenko VF, Orlov MY, Kopecky KJ, Davis S. Iodine deficiency, radiation dose, and the risk of thyroid cancer among children and adolescents in the Bryansk region of Russia following the Chernobyl power station accident. Int J Epidemiol. 2003;32:584–91.
Verger P, Aurengo A, Geoffroy B, Le Guen B. Iodine kinetics and effectiveness of stable iodine prophylaxis after intake of radioactive iodine: a review. Thyroid. 2001;11:353–60.
Reiners C, Schneider R. Potassium iodide (KI) to block the thyroid from exposure to I-131: current questions and answers to be discussed. Radiat Environ Biophys. 2013;52:189–93.
World Health Organization. Iodine thyroid blocking: guidelines for use in planning for and responding to radiological and nuclear emergencies. Geneva; 2017, p. 46.
Saxena KM, Chapman EM, Pryles CV. Minimal dosage of iodide required to suppress uptake of iodine-131 by normal thyroid. Science. 1962;138:431–3.
Cuddihy RG. Thyroidal iodine-131 uptake, turnover and blocking in adults and adolescents. Health Phys. 1966;12:1021–5.
Kunii Y, Uruno T, Mukasa K, Sekiya K, Iwaku K, Suzuki A, et al. Inhibitory effect of low-dose inorganic iodine on thyroidal radioactive iodine uptake in healthy Japanese adults. Endocr J. 2016;63:21–7.
Sternthal E, Lipworth L, Stanley B. Suppression of thyroid radioiodine uptake by various doses of stable iodide. N Engl J Med. 1980;303:1083–8.
Blum M, Eisenbud M. Reduction of thyroid irradiation from 131-I by potassium iodide. J Am Med Assoc. 1967;200:1036–40.
Phan G, Rebiere F, Suhard D, Legrand A, Carpentier F, Sontag T, et al. Optimal KI prophylactic dose determination for thyroid radiation protection after a single administration in adult rats. Dose-Response. 2017;15:1–8.
Landon G, Bouvier-Capely C, Legrand A, Sontag T, Finance G, Saint-Hilaire M, et al. Validation and comparison of two calibration methods for themeasurement of stable iodine in the urinary matrix by ICP-MS: standard addition vs external calibration. Am J Anal Chem. 2017;8:245–63.
Gabrielsson J, Meibohm B, Weiner D. Pattern recognition in pharmacokinetic data analysis. AAPS J. 2016;18:47–63.
Gabrielsson J, Weiner D. Pharmacokinetics and pharmacodynamic data analysis: concepts and applications. Stockholm: Swedish Academy of Pharmaceutical Sciences; 2016.
Akaike H. A new look at the statistical model identification. IEEE Trans Autom Control. 1974;19:716–23.
Gabrielsson J, Weiner D. Akaike and Schwarz criteria. Pharmacokinetics and pharmacodynamic data analysis: concepts and applications. Stockholm: Swedish Academy of Pharmaceutical Sciences; 2016. p. 389.
Schwarz G. Estimating the dimension of a model. Ann Stat. 1978;6:461–4.
Abrahamyan HG, Hovhannisyan AS. Comparative pharmacokinetics of iodide anion at administration of iodine preparations. New Armenian Medical J. 2011;5:26–32.
Berkovski V. New iodine models family for simulation of short-term biokinetics processes, pregnancy and lactation. Food Nutr Bull. 2002;23:87–94.
McLanahan ED, Andersen ME, Fisher JW. A biologically based dose-response model for dietary iodide and the hypothalamic-pituitary-thyroid axis in the adult rat: evaluation of iodide deficiency. Toxicol Sci. 2008;102:241–53.
Jang M, Kim HK, Choi CW, Kang CS. Age-dependent potassium iodide effect on the thyroid irradiation by 131I and 133I in the nuclear emergency. Radiat Prot Dosim. 2008;130:499–502.
Jang M, Kim HK, Choi CW, Kang CS. Thyroid dose estimation with potassium iodide (KI) administration in a nuclear emergency. Radiat Prot Dosim. 2008;132:303–7.
Koutras DA, Pharmakiotis AD, Koliopoulos N, Tsoukalos J, Souvatzoglou A, Sfontouris J. The plasma inorganic iodine and the pituitary-thyroid axis in pregnancy. J Endocrinol Investig. 1978;1:227–31.
Nagataki S, Shizume K, Nakao K. Effect of iodide on thyroidal iodine turnover in hyperthyroid subjects. J Clin Endocrinol Metab. 1970;30:469–78.
Liberman CS, Pino SC, Fang SL, Braverman LE, Emerson CH. Circulating iodide concentrations during and after Pregnancy1. J Clin Endocrinol Metab. 1998;83:3545–9.
Paul T, Meyers B, Witorsch RJ, Pino S, Chipkin S, Ingbar SH, et al. The effect of small increases in dietary iodine on thyroid function in euthyroid subjects. Metabolism. 1988;37:121–4.
Abraham GE. Serum inorganic iodide levels following ingestion of a tablet form of Lugol solution: evidence for an enterohepatic circulation of iodine. The Original Internist. 2004;11:29–34.
Delgado G, Muñoz-Torres C, Orozco-Esquivel T, Anguiano B, Aceves C. Total iodine quantification in fluids and tissues from iodine- or iodide-supplemented rats by ion chromatography following microwave-assisted digestion. Thyroid. 2015;25:352–60.
Gilbert ME, McLanahan ED, Hedge J, Crofton KM, Fisher JW, Valentin-Blasini L, et al. Marginal iodide deficiency and thyroid function: dose-response analysis for quantitative pharmacokinetic modeling. Toxicology. 2011;283:41–8.
Shelor CP, Dasgupta PK. Review of analytical methods for the quantification of iodine in complex matrices. Anal Chim Acta. 2011;702:16–36.
Sun X, Lu L, Shan L, Shan Z, Teng W. Iodine storage and metabolism of mild to moderate iodine-deficient pregnant rats. Thyroid. 2017;27:846–51.
Leggett RW. A physiological systems model for iodine for use in radiation protection. Radiat Res. 2010;174:496–516.
McLanahan ED, Campbell JL Jr, Ferguson DC, Harmon B, Hedge JM, Crofton KM, et al. Low-dose effects of ammonium perchlorate on the hypothalamic-pituitary-thyroid axis of adult male rats pretreated with PCB126. Toxicol Sci. 2007;97:308–17.
Clewell RA, Merrill EA, Yu KO, Mahle DA, Sterner TR, Mattie DR, et al. Predicting fetal perchlorate dose and inhibition of iodide kinetics during gestation: a physiologically-based pharmacokinetic analysis of perchlorate and iodide kinetics in the rat. Toxicol Sci. 2003;73:235–55.
Leggett R. An age-specific biokinetic model for iodine. J Radiol Prot. 2017;37:864–82.
Ramsden D, Passant FH, Peabody CO, Speight RG. Radioiodine uptakes in the thyroid studies of the blocking and subsequent recovery of the gland following the administration of stable iodine. Health Phys. 1967;13:633–46.
Braverman ER, Blum K, Loeffke B, Baker R, Kreuk F, Yang SP, et al. Managing terrorism or accidental nuclear errors, preparing for iodine-131 emergencies: a comprehensive review. Int J Environ Res Public Health. 2014;11:4158–200.
Nagataki S, Ingbar SH. Relation between qualitative and quantitative alterations in thyroid hormone synthesis induced by varying doses of iodide. Endocrinology. 1964;74:731–6.
Lebsir D, Manens L, Grison S, Lestaevel P, Ebrahimian Chiusa T, Suhard D, et al. Effects of repeated potassium iodide administration on genes involved in synthesis and secretion of thyroid hormone in adult male rats. Mol Cell Endocrinol. 2018;474:119–26.
Zanzonico PB, Becker DV. Effects of time of administration and dietary iodine levels on potassium iodide (KI) blockade of thyroid irradiation by 131I from radioactive fallout. Health Phys. 2000;78:660–7.
Acknowledgments and Disclosures
The authors are thankful to the Central Army Pharmacy for providing solutions of KI, Dr. Delphine Denais-Lalieve (DVM), Mélanie Berchet, Amandine Sache, Romain Granger and Frédéric Voyer from the animal experiment support group of IRSN for animal care. These studies were supported by the research program in Nuclear Safety and Radiation Protection of the French National Research Agency (ANR) (contract number ANR-11-RSNR-0019). The authors declared no potential conflicts of interest with respect to research, authorship and publication of this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Phan, G., Chioukh, R., Suhard, D. et al. Repeated KI Prophylaxis in Case of Prolonged Exposure to Iodine Radioisotopes: Pharmacokinetic Studies in Adult Rats. Pharm Res 35, 227 (2018). https://doi.org/10.1007/s11095-018-2515-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11095-018-2515-1