Abstract
Purpose
Collagen-induced arthritic (CIA) rats are used commonly for preclinical pharmacologic research into rheumatoid arthritis (RA). Dexamethasone (DEX), a potent corticosteroid (CS), remains an important component in combination therapy for RA. Although sex differences in RA and CS pharmacokinetics/pharmacodynamics (PK/PD) have been documented in humans, there has been no such comprehensive evaluation of sex differences in CIA rats.
Methods
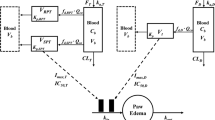
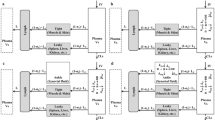
Paw size measurements were obtained for males and females from four groups of animals: healthy controls, non-drug treated arthritic animals, and both 0.225 and 2.25 mg/kg DEX-treated arthritic animals. A turnover model for disease progression, minimal PBPK model for drug concentrations, and inhibitory indirect response model were applied using population PK/PD modeling.
Results
The clearances of DEX were 43% greater in males, but other PK parameters were similar. The temporal profiles of paw swelling exhibited earlier progression, peak edema times, and disease remission in females. DEX suppressed paw edema well in both males and females with similar capacity (Imax) values (=1.0), but DEX potency was less in females with higher IC50 values (0.101 versus 0.015 ng/mL).
Conclusions
The pharmacology of DEX was well characterized in CIA rats. This study addresses knowledge gaps about sex differences and can be a guide for more mechanistic assessment of sex, drug, and disease differences in RA.





Similar content being viewed by others
Abbreviations
- AUC:
-
Area under the curve
- CS:
-
Corticosteroids
- CIA:
-
Collagen-induced arthritis
- DEX:
-
Dexamethasone
- DIS:
-
Disease
- GR:
-
Glucocorticoid receptor
- IIV:
-
Inter-individual variability
- IL:
-
Interleukin
- ISF:
-
Interstitial fluid
- mPBPK:
-
Minimal physiologically-based pharmacokinetic
- OFV:
-
Objective function value
- PD:
-
Pharmacodynamics
- PK:
-
Pharmacokinetics
- RA:
-
Rheumatoid arthritis
- SC:
-
Subcutaneous
References
Scott DL, Wolfe F, Huizinga TW. Rheumatoid arthritis. Lancet. 2010;376(9746):1094–108.
Gaffo A, Saag KG, Curtis JR. Treatment of rheumatoid arthritis. Am J Health Syst Pharm. 2006;63(24):2451–65.
Mateen S, Zafar A, Moin S, Khan AQ, Zubair S. Understanding the role of cytokines in the pathogenesis of rheumatoid arthritis. Clin Chim Acta. 2016;455:161–71.
McInnes IB, Schett G. The pathogenesis of rheumatoid arthritis. N Engl J Med. 2011;365(23):2205–19.
Brown PM, Pratt AG, Isaacs JD. Mechanism of action of methotrexate in rheumatoid arthritis, and the search for biomarkers. Nat Rev Rheumatol. 2016;12(12):731–42.
Baschant U, Lane NE, Tuckermann J. The multiple facets of glucocorticoid action in rheumatoid arthritis. Nat Rev Rheumatol. 2012;8(11):645–55.
Ronchetti S, Migliorati G. Riccardi C. GILZ as a mediator of the anti-inflammatory effects of glucocorticoids. Front Endocrinol (Lausanne). 2015;6:170.
Losel R, Wehling M. Nongenomic actions of steroid hormones. Nat Rev Mol Cell Biol. 2003;4(1):46–56.
Cato AC, Nestl A. Mink S. Rapid actions of steroid receptors in cellular signaling pathways. Sci STKE. 2002;2002(138):re9.
Jawaheer D, Lum RF, Gregersen PK, Criswell LA. Influence of male sex on disease phenotype in familial rheumatoid arthritis. Arthritis Rheum. 2006;54(10):3087–94.
Kovacs WJ, Olsen NJ. Sexual dimorphism of RA manifestations: genes, hormones and behavior. Nat Rev Rheumatol. 2011;7(5):307–10.
van Vollenhoven RF. Sex differences in rheumatoid arthritis: More than meets the eye. BMC Med. 2009;7:12.
Danska JS. Sex matters for mechanism. Sci Transl Med 2014;6(258):258fs240.
Jorgensen TN. Sex disparities in the immune response. Cell Immunol. 2015;294(2):61–2.
Lew KH, Ludwig EA, Milad MA, Donovan K, Middleton E Jr, Ferry JJ, et al. Gender-based effects on methylprednisolone pharmacokinetics and pharmacodynamics. Clin Pharmacol Ther. 1993;54(4):402–14.
Kovats S. Estrogen receptors regulate innate immune cells and signaling pathways. Cell Immunol. 2015;294(2):63–9.
Gubbels Bupp MR. Sex, the aging immune system, and chronic disease. Cell Immunol. 2015;294(2):102–10.
Hegen M, Keith JC Jr, Collins M, Nickerson-Nutter CL. Utility of animal models for identification of potential therapeutics for rheumatoid arthritis. Ann Rheum Dis. 2008;67(11):1505–15.
Bolon B, Stolina M, King C, Middleton S, Gasser J, Zack D, et al. Rodent preclinical models for developing novel antiarthritic molecules: comparative biology and preferred methods for evaluating efficacy. J Biomed Biotechnol. 2011;2011:569068.
Moudgil KD, Kim P, Brahn E. Advances in rheumatoid arthritis animal models. Curr Rheumatol Rep. 2011;13(5):456–63.
Earp JC, DuBois DC, Almon RR, Jusko WJ. Quantitative dynamic models of arthritis progression in the rat. Pharm Res. 2009;26(1):196–203.
Earp JC, DuBois DC, Molano DS, Pyszczynski NA, Almon RR, Jusko WJ. Modeling corticosteroid effects in a rat model of rheumatoid arthritis II: Mechanistic pharmacodynamic model for dexamethasone effects in Lewis rats with collagen-induced arthritis. J Pharmacol Exp Ther. 2008;326(2):546–54.
Earp JC, DuBois DC, Molano DS, Pyszczynski NA, Keller CE, Almon RR, et al. Modeling corticosteroid effects in a rat model of rheumatoid arthritis I: Mechanistic disease progression model for the time course of collagen-induced arthritis in Lewis rats. J Pharmacol Exp Ther. 2008;326(2):532–45.
Stolina M, Bolon B, Dwyer D, Middleton S, Duryea D, Kostenuik PJ, et al. The evolving systemic and local biomarker milieu at different stages of disease progression in rat collagen-induced arthritis. Biomarkers. 2008;13(7):692–712.
Guo W, Yu D, Wang X, Luo C, Chen Y, Lei W, et al. Anti-inflammatory effects of interleukin-23 receptor cytokine-binding homology region rebalance T cell distribution in rodent collagen-induced arthritis. Oncotarget. 2016;7(22):31800–13.
Li X, DuBois DC, Song D, Almon RR, Jusko WJ, Chen X. Modeling combined immunosuppressive and anti-inflammatory effects of dexamethasone and naproxen in rats predicts the steroid-sparing potential of naproxen. Drug Metab Dispos. 2017;45(7):834–45.
Cao Y, Jusko WJ. Applications of minimal physiologically-based pharmacokinetic models. J Pharmacokinet Pharmacodyn. 2012;39(6):711–23.
Samtani MN, Jusko WJ. Comparison of dexamethasone pharmacokinetics in female rats after intravenous and intramuscular administration. Biopharm Drug Dispos. 2005;26(3):85–91.
Earp JC, Pyszczynski NA, Molano DS, Jusko WJ. Pharmacokinetics of dexamethasone in a rat model of rheumatoid arthritis. Biopharm Drug Dispos. 2008;29(6):366–72.
McNamara PJ, Gibaldi M, Stoeckel K. Fraction unbound in interstitial fluid. J Pharm Sci. 1983;72(7):834–6.
Post TM, Freijer JI, DeJongh J, Danhof M. Disease system analysis: basic disease progression models in degenerative disease. Pharm Res. 2005;22(7):1038–49.
Dayneka NL, Garg V, Jusko WJ. Comparison of four basic models of indirect pharmacodynamic responses. J Pharmacokinet Biopharm. 1993;21(4):457–78.
Beal SL, Sheiner LB, Boeckmann AL, Bauer RJ. NONMEM Users Guides, Icon Development Solutions, Ellicott City, MD, 2009.
Keizer RJ, Karlsson MO, Hooker A. Modeling and simulation workbench for NONMEM: Tutorial on Pirana, PsN, and Xpose. CPT Pharmacometrics Syst Pharmacol. 2013;2:e50.
Lindbom L, Pihlgren P, Jonsson EN. PsN-Toolkit--a collection of computer intensive statistical methods for non-linear mixed effect modeling using NONMEM. Comput Methods Prog Biomed. 2005;79(3):241–57.
R-Core Team. R: a language and environment for statistical computing. Vienna: Foundation for Statistical Computing; 2008.
Lon HK, Liu D, DuBois DC, Almon RR, Jusko WJ. Modeling pharmacokinetics/pharmacodynamics of abatacept and disease progression in collagen-induced arthritic rats: a population approach. J Pharmacokinet Pharmacodyn. 2013;40(6):701–12.
Tomlinson ES, Maggs JL, Park BK, Back DJ. Dexamethasone metabolism in vitro: species differences. J Steroid Biochem Mol Biol. 1997;62(4):345–52.
Lamiable D, Vistelle R, Fay R, Bensussan B, Millart H, Wiczewski M, et al. Influence of sex and oestrogen replacement on the disposition of dexamethasone in rats. Fundam Clin Pharmacol. 1991;5(8):733–40.
Kato R, Yamazoe Y. Sex-specific cytochrome P450 as a cause of sex- and species-related differences in drug toxicity. Toxicol Lett. 1992;64–65 Spec No:661–667.
Maggs JL, Morgan P, Park BK. The sexually differentiated metabolism of [6,7-3H]17 beta-oestradiol in rats: male-specific 15 alpha- and male-selective 16 alpha-hydroxylation and female-selective catechol formation. J Steroid Biochem Mol Biol. 1992;42(1):65–76.
Li X, DuBois DC, Almon RR, Jusko WJ. Modeling sex differences in pharmacokinetics, pharmacodynamics, and disease progression effects of naproxen in rats with collagen-induced arthritis. Drug Metab Dispos. 2017;45(5):484–91.
Cutolo M, Villaggio B, Seriolo B, Montagna P, Capellino S, Straub RH, et al. Synovial fluid estrogens in rheumatoid arthritis. Autoimmun Rev. 2004;3(3):193–8.
Shah DK, Betts AM. Towards a platform PBPK model to characterize the plasma and tissue disposition of monoclonal antibodies in preclinical species and human. J Pharmacokinet Pharmacodyn. 2012;39(1):67–86.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, D., DuBois, D.C., Almon, R.R. et al. Modeling Sex Differences in Anti-inflammatory Effects of Dexamethasone in Arthritic Rats. Pharm Res 35, 203 (2018). https://doi.org/10.1007/s11095-018-2483-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11095-018-2483-5