Abstract
Purpose
We designed electrospun polycaprolactone mats consisting of nanofibers and microbeads for extended delivery of dexamethasone.
Methods
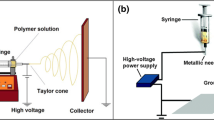
Thin flexible dexamethasone loaded polycaprolactone mats were prepared by electrospinning. The solvents, polymer loading, voltage and tip-to-collector distance were varied to explore the effects on microstructure of the mats. The microstructure was determined by scanning electron microscope imaging; drug transport was measured and modeled, and X-ray diffraction was used to gauge the crystallinity. Drug transport and X-ray diffraction studies were also conducted with a spin cast film for comparison.
Results
Thin mats, about 10 μm in thickness, were prepared by electrospinning. By controlling the voltage and tip-to-collector distance, we achieved a hybrid structure comprising of nanorods (nanofibers) and microbeads. The release profiles were fitted to the diffusion equation to obtain the diffusivities in the spheres and the rods. The diffusivity in the electrospun nanofibers was significantly lower compared to the casted films due to increased crystallinity, which was estimated from X-ray diffraction analysis. The electrospun hybrid mats sustained drug release for the desired duration of a month, in spite of the small thickness of about 10 μm. By comparison, a ten-fold thicker cast film sustains release for about the same duration suggesting about 100-fold decrease in diffusivity in the electrospun mats due to increased crystallinity.
Conclusions
Electrospun polycaprolactone mats are optimal for achieving long release durations due to increased crystallinity. Designing a hybrid structure by controlling the electrospinning parameters can be a useful approach to increase the release durations.







Similar content being viewed by others
Abbreviations
- DCM:
-
Dichloromethane
- DMF:
-
Dimethylformamide
- FWHM:
-
Full width at half maximum
- PCL:
-
Polycaprolactone
- SEM:
-
Scanning electron microscope
- XRD:
-
X-ray diffraction
References
Wagoner MD. Chemical injuries of the eye: current concepts in pathophysiology and therapy. Surv Ophthalmol. 1997;41(4):275–13.
Gaudio P. A review of evidence guiding the use of corticosteroids in the treatment of intraocular inflammation. Ocul Immunol Inflamm. 2004;12(3):169–92.
Dexamethasone dosing information. Drugs.com.; Available from: http://www.drugs.com/mtm/dexamethasone-ophthalmic.html.
Ashburn FS, Goldberg I, Kass MA. Compliance with ocular therapy. Surv Ophthalmol. 1980;24(4):237–48.
Schwartz B. The response of ocular pressure to corticosteroids. Int Ophthalmol Clin. 1966;6(4):929–89.
Urban Jr RC, Cotlier E. Corticosteroid-induced cataracts. Surv Ophthalmol. 1986;31(2):102–10.
Clark AF, Yorio T. Ophthalmic drug discovery. Nat Rev Drug Discov. 2003;2(6):448–59.
Hsu K-H, Gause S, Chauhan A. Review of ophthalmic drug delivery by contact lenses. J Drug Delivery Sci Technol. 2014;24(2):123–35.
Saettone MF, Salminen L. Ocular inserts for topical delivery. Adv Drug Deliv Rev. 1995;16(1):95–106.
Garduño-Anaya MA, De Toledo HC, Hinojosa-González R, Pane-Pianese C, Ríos-Castañeda LC. Dexamethasone inner ear perfusion by intratympanic injection in unilateral Meniere’s disease: a two-year prospective, placebo-controlled, double-blind, randomized trial. Otolaryngol Head Neck. 2005;133(2):285–94.
Chang-Lin J-E, Attar M, Acheampong AA, Robinson MR, Whitcup SM, Kuppermann BD, et al. Pharmacokinetics and pharmacodynamics of a sustained-release dexamethasone intravitreal implant. Invest Ophthalmol Vis Sci. 2011;52(1):80–6.
Agarwal S, Wendorff JH, Greiner A. Use of electrospinning technique for biomedical applications. Polymer. 2008;49(26):5603–21.
Neppalli R, Marega C, Marigo A, Bajgai MP, Kim HY, Causin V. Poly (ε-caprolactone) filled with electrospun nylon fibres: a model for a facile composite fabrication. Eur Polym J. 2010;46(5):968–76.
Park EK, Kim SY, Lee SB, Lee YM. Folate-conjugated methoxy poly (ethylene glycol)/poly (ɛ-caprolactone) amphiphilic block copolymeric micelles for tumor-targeted drug delivery. J Control Release. 2005;109(1):158–68.
Rai B, Teoh S-H, Hutmacher D, Cao T, Ho K. Novel PCL-based honeycomb scaffolds as drug delivery systems for rhBMP-2. Biomaterials. 2005;26(17):3739–48.
Kim H-W, Knowles JC, Kim H-E. Hydroxyapatite/poly (ε-caprolactone) composite coatings on hydroxyapatite porous bone scaffold for drug delivery. Biomaterials. 2004;25(7):1279–87.
Rodoplu D, Mutlu M. Effects of electrospinning setup and process parameters on nanofiber morphology intended for the modification of quartz crystal microbalance surfaces. J Eng Fibers Fabr. 2012;2:118–23.
Koski A, Yim K, Shivkumar S. Effect of molecular weight on fibrous PVA produced by electrospinning. Mater Lett. 2004;58(3):493–7.
Bhardwaj N, Kundu SC. Electrospinning: a fascinating fiber fabrication technique. Biotechnol Adv. 2010;28(3):325–47.
Huang Z-M, Zhang Y-Z, Kotaki M, Ramakrishna S. A review on polymer nanofibers by electrospinning and their applications in nanocomposites. Compos Sci Technol. 2003;63(15):2223–53.
Ramakrishna S, Fujihara K, Teo W-E, Lim T-C, Ma Z. An introduction to electrospinning and nanofibers. New Jersey: World Scientific; 2005.
Kim J, Chauhan A. Dexamethasone transport and ocular delivery from poly(hydroxyethyl methacrylate) gels. Int J Pharm. 2008;353(1–2):205–22.
Lim C, Tan E, Ng S. Effects of crystalline morphology on the tensile properties of electrospun polymer nanofibers. Appl Phys Lett. 2008;92(14):141908.
Wang X, Zhao H, Turng L-S, Li Q. Crystalline morphology of electrospun poly (ε-caprolactone)(PCL) nanofibers. Ind Eng Chem Res. 2013;52(13):4939–49.
Fong H, Chun I, Reneker D. Beaded nanofibers formed during electrospinning. Polymer. 1999;40(16):4585–92.
Eda G, Shivkumar S. Bead structure variations during electrospinning of polystyrene. J Mater Sci. 2006;41(17):5704–8.
Eda G, Shivkumar S. Bead‐to‐fiber transition in electrospun polystyrene. J Appl Polym Sci. 2007;106(1):475–87.
Thompson C, Chase G, Yarin A, Reneker D. Effects of parameters on nanofiber diameter determined from electrospinning model. Polymer. 2007;48(23):6913–22.
Wannatong L, Sirivat A, Supaphol P. Effects of solvents on electrospun polymeric fibers: preliminary study on polystyrene. Polym Int. 2004;53(11):1851–9.
Siepmann J, Siegel RA, Rathbone MJ. Diffusion controlled drug delivery systems. In: Fundamentals and applications of controlled release drug delivery. New York: Springer Science & Business Media; 2011. p. 127–51.
Sung H-J, Meredith C, Johnson C, Galis ZS. The effect of scaffold degradation rate on three-dimensional cell growth and angiogenesis. Biomaterials. 2004;25(26):5735–42.
Sun H, Mei L, Song C, Cui X, Wang P. The in vivo degradation, absorption and excretion of PCL-based implant. Biomaterials. 2006;27(9):1735–40.
Peng C-C, Chauhan A. Ion transport in silicone hydrogel contact lenses. J Membr Sci. 2012;399:95–105.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hsu, KH., Fang, SP., Lin, CL. et al. Hybrid Electrospun Polycaprolactone Mats Consisting of Nanofibers and Microbeads for Extended Release of Dexamethasone. Pharm Res 33, 1509–1516 (2016). https://doi.org/10.1007/s11095-016-1894-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-016-1894-4