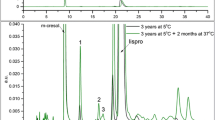
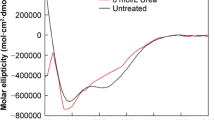
Abstract
Purpose
To identify High Molecular Weight Products (HMWP) formed in human insulin formulation during storage.
Methods
Commercial formulation of human insulin was stored at 37°C for 1 year and HMWP was isolated using preparative size exclusion chromatography (SEC) and reverse phase (RP) chromatography. The primary structure of the isolated species was analysed using liquid chromatography mass spectrometry (LC-MS) and tandem mass spectrometry (MS/MS). To test the hypothesis that amino groups of insulin are involved in HMWP formation, the HMWP content of various formulations spiked with amine compounds or formulations of insulin with modified amino groups was measured.
Results
More than 20 species of HMWP were observed and 16 species were identified using LC-MS. All identified species were covalent dimers of human insulin linked via A21Asn and B29Lys, formed via the formation of an anhydride intermediate at A21Asn. Two types of HMWP were identified, with the covalent link in the open or closed (succinimidyl) form. Some species also contained single deamidation at B3 or the desPhe(B1)-N-oxalyl-Val(B2) modification. Reduced rate of HMWP formation was observed after addition of L-lysine, L-arginine or piperazine or when insulin analogues with methylated N-terminals and side chain amines and A21Gly mutation were used. Formulations of human insulin without zinc and m-cresol were found to contain a different pool of HMWP.
Conclusions
HMWP formed in formulation of human insulin at pH 7.4 with zinc and m-cresol consists primarily of covalent dimers linked via A21Asn and B29Lys. Insulin formulation properties determine the amount and identity of formed HMWP.











Similar content being viewed by others
Abbreviations
- HI:
-
Human insulin
- HMWP:
-
High Molecular Weight Products
- LC-MS:
-
Liquid chromatography mass spectrometry
- MS:
-
Mass spectrometry
- MS/MS:
-
Tandem mass spectrometry
- RP:
-
Reverse phase (chromatography)
- SEC:
-
Size exclusion chromatography
- TCEP:
-
Tris(2-carboxy-ethyl)phosphine
- V8:
-
Endoproteinase Glu-C V8 enzyme
References
Wang W. Instability, stabilization, and formulation of liquid protein pharmaceuticals. Int J Pharm. 1999;185(2):129–88.
Frokjaer S, Otzen DE. Protein drug stability: a formulation challenge. Nat Rev Drug Discov. 2005;4(4):298–306.
Hermeling S, Crommelin DJ, Schellekens H, Jiskoot W. Structure-immunogenicity relationships of therapeutic proteins. Pharm Res. 2004;21(6):897–903.
Manning MC, Chou DK, Murphy BM, Payne RW, Katayama DS. Stability of protein pharmaceuticals: an update. Pharm Res. 2010;27(4):544–75.
Hermeling S, Schellekens H, Maas C, Gebbink MF, Crommelin DJ, Jiskoot W. Antibody response to aggregated human interferon alpha2b in wild-type and transgenic immune tolerant mice depends on type and level of aggregation. J Pharm Sci. 2006;95(5):1084–96.
van Beers MM, Sauerborn M, Gilli F, Brinks V, Schellekens H, Jiskoot W. Oxidized and aggregated recombinant human interferon beta is immunogenic in human interferon beta transgenic mice. Pharm Res. 2011;28(10):2393–402.
Doyle HA, Gee RJ, Mamula MJ. Altered immunogenicity of isoaspartate containing proteins. Autoimmunity. 2007;40(2):131–7.
Jars MU, Hvass A, Waaben D. Insulin aspart (AspB28 human insulin) derivatives formed in pharmaceutical solutions. Pharm Res. 2002;19(5):621–8.
Carpenter JF, Randolph TW, Jiskoot W, Crommelin DJ, Middaugh CR, Winter G, et al. Overlooking subvisible particles in therapeutic protein products: gaps that may compromise product quality. J Pharm Sci. 2009;98(4):1201–5.
Jiskoot W, Randolph TW, Volkin DB, Middaugh CR, Schöneich C, Winter G, et al. Protein instability and immunogenicity: roadblocks to clinical application of injectable protein delivery systems for sustained release. J Pharm Sci. 2012;101(3):946–54.
Singh SK, Afonina N, Awwad M, Bechtold-Peters K, Blue JT, Chou D, et al. An industry perspective on the monitoring of subvisible particles as a quality attribute for protein therapeutics. J Pharm Sci. 2010;99(8):3302–21.
Singh SK. Impact of product-related factors on immunogenicity of biotherapeutics. J Pharm Sci. 2011;100(2):354–87.
Rosenberg AS. Effects of protein aggregates: an immunologic perspective. AAPS J. 2006;8(3):501–7.
Insulin human. European pharmacopoeia. 7 ed. Strasbourg: Council of Europe; 2013
Brange J, Hallund O, Sørensen E. Chemical stability of insulin. 5. Isolation, characterization and identification of insulin transformation products. Acta Pharm Nord. 1992;4(4):223–32.
Maislos M, Mead PM, Gaynor DH, Robbins DC. The source of the circulating aggregate of insulin type I diabetic patients is therapeutic insulin. J Clin Invest. 1986;77(3):717–23.
Schernthaner G. Immunogenicity and allergenic potential of animal and human insulins. Diabetes Care. 1993;16(3):155–65.
Ratner ER, Phillips TM, Steiner M. Persistent cutaneous insulin allergy resulting from high molecular weight insulin aggregates. Diabetes. 1990;39(6):728–33.
Robbins DC, Mead PM. Free covalent aggregates of therapeutic insulin in blood of insulin-dependent diabetics. Diabetes. 1987;36(2):147–51.
Brems DN, Brown PL, Bryant C, Chance RE, Green LK, Long HB, et al. Improved insulin stability through amino acid substitution. Protein Eng. 1992;5(6):519–25.
Darrington RT, Anderson BD. Evidence for a common intermediate in insulin deamidation and covalent dimer formation: effects of pH and aniline trapping in dilute acidic solutions. J Pharm Sci. 1995;84(3):275–82.
Darrington RT, Anderson BD. Effects of insulin concentration and self-association on the partitioning of Its A-21 cyclic anhydride intermediate to desamido insulin and covalent dimer. Pharm Res. 1995;12(7):1077–84.
DeHart MP, Anderson BD. Kinetics and mechanisms of deamidation and covalent amide-linked adduct formation in amorphous lyophiles of a model asparagine-containing peptide. Pharm Res. 2012;29(10):2722–37.
Torosantucci R, Mozziconacci O, Sharov V, Schöneich C, Jiskoot W. Chemical modifications in aggregates of recombinant human insulin induced by metal-catalyzed oxidation: covalent cross-linking via michael addition to tyrosine oxidation products. Pharm Res. 2012;29(8):2276–93.
Printz M, Friess W. Simultaneous detection and analysis of protein aggregation and protein unfolding by size exclusion chromatography with post column addition of fluorescent dye BisANS. J Pharm Sci. 2012;101(2):826–37.
Tantipolphan R, Romeijn S, Engelsman J, Torosantucci R, Rasmussen T, Jiskoot W. Elution behavior of insulin on high-performance size exclusion chromatography at neutral pH. J Pharm Biomed Anal. 2010;52(2):195–202.
Carpenter FH. Relationship of structure to biological activity of insulin as revealed by degradative studies. Am J Med. 1966;40(5):750–8.
Brange J, Havelund S, Hougaard P. Chemical stability of insulin. 2. Formation of higher molecular weight transformation products during storage of pharmaceutical preparations. Pharm Res. 1992;9(6):727–34.
Hawe A, Wiggenhorn M, Van de Weert M, Garbe JHO, Mahler HC, Jiskoot W. Forced degradation of therapeutic proteins. J Pharm Sci. 2012;101(3):895–913.
Huus K, Havelund S, Olsen HB, Van de Weert M, Frokjaer S. Thermal dissociation and unfolding of insulin. Biochemistry. 2005;44(33):11171–7.
Darrington RT, Anderson BD. The role of intramolecular nucleophilic catalysis and the effects of self-association on the deamidation of human insulin at low pH. Pharm Res. 1994;11(6):784–93.
Geiger T, Clarke S. Deamidation, isomerization, and racemization at asparaginyl and aspartyl residues in peptides. Succinimide-linked reactions that contribute to protein degradation. J Biol Chem. 1987;262(2):785–94.
Poulsen C, Jacobsen D, Palm L. Effect of ethylenediamine on chemical degradation of insulin aspart in pharmaceutical solutions. Pharm Res. 2008;25(11):2534–44.
Markussen J, Diers I, Hougaard P, Langkjær L, Norris K, Snel L, et al. Soluble, prolonged-acting insulin derivatives. III. Degree of protraction, crystallizability and chemical stability of insulins substituted in positions A21, B13, B23, B27 and B30. Protein Eng. 1988;2(2):157–66.
Teska BM, Alarcón J, Pettis RJ, Randolph TW, Carpenter JF. Effects of phenol and meta-cresol depletion on insulin analog stability at physiological temperature. J Pharm Sci. 2014;103(8):2255–67.
Brange J, Langkjær L. Chemical stability of insulin. 3. Influence of excipients, formulation, and pH. Acta Pharm Nord. 1992;4(4):149–58.
Derewenda U, Derewenda Z, Dodson EJ, Dodson GG, Reynolds CD, Smith GD, et al. Phenol stabilizes more helix in a new symmetrical zinc insulin hexamer. Nature. 1989;338(6216):594–6.
Kaarsholm NC, Ko HC, Dunn MF. Comparison of solution structural flexibility and zinc binding domains for insulin, proinsulin and miniproinsulin. Biochemistry. 1989;28(10):4427–35.
Peters B, Trout BL. Asparagine deamidation: pH-dependent mechanism from density functional theory. Biochemistry. 2006;45(16):5384–92.
ACKNOWLEDGMENTS AND DISCLOSURES
This study was jointly funded by Innovation Fund Denmark and Novo Nordisk A/S as part of the Industrial Ph.D. programme (Ministry of Higher Education and Science). Laboratory facilities, equipment, and materials were provided by Novo Nordisk. The authors would like to thank Peter Madsen and Thomas Høeg-Jensen from Diabetes Protein Engineering (Novo Nordisk A/S) for providing the insulin analogues.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
Synthesized insulin analogues. Analogue 1 (a), analogue 2 (b) (GIF 10 kb)
Fig. S3
Preparative RP test run to evaluate the number of HMWP species in isolated preparative SEC fractions of HMWP. HMWP species eluted after residual human insulin monomer (A) until column wash gradient was initiated (B) (GIF 12 kb)
Fig. S4
Mass spectra of cross-linked peptide A-II + B-III obtained from samples of HMWP treated with V8 and TCEP. HMWP with intact mass 11590.31 Da (top), HMWP with intact mass 11572.31 Da (bottom) (GIF 17 kb)
Fig. S5
MS/MS of HMWP A-II+B-III peptide (a–b). HMWP 11,590 Da, parrent ion 537.87 m/z (top), HMWP 11,572 Da, parrent ion 531.91 m/z (bottom) (GIF 33 kb)
Fig. S6
MS of B-I peptide obtained from sample of HMWP treated with V8 and TCEP, mass range 440–780 m/z. HMWP B-I peptide with deamidation (+1 Da), deconvoluted mass of 1482.69 Da (top), human insulin B-I peptide, deconvoluted mass of 1481.71 Da (bottom) (GIF 14 kb)
Fig. S7
MS/MS of B-I peptide (a–e) obtained from sample of HMWP treated with V8 and TCEP (top) and human insulin B-I peptide (bottom) (GIF 116 kb)
Fig. S8
MS of B-I peptide obtained from sample of HMWP treated with V8 and TCEP. HMWP B-I peptide with desPhe(B1)-N-oxalyl-Val(B2) modification, 1406.62 Da (top), human insulin B-I peptide, 1481.71 Da (bottom) (GIF 14 kb)
Fig. S9
MS/MS of B-I peptide containing desPhe(B1)-N-oxalyl-Val(B2) modification (a–d) obtained from HMWP treated with V8 and TCEP (top) and human insulin reference (bottom) (GIF 72 kb)
Fig. S10
LC-MS of human insulin formulation spiked with L-arginine (top), lysine (middle) or piperazine (bottom). Calculated monoisotopic masses based on [M+6H]6+: HI+arginine: 5960.72 Da (994.4612 m/z), −HI+lysine: 5932.73 (989.7968 m/z), HI+piperazine 5872.69 Da (989.7968 m/z) (GIF 16 kb)
Fig. S11
Nano-spray MS/MS of human insulin linked to arginine or lysine (a-d) obtained from human insulin formulation spiked with arginine (top) or lysine (middle) and human insulin control (bottom) (GIF 51.2 kb)
Rights and permissions
About this article
Cite this article
Hjorth, C.F., Hubálek, F., Andersson, J. et al. Purification and Identification of High Molecular Weight Products Formed During Storage of Neutral Formulation of Human Insulin. Pharm Res 32, 2072–2085 (2015). https://doi.org/10.1007/s11095-014-1600-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-014-1600-3