Abstract
Purpose
Amorphous drug-polymer solid dispersions have been found to result in improved drug dissolution rates when compared to their crystalline counterparts. However, when the drug exists in the amorphous form it will possess a higher Gibb’s free energy than its associated crystalline state and can recrystallize. Drug-polymer phase diagrams constructed through the application of the Flory Huggins (F-H) theory contain a wealth of information regarding thermodynamic and kinetic stability of the amorphous drug-polymer system. This study was aimed to evaluate the effects of various experimental conditions on the solubility and miscibility detections of drug-polymer binary system.
Methods
Felodipine (FD)-Polyvinylpyrrolidone (PVP) K15 (PVPK15) and FD-Polyvinylpyrrolidone/vinyl acetate (PVP/VA64) were the selected systems for this research. Physical mixtures with different drug loadings were mixed and ball milled. These samples were then processed using Differential Scanning Calorimetry (DSC) and measurements of melting point (Tend) and glass transition (Tg) were detected using heating rates of 0.5, 1.0 and 5.0°C/min.
Results
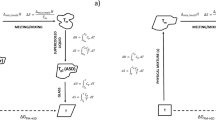
The melting point depression data was then used to calculate the F-H interaction parameter (χ) and extrapolated to lower temperatures to complete the liquid–solid transition curves. The theoretical binodal and spinodal curves were also constructed which were used to identify regions within the phase diagram. The effects of polymer selection, DSC heating rate, time above parent polymer Tg and polymer molecular weight were investigated by identifying amorphous drug miscibility limits at pharmaceutically relevant temperatures.
Conclusion
The potential implications of these findings when applied to a non-ambient processing method such as Hot Melt Extrusion (HME) are also discussed.










Similar content being viewed by others
References
Ku MS, Dulin W. A biopharmaceutical classification-based Right-First-Time formulation approach to reduce human pharmacokinetic variability and project cycle time from First-In-Human to clinical Proof-Of-Concept. Pharm Dev Technol. 2012;17:285–302.
Andrews GP, Abu-Diak O, Kusmanto F, Hornsby P, Hui Z, Jones DS. Physicochemical characterization and drug-release properties of celecoxib hot-melt extruded glass solutions. J Pharm Pharmacol. 2010;62:1580–90.
Merisko-Liversidge EM, Liversidge GG. Drug Nanoparticles: Formulating Poorly Water-Soluble Compounds. Toxicol Pathol. 2008;36:43–8.
Kawabata Y, Wada K, Nakatani M, Yamada S, Onoue S. Formulation design for poorly water-soluble drugs based on biopharmaceutics classification system: basic approaches and practical applications. Int J Pharm. 2011;420:1–10.
Huang L, Tong W. Impact of solid state properties on developability assessment of drug candidates. Adv Drug Deliv Rev. 2004;56:321–34.
Hancock B, Zografi G. Characteristics and significance of the amorphous state in pharmaceutical systems. J Pharm Sci. 1997;86:1–12.
Leuner C, Dressman J. Improving drug solubility for oral delivery using solid dispersions. Eur J Pharm Biopharm. 2000;50:47–60.
Alam MA, Al-Jenoobi FI, Al-mohizea AM. Commercially bioavailable proprietary technologies and their marketed products. Drug Discov Today. 2013;18:936–49.
Lewis S, Dhirendra K, Udupa N, Atin K. Solid dispersions: a review. J Pharm Sci. 2009;22:234.
Laitinen R, Lobmann K, Strachan CJ, Grohganz H, Rades T. Emerging trends in the stabilization of amorphous drugs. Int J Pharm. 2013;453:65–79.
Tian Y, Booth J, Meehan E, Jones DS, Li S, Andrews GP. Construction of Drug-Polymer Thermodynamic Phase Diagrams Using Flory-Huggins Interaction Theory: identifying the Relevance of Temperature and Drug Weight Fraction to Phase Separation within Solid Dispersions. Mol Pharm. 2013;10:236–48.
Mahieu A, Willart J, Dudognon E, Danede F, Descamps M. A New Protocol To Determine the Solubility of Drugs into Polymer Matrixes. Mol Pharm. 2013;10:560–6.
Friesen DT, Shanker R, Crew M, Smithey DT, Curatolo WJ, Nightingale JAS. Hydroxypropyl Methylcellulose Acetate Succinate-Based Spray-Dried Dispersions: an Overview. Mol Pharm. 2008;5:1003–19.
Lin D, Huang Y. A thermal analysis method to predict the complete phase diagram of drug-polymer solid dispersions. Int J Pharm. 2010;399:109–15.
Marsac PJ, Li T, Taylor LS. Estimation of Drug-Polymer Miscibility and Solubility in Amorphous Solid Dispersions Using Experimentally Determined Interaction Parameters. Pharm Res. 2009;26:139–51.
Marsac PJ, Shamblin SL, Taylor LS. Theoretical and practical approaches for prediction of drug-polymer miscibility and solubility. Pharm Res. 2006;23:2417–26.
Albers J, Matthee K, Knop K, Kleinebudde P. Evaluation of Predictive Models for Stable Solid Solution Formation. J Pharm Sci. 2011;100:667–80.
Zhao Y, Inbar P, Chokshi HP, Malick AW, Choi DS. Prediction of the Thermal Phase Diagram of Amorphous Solid Dispersions by Flory-Huggins Theory. J Pharm Sci. 2011;100:3196–207.
Caron V, Tajber L, Corrigan OI, Healy AM. A Comparison of Spray Drying and Milling in the Production of Amorphous Dispersions of Sulfathiazole/Polyvinylpyrrolidone and Sulfadimidine/Polyvinylpyrrolidone. Mol Pharm. 2011;8:532–42.
Tian Y, Caron V, Jones DS, Healy A, Andrews GP, Tian Y, et al. Using Flory–Huggins phase diagrams as a pre‐formulation tool for the production of amorphous solid dispersions: a comparison between hot‐melt extrusion and spray drying. J Pharm Pharmacol. 2014;66:256.
Yang M, Wang P, Gogos C. Prediction of acetaminophen’s solubility in poly(ethylene oxide) at room temperature using the Flory-Huggins theory. Drug Dev Ind Pharm. 2013;39:102–8.
Rubinstein M, Colby RH. Polymer physics. 1st ed. New York, USA: Oxford University Press; 2003.
van Krevelen D. Properties of polymers: their estimation and correlation with chemical structure. Amsterdam: Elsevier; 1976.
Forster A, Hempenstall J, Tucker I, Rades T. Selection of excipients for melt extrusion with two poorly water-soluble drugs by solubility parameter calculation and thermal analysis. Int J Pharm. 2001;226:147–61.
Tao J, Sun Y, Zhang GGZ, Yu L. Solubility of Small-Molecule Crystals in Polymers: d-Mannitol in PVP, Indomethacin in PVP/VA, and Nifedipine in PVP/VA. Pharm Res. 2009;26:855–64.
Thakral S, Thakral NK. Prediction of drug-polymer miscibility through the use of solubility parameter based flory-huggins interaction parameter and the experimental validation: PEG as model polymer. J Pharm Sci. 2013;102:2254–63.
Young RJ, Lovell PA. Introduction to polymers. 2nd ed. London: Chapman & Hall; 1991.
Gupta J, Nunes C, Vyas S, Jonnalagadda S. Prediction of Solubility Parameters and Miscibility of Pharmaceutical Compounds by Molecular Dynamics Simulations. J Phys Chem B. 2011;115:2014–23.
Marsac PJ, Konno H, Taylor LS. A comparison of the physical stability of amorphous felodipine and nifedipine systems. Pharm Res. 2006;23:2306–16.
Coleman N, Craig D. Modulated temperature differential scanning calorimetry: A novel approach to pharmaceutical thermal analysis. Int J Pharm. 1996;135:13–29.
Kestur US, Lee H, Santiago D, Rinaldi C, Won Y, Taylor LS. Effects of the molecular weight and concentration of polymer additives, and temperature on the melt crystallization kinetics of a small drug molecule. Cryst Growth Des. 2010;10:3585–95.
Baird JA, Taylor LS. Evaluation of amorphous solid dispersion properties using thermal analysis techniques. Adv Drug Deliv Rev. 2012;64:396–421.
Keen JM, Martin C, Machado A, Sandhu H, McGinity JW, DiNunzio JC. Investigation of process temperature and screw speed on properties of a pharmaceutical solid dispersion using corotating and counter-rotating twin-screw extruders. J Pharm Pharmacol. 2014;66:204–17.
F. Qian, J. Huang, M.A. Hussain, Drug-Polymer Solubility and Miscibility: Stability Consideration and Practical Challenges in Amorphous Solid Dispersion Development, J. Pharm. Sci. 99 (2010).
Gramaglia D, Conway B, Kett V, Malcolm R, Batchelor H. High speed DSC (hyper-DSC) as a tool to measure the solubility of a drug within a solid or semi-solid matrix. Int J Pharm. 2005;301:1–5.
Al-Obaidi H, Lawrence MJ, Al-Saden N, Ke P. Investigation of griseofulvin and hydroxypropylmethyl cellulose acetate succinate miscibility in ball milled solid dispersions. Int J Pharm. 2013;443:95–102.
Mohan R, Lorenz H, Myerson A. Solubility measurement using differential scanning calorimetry. Ind Eng Chem Res. 2002;41:4854–62.
Pijpers TFJ, Mathot VBF, Goderis B, Scherrenberg RL. van der Vegte, Eric W., High speed calorimetry for the study of kinetics of (de)vitrification, crystallization, and melting of macromolecules. Macromolecules. 2002;35:3601–13.
TANAKA H, NISHI T. Local Phase-Separation at the Growth Front of a Polymer Spherulite during Crystallization and Nonlinear Spherulitic Growth in a Polymer Mixture with a Phase-Diagram. Phys Rev a. 1989;39:783–94.
TANAKA H, NISHI T. New Types of Phase-Separation Behavior during the Crystallization Process in Polymer Blends with Phase-Diagram. Phys Rev Lett. 1985;55:1102–5.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Donnelly, C., Tian, Y., Potter, C. et al. Probing the Effects of Experimental Conditions on the Character of Drug-Polymer Phase Diagrams Constructed Using Flory-Huggins Theory. Pharm Res 32, 167–179 (2015). https://doi.org/10.1007/s11095-014-1453-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-014-1453-9