ABSTRACT
Purpose
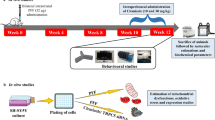
To prepare an angiopep-conjugated dendrigraft poly-L-lysine (DGL)-based gene delivery system and evaluate the neuroprotective effects in the rotenone-induced chronic model of Parkinson’s disease (PD).
Methods
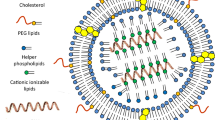
Angiopep was applied as a ligand specifically binding to low-density lipoprotein receptor-related protein (LRP) which is overexpressed on blood-brain barrier (BBB), and conjugated to biodegradable DGL via hydrophilic polyethyleneglycol (PEG), yielding DGL-PEG-angiopep (DPA). In vitro characterization was carried out. The neuroprotective effects were evaluated in a chronic parkinsonian model induced by rotenone using a regimen of multiple dosing intravenous administrations.
Results
The successful synthesis of DPA was demonstrated via 1H-NMR. After encapsulating the therapeutic gene encoding human glial cell line-derived neurotrophic factor (hGDNF), DPA/hGDNF NPs showed a sphere-like shape with the size of 119 ± 12 nm and zeta potential of 8.2 ± 0.7 mV. Angiopep-conjugated NPs exhibited higher cellular uptake and gene expression in brain cells compared to unmodified counterpart. The pharmacodynamic results showed that rats in the group with five injections of DPA/hGDNF NPs obtained best improved locomotor activity and apparent recovery of dopaminergic neurons compared to those in other groups.
Conclusion
This work provides a practical non-viral gene vector for long-term gene therapy of chronic neurodegenerative disorders.








Similar content being viewed by others
Abbreviations
- BBB:
-
blood-brain barrier
- BCECs:
-
brain capillary endothelial cells
- DGL:
-
dendrigraft poly-L-lysine
- DPA:
-
DGL-PEG-angiopep
- GFP:
-
green fluorenscent protein
- hGDNF :
-
the gene encoding human glial cell line-derived neurotrophic factor
- LRP:
-
low-density lipoprotein receptor-related protein
- NMR:
-
nuclear magnetic resonance
- NPs:
-
nanoparticles
- PAMAM:
-
polyamidoamine
- PBS:
-
phosphate-buffered solution
- PD:
-
Parkinson’s disease
- PEG:
-
polyethylene glycol
- PPA:
-
PAMAM-PEG-angiopep
- TH:
-
tyrosine hydroxylase
REFERENCES
Di Stefano A, Sozio P, Cerasa LS, Iannitelli A. L-Dopa prodrugs: an overview of trends for improving Parkinson’s disease treatment. Curr Pharm Des. 2011;17(32):3482–93.
Reichmann H, Emre M. Optimizing levodopa therapy to treat wearing-off symptoms in Parkinson’s disease: focus on levodopa/carbidopa/entacapone. Expert Rev Neurother. 2012;12(2):119–31.
Björklund A, Björklund T, Kirik D. Gene therapy for dopamine replacement in Parkinson’s disease. Sci Transl Med. 2009;1(2):2ps2.
Huang RQ, Ke WL, Liu Y, Wu DD, Feng LY, Jiang C, et al. Gene therapy using lactoferrin-modified nanoparticles in a rotenone-induced chronic Parkinson model. J Neurol Sci. 2010;290(1–2):123–30.
Boado RJ, Pardridge WM. The Trojan horse liposome technology for nonviral gene transfer across the blood-brain barrier. J Drug Deliv. 2011;2011:296151.
Wagner S, Zensi A, Wien SL, Tschickardt SE, Maier W, Vogel T, et al. Uptake mechanism of ApoE-modified nanoparticles on brain capillary endothelial cells as a blood-brain barrier model. PLoS One. 2012;7(3):e32568.
Demeule M, Currie JC, Bertrand Y, Ché C, Nguyen T, Régina A, et al. Involvement of the low-density lipoprotein receptor-related protein in the transcytosis of the brain delivery vector angiopep-2. J Neurochem. 2008;106(4):1534–44.
Ke WL, Shao K, Huang RQ, Han L, Liu Y, Li JF, et al. Gene delivery targeted to the brain using an Angiopep-conjugated polyethyleneglycol-modified polyamidoamine dendrimer. Biomaterials. 2009;30(36):6976–85.
Huang RQ, Liu SH, Shao K, Han L, Ke WL, Liu Y, et al. Evaluation and mechanism studies of PEGylated dendrigraft poly-L-lysines as novel gene delivery vectors. Nanotechnology. 2010;21(26):265101.
Huang RQ, Ke WL, Han L, Li JF, Liu SH, Jiang C. Targeted delivery of chlorotoxin-modified DNA-loaded nanoparticles to glioma via intravenous administration. Biomaterials. 2011;32(9):2399–406.
Xie Y, Ye LY, Zhang XB, Cui W, Lou JN, Nagai T, et al. Transport of nerve growth factor encapsulated into liposomes across the blood-brain barrier: In vitro and in vivo studies. J Control Release. 2005;105(1–2):106–19.
Canzoniero LM, Adornetto A, Secondo A, Magi S, Dell’aversano C, Scorziello A, et al. Involvement of the nitric oxide/protein kinase G pathway in polychlorinated biphenyl-induced cell death in SH-SY 5Y neuroblastoma cells. J Neurosci Res. 2006;84(3):692–7.
Huang RQ, Han L, Li JH, Ren FL, Ke WL, Jiang C, et al. Neuroprotection in a 6-OHDA-lesioned Parkinson model using Lactoferrin-modified Nanoparticles. J Gene Med. 2009;11(9):754–63.
Huang RQ, Ke WL, Liu Y, Jiang C, Pei YY. The use of lactoferrin as a ligand for targeting the polyamidoamine-based gene delivery system to the brain. Biomaterials. 2008;29(2):238–46.
Spuch C, Navarro C. Liposomes for targeted delivery of active agents against neurodegenerative diseases (Alzheimer’s disease and Parkinson’s disease). J Drug Deliv. 2011;2011:469679.
Shao K, Huang RQ, Li JF, Han L, Ye LY, Lou JN, et al. Angiopep-2 modified PE-PEG based polymeric micelles for amphotericin B delivery targeted to the brain. J Control Release. 2010;147(1):118–26.
Alam M, Schmidt WJ. Rotenone destroys dopaminergic neurons and induces parkinsonian symptoms in rats. Behav Brain Res. 2002;136(1):317–24.
Samantaray S, Knaryan VH, Guyton MK, Matzelle DD, Ray SK, Banik NL. The parkinsonian neurotoxin rotenone activates calpain and caspase-3 leading to motoneuron degeneration in spinal cord of Lewis rats. Neuroscience. 2007;146(2):741–55.
Hirsch EC, Höglinger G, Rousselet E, Breidert T, Parain K, Feger J, et al. Animal models of Parkinson’s disease in rodents induced by toxins: an update. J Neural Transm Suppl. 2003;65:89–100.
Sherer TB, Betarbet R, Kim JH, Greenamyre JT. Selective microglial activation in the rat rotenone model of Parkinson’s disease. Neurosci Lett. 2003;341(2):87–90.
Mercanti G, Bazzu G, Giusti P. A 6-hydroxydopamine in vivo model of Parkinson’s disease. Methods Mol Biol. 2012;846:355–64.
Li JF, Zhou L, Ye DY, Huang SX, Shao K, Huang RQ, et al. Choline-derivate-modified nanoparticles for brain-targeting gene delivery. Adv Mater. 2011;23(39):4516–20.
Liu SH, Guo YB, Huang RQ, Li JF, Huang SX, Kuang YY, et al. Gene and doxorubicin co-delivery system for targeting therapy of glioma. Biomaterials. 2012;33(19):4907–16.
Tsogas I, Theodossiou T, Sideratou Z, Paleos CM, Collet H, Rossi JC, et al. Interaction and transport of poly(L-lysine) dendrigrafts through liposomal and cellular membranes: the role of generation and surface functionalization. Biomacromolecules. 2007;8(10):3263–70.
Duarte EP, Curcio M, Canzoniero LM, Duarte CB. Neuroprotection by GDNF in the ischemic brain. Growth Factors. 2012;30(4):242–57.
Boger HA, Middaugh LD, Huang P, Zaman V, Smith AC, Hoffer BJ, et al. A partial GDNF depletion leads to earlier age-related deterioration of motor function and tyrosine hydroxylase expression in the substantia nigra. Exp Neurol. 2006;202(2):336–47.
Jiang C, Koyabu N, Yonemitsu Y, Shimazoe T, Watanabe S, Naito M, et al. In vivo delivery of glial cell-derived neurotrophic factor across the blood-brain barrier by gene transfer into brain capillary endothelial cells. Hum Gene Ther. 2003;14(12):1181–91.
ACKNOWLEDGMENTS AND DISCLOSURES
Rongqin Huang and Haojun Ma contributed equally to this work. This work was supported by the grants from Specialized Research Fund for Doctoral Program (20090071120066), “Zhuo Xue” Talent Plan of Fudan University, “Chen Guang” project supported by Shanghai Municipal Education Commission and Shanghai Education Development Foundation, and National Key Basic Research Program (2013CB932502) of China (973 Program).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, R., Ma, H., Guo, Y. et al. Angiopep-Conjugated Nanoparticles for Targeted Long-Term Gene Therapy of Parkinson’s Disease. Pharm Res 30, 2549–2559 (2013). https://doi.org/10.1007/s11095-013-1005-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-013-1005-8