Abstract
Purpose
Computer simulations are utilized during pharmaceutical development in order to design appropriate formulation based on the absorption, distribution, metabolism, and excretion (ADME) and physicochemical properties of target compounds, so that adequate prescriptions are offered to patients. Oro-cecal transit time (OCTT) is an important factor affecting these simulations because the absorption of drug that administered orally and the resultant pharmacokinetic profile are expressed as a function of time. Given the large intra- and inter-individual variance in OCTT, it is unsurprising that an accurate model has not yet been proposed.
Methods
We conducted a meta-analysis using subject-level data to construct a statistical model that predicted OCTT. Literature that utilized lactulose to measure OCTT was identified and analyzed using a mixed-effects model.
Results
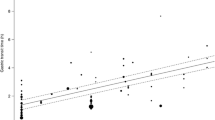
The OCTTs of fasting healthy subjects were expressed using a linear model, with the amount of lactulose as the single significant explanatory factor. We found that this model could statistically distinguish the OCTTs of subjects with altered physical status from those of healthy people. Specifically, cystic fibrosis and celiac disease most significantly affected OCTT.
Conclusion
The OCTT models developed herein incorporate inter-subject variations and can contribute to providing more accurate predictions of drug pharmacokinetic profiles.


Similar content being viewed by others
Abbreviations
- ADME:
-
absorption, distribution, metabolism and elimination
- FDA:
-
Food and Drug Administration
- GET:
-
gastric emptying time
- GI:
-
gastrointestinal
- LHBT:
-
lactulose hydrogen breath test
- OCTT:
-
oro-cecal transit time
- PK:
-
pharmacokinetics
- SITT:
-
small intestine transit time
References
Varum FJ, Merchant HA, Basit AW. Oral modified-release formulations in motion: the relationship between gastrointestinal transit and drug absorption. Int J Pharm. 2010;395(1–2):26–36.
Yu LX, Lipka E, Crison JR, Amidon GL. Transport approaches to the biopharmaceutical design of oral drug delivery systems: prediction of intestinal absorption. Adv Drug Deliv Rev. 1996;19(3):359–76.
Willmann S, Schmitt W, Keldenich J, Lippert J, Dressman JB. A physiological model for the estimation of the fraction dose absorbed in humans. J Med Chem. 2004;47(16):4022–31.
Caride VJ, Prokop EK, Troncale FJ, Buddoura W, Winchenbach K, McCallum RW. Scintigraphic determination of small intestinal transit time: comparison with the hydrogen breath technique. Gastroenterology. 1984;86(4):714–20.
Weitschies W, Kosch O, Mönnikes H, Trahms L. Magnetic Marker Monitoring: an application of biomagnetic measurement instrumentation and principles for the determination of the gastrointestinal behavior of magnetically marked solid dosage forms. Adv Drug Deliv Rev. 2005;57(8):1210–22.
Wutzke KD, Heine WE, Plath C, Leitzmann P, Radke M, Mohr C, Richter I, Gülzow HU, Hobusch D. Evaluation of oro-coecal transit time: a comparison of the lactose-[13C, 15N]ureide 13CO2- and the lactulose H2-breath test in humans. Eur J Clin Nutr. 1997;51(1):11–9.
La Brooy SJ, Male PJ, Beavis AK, Misiewicz JJ. Assessment of the reproducibility of the lactulose H2 breath test as a measure of mouth to caecum transit time. Gut. 1983;24(10):893–6.
Ladas SD, Latoufis C, Giannopoulou H, Hatziioannou J, Raptis SA. Reproducible lactulose hydrogen breath test as a measure of mouth-to-cecum transit time. Dig Dis Sci. 1989;34(6):919–24.
Diggory RT, Cuschieri A. The effect of dose and osmolality of lactulose on the oral-caecal transit time determined by the hydrogen breath test and the reproducibility of the test in normal subjects. Ann Clin Res. 1985;17(6):331–3.
Stewart LA, Clarke MJ. Practical methodology of meta-analyses (overviews) using updated individual patient data. Cochrane Working Group. Stat Med. 1995;14(19):2057–79.
Berlin JA, Santanna J, Schmid CH, Szczech LA, Feldman HI, Anti-Lymphocyte Antibody Induction Therapy Study Group. Individual patient- versus group-level data meta-regressions for the investigation of treatment effect modifiers: ecological bias rears its ugly head. Stat Med. 2002;21(3):371–87.
Lambert PC, Sutton AJ, Abrams KR, Jones DR. A comparison of summary patient-level covariates in meta-regression with individual patient data meta-analysis. J Clin Epidemiol. 2002;55(1):86–94.
Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analysis: the PRISMA statement. PLOS Medicine. 2010;6(7)e1000097:1–6.
Fadda HM, McConnell EL, Short MD, Basit AW. Meal-induced acceleration of tablet transit through the human small intestine. Pharm Res. 2009;26(2):356–60.
Priebe MG, Wachters-Hagedoorn RE, Stellaard F, Heiner AM, Elzinga H, Vonk RJ. Oro-cecal transit time: influence of a subsequent meal. Eur J Clin Invest. 2004;34(6):417–21.
Staniforth DH, Rose D. Statistical analysis of the lactulose/breath hydrogen test in the measurement of orocaecal transit: its variability and predictive value in assessing drug action. Gut. 1989;30(2):171–5.
Engage Digitizer: 2007. http://digitizer.sourceforge.net/
Matsumoto T, Iida M, Hirakawa M, Hirakawa K, Kuroki F, Lee S, Nanbu T, Fujishima M. Breath hydrogen test using water-diluted lactulose in patients with gastrointestinal amyloidosis. Dig Dis Sci. 1991;36(12):1756–60.
Chiarioni G, Bassotti G, Germani U, Battaglia E, Brentegani MT, Morelli A, Vantini I. Gluten-free diet normalizes mouth-to-cecum transit of a caloric meal in adult patients with celiac disease. Dig Dis Sci. 1997;42(10):2100–5.
Basilisco G, Camboni G, Bozzani A, Vita P, Doldi S, Bianchi PA. Orocecal transit delay in obese patients. Dig Dis Sci. 1989;34(4):509–12.
Camboni G, Basilisco G, Bozzani A, Bianchi PA. Repeatability of lactulose hydrogen breath test in subjects with normal or prolonged orocecal transit. Dig Dis Sci. 1988;33(12):1525–7.
Bali A, Stableforth DE, Asquith P. Prolonged small-intestinal transit time in cystic fibrosis. Br Med J (Clin Res Ed). 1983;287(6398):1011–3.
Scarpello JH, Greaves M, Sladen GE. Small intestinal transit in diabetics. Br Med J. 1976;2(6046):1225–6.
Corbett CL, Thomas S, Read NW, Hobson N, Bergman I, Holdsworth CD. Electrochemical detector for breath hydrogen determination: measurement of small bowel transit time in normal subjects and patients with the irritable bowel syndrome. Gut. 1981;22(10):836–40.
Gorard DA, Libby GW, Farthing MJ. Influence of antidepressants on whole gut and orocaecal transit times in health and irritable bowel syndrome. Aliment Pharmacol Ther. 1994;8(2):159–66.
Pilotto A, Franceschi M, Del Favero G, Fabrello R, Di Mario F, Valerio G. The effect of aging on oro-cecal transit time in normal subjects and patients with gallstone disease. Aging (Milano). 1995;7(4):234–7.
Szilagyi A, Salomon R, Smith BE, Martin M, Seidman E. Determinants of prolonged oral cecal transit time during late phase pregnancy. Clin Invest Med. 1996;19(1):20–7.
Caride VJ, Prokop EK, Troncale FJ, Buddoura W, Winchenbach K, McCallum RW. Scintigraphic determination of small intestinal transit time: comparison with the hydrogen breath technique. Gastroenterology. 1984;86(4):714–20.
Pressman JH, Hofmann AF, Witztum KF, Gertler SL, Steinbach JH, Stokes K, Kelts DG, Stone DM, Jones BR, Dharmsathaphorn K. Limitations of indirect methods of estimating small bowel transit in man. Dig Dis Sci. 1987;32(7):689–99.
Lehtola J, Jauhonen P, Kesäniemi A, Wikberg R, Gordin A. Effect of erythromycin on the oro-caecal transit time in man. Eur J Clin Pharmacol. 1990;39(6):555–8.
Pfeiffer A, Schmidt T, Höller T, Herrmann H, Pehl C, Wendl B, Kaess H. Effect of trospium chloride on gastrointestinal motility in humans. Eur J Clin Pharmacol. 1993;44(3):219–23.
Dukowicz AC, Lacy BE, Levine GM. Small intestinal bacterial overgrowth: a comprehensive review. Gastroenterol Hepatol (N Y). 2007;3(2):112–22.
Basilisco G, Risicato R, Bnazzi P, Di Sario A, Portincasa P. H2-breath testing for evaluation of oro-caecal transit time. Aliment Pharmacol Ther. 2009;29(Suppl1):1–49.
Hirakawa M, Iida M, Kohrogi N, Fujishima M. Hydrogen breath test assessment of orocecal transit time: comparison with barium meal study. Am J Gastroenterol. 1988;83(12):1361–3.
Karalis V, Macheras P, Van Peer A, Shah VP. Bioavailability and bioequivalence: focus on physiological factors and variability. Pharm Res. 2008;25(8):1956–62.
Jiang W, Kim S, Zhang X, Lionberger RA, Davit BM, Conner DP, Yu LX. The role of predictive biopharmaceutical modeling and simulation in drug development and regulatory evaluation. Int J Pharm. 2011;418(2):151–60.
Bryson JC, Dukes GE, Kirby MG, Heizer WD, Powell JR. Effect of altering small bowel transit time on sustained release theophylline absorption. J Clin Pharmacol. 1989;29(8):733–8.
Zariffa NM, Patterson SD. Population and individual bioequivalence: lessons from real data and simulation studies. J Clin Pharmacol. 2001;41(8):811–22.
Hamburg MA, Collins FS. The path to personalized medicine. N Engl J Med. 2010;22, 363(4):301–304.
U.S. Department of Health and Human Services, FDA, CDER. Guidance for Industry: Food-Effect Bioavailability and Fed Bioequivalence Studies. 2002. http://www.fda.gov/downloads/regulatoryinformation/guidances/ucm126833.pdf
Basilisco G, Camboni G, Bozzani A, Paravicini M, Bianchi PA. Oral naloxone antagonizes loperamide-induced delay of orocecal transit. Dig Dis Sci. 1987;32(8):829–32.
Beaugerie L, Burger AJ, Cadranel JF, Lamy P, Gendre JP, Le Quintrec Y. Modulation of orocaecal transit time by hypnosis. Gut. 1991;32(4):393–4.
Gorard DA, Libby GW, Farthing MJ. 5-Hydroxytryptamine and human small intestinal motility: effect of inhibiting 5-hydroxytryptamine reuptake. Gut. 1994;35(4):496–500.
Gorard DA, Gomborone JE, Libby GW, Farthing MJ. Intestinal transit in anxiety and depression. Gut. 1996;39(4):551–5.
Meshkinpour H, Kemp C, Fairshter R. Effect of aerobic exercise on mouth-to-cecum transit time. Gastroenterology. 1989;96(3):938–41.
Nordgaard I, Rumessen JJ, Nielsen SA, Gudmand-Høyer E. Absorption of wheat starch in patients resected for left-sided colonic cancer. Scand J Gastroenterol. 1992;27(8):632–4.
Rubinoff MJ, Piccione PR, Holt PR. Clonidine prolongs human small intestine transit time: use of the lactulose-breath hydrogen test. Am J Gastroenterol. 1989;84(4):372–4.
Rumessen JJ, Hamberg O, Gudmand-Høyer E. Influence of orocaecal transit time on hydrogen excretion after carbohydrate malabsorption. Gut. 1989;30(6):811–4.
Staniforth DH. Effect of drugs on oro-caecal transit time assessed by the lactulose/breath hydrogen method. Eur J Clin Pharmacol. 1987;33(1):55–8.
Van Wyk M, Sommers DK, Steyn AG. Evaluation of gastrointestinal motility using the hydrogen breath test. Br J Clin Pharmacol. 1985;20(5):479–81.
Vazquez-Olivencia W, Shah P, Pitchumoni CS. The effect of red and black pepper on orocecal transit time. J Am Coll Nutr. 1992;11(2):228–31.
Yuan CS, Foss JF, O’Connor M, Toledano A, Roizen MF, Moss J. Methylnaltrexone prevents morphine-induced delay in oral-cecal transit time without affecting analgesia: a double-blind randomized placebo-controlled trial. Clin Pharmacol Ther. 1996;59(4):469–75.
Yuan CS, Foss JF, Osinski J, Toledano A, Roizen MF, Moss J. The safety and efficacy of oral methylnaltrexone in preventing morphine-induced delay in oral-cecal transit time. Clin Pharmacol Ther. 1997;61(4):467–75.
Yuan CS, Foss JF, O’Connor M, Karrison T, Osinski J, Roizen MF, Moss J. Effects of enteric-coated methylnaltrexone in preventing opioid-induced delay in oral-cecal transit time. Clin Pharmacol Ther. 2000;67(4):398–404.
Yuan CS, Wei G, Foss JF, O’Connor M, Karrison T, Osinski J. Effects of subcutaneous methylnaltrexone on morphine-induced peripherally mediated side effects: a double-blind randomized placebo-controlled trial. J Pharmacol Exp Ther. 2002;300(1):118–23.
Yuan CS, Doshan H, Charney MR, O’connor M, Karrison T, Maleckar SA, Israel RJ, Moss J. Tolerability, gut effects, and pharmacokinetics of methylnaltrexone following repeated intravenous administration in humans. J Clin Pharmacol. 2005;45(5):538–46.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kokubo, T., Matsui, S. & Ishiguro, M. Meta-analysis of Oro-cecal Transit Time in Fasting Subjects. Pharm Res 30, 402–411 (2013). https://doi.org/10.1007/s11095-012-0882-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-012-0882-6