ABSTRACT
Purpose
A challenge in the field of nanobubbles, including lipobubbles and polymeric nanobubbles, is identification of formulation approaches to enhance circulation time or “bubble life” in the specific organ to allow for organ visualization. The aim of this study was to investigate the potential of two specific preparation approaches, polymeric surface modification to lipobubbles and a one-step approach for the preparation of ionotropically originated polymeric hydrogel nanobubbles for the production of biocompatible, biodegradable, and sufficiently echogenic (‘flexible’) bubbles, preferably within the nanometer range, that possess an enhanced in vivo lifetime compared to an unmodified lipobubble to allow visualization of the lymph node vasculature.
Methods
In the first approach, formed liposomes (basic and polymerically enhanced) were sequentially layered with appropriate cationic and anionic polyelectrolytes followed by transformation into polymer-coated nanobubbles. In addition, a one-step approach was employed for the fabrication of ionotropically originated polymeric hydrogel bubbles.
Results
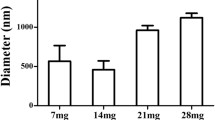
Bubble lifetime was marginally enhanced by self-deposition of polyelectrolytes onto the normal lipobubble, however, not significantly (P = 0.0634). In general, formulations possessing a higher ratio of anionic:cationic coats and highly anionic overall surface charge (−20.62 mV to −17.54 mV) possessed an enhanced lifetime. The improvement in bubble lifetime was significant when a purely polymeric polyionic hydrogel bubble shell was instituted compared to a normal unmodified lipobubble (P = 0.004). There was enhanced persistence of these systems compared to lipobubbles, attributed to the highly flexible, interconnected hydrogel shell which minimized gas leakage. The prolonged contrast signal may also be attributed to a degree of polymeric deposition/endothelial attachment.
Conclusions
This study identified the relevance of polymeric modifications to nanobubbles for an improved circulating lifetime, which would be essential for application of these systems in passive drug or gene targeting via the enhanced permeability and retention effect.





Similar content being viewed by others
REFERENCES
Hernot S, Klibanov AL. Microbubbles in ultrasound-triggered drug and gene delivery. Adv Drug Deliv Rev (Ultrasound in Drug and Gene Delivery). 2008;60:1153–66.
Kodama T, Tomita N, Horie S, Sax N, Iwasaki H, Suzuki R, et al. Morphological study of acoustic liposomes using transmission electron microscopy. J Electron Microsc. 2010;59:187–96.
Takeuchi H, Yamamoto H, Toyoda T, Toyobuku H, Hino T, Kawashima Y. Physical stability of size controlled small unilameller liposomes coated with a modified polyvinyl alcohol. Int J Pharm. 1998;164:103–11.
Walker HW, Grant SB. Influence of surface charge and particle size on the stabilization of colloidal particles by model polyelectrolytes. Colloids Surf A-Physicochem Eng Asp. 1998;135:123–33.
Bogdanovic G, Sennerfors T, Zhmud B, Tiberg F. Formation and structure of polyelectrolyte and nanoparticle multilayers: effect of particle characteristics. J Colloid Interface Sci. 2002;255:44–51.
Yap HP, Quinn JF, Ng SM, Cho J, Caruso F. Colloid surface engineering via deposition of multilayered thin films from polyelectrolyte blend solutions. Langmuir. 2005;21:4328–33.
Haidar ZS, Hamdy RC, Tabrizian M. Protein release kinetics for core-shell hybrid nanoparticles based on the layer-by-layer assembly of alginate and chitosan on liposomes. Biomaterials. 2008;29(9):1207–15.
Decher G. Fuzzy nanoassemblies: toward layered polymeric multicomposites. Science. 1997;277:1232–7.
Feinstein SB, Lang RM, Dick C, Neumann A, Al-Sadir J, Chua KG, et al. Contrast echocardiography during coronary arteriography in humans: perfusion and anatomic studies. J Am Coll Cardiol. 1988;11:59–65.
Shapiro JR, Reisner SA, Lichtenberg GS, Meltzer RS. Intravenous contrast echocardiography with use of sonicated albumin in humans: systolic disappearance of left ventricular contrast after transpulmonary transmission. J Am Coll Cardiol. 1990;16:1603–7.
Wheatley MA, Schropet B, Shen P. Contrast agents for diagnostic ultrasound: development and evaluation of polymer-coated microbubbles. Biomatenals. 1990;11:713–8.
Kwok KK, Groves MJ, Burgess DJ. Production of 5–15βm diameter alginate-polylysine microcapsules by an air-atomization technique. Pharm Res. 1991;8:341–4.
Schneider M, Bussat P, Barrau M-B, Arditi M, Yan F, Hybl E. Polymeric microballoons as ultrasound contrast agents: Physical and ultrasonic properties compared with sonicated albumin. Invest Radiol. 1992;27:134–9.
Cohen S, Andrianov AK, Wheatley M, Allcock HR, Langer RS. Gas-filled polymeric microbubbles for ultrasound imaging. United States Patent 5487390, 1996.
Matsumura Y, Maeda H. A new concept for macromolecular therapeutics in cancer-chemotherapy—mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986;46:6387–92.
Agnihotri SA, Mallikarjuna NN, Aminabhavi TM. Recent advances on chitosan-based micro- and nanoparticles in drug delivery. J Control Release. 2004;100:5–28.
Douglas KL, Tabrizian MJ. Effect of experimental parameters on the formation of alginate–chitosan nanoparticles and evaluation of their potential application as DNA carrier. J Biomater Sci, Polym Ed. 2005;16:43–56.
Motwani SK, Chopra S, Talegaonkar S, Kohli K, Ahmad FJ, Khar RK. Chitosan–sodium alginate nanoparticles as submicroscopic reservoirs for ocular delivery: formulation, optimisation and in vitro characterisation. Eur J Pharm Biopharm. 2008;68:513–25.
Heskins M, Guillet JE. Solution properties of poly(N-isopropylacrylamide). J Macromol Sci Chem. 1968;A2:1441–55.
Chung JE, Yokoyama M, Yamato M, Aoyagi T, Sakurai Y, Okano T. Thermo-responsive drug delivery from polymeric micelles constructed using block copolymers of poly(N-isopropylacrylamide) and poly(butylmethacrylate). J Control Release. 1999;62:115–27.
Murphy ED, Roths JB. Autoimmunity and lymphoproliferation: induction by mutant gene lpr, and acceleration by a male-associated factor in strain BXSB mice. New York: Elsevier North Holland Inc.; 1978.
O’ Doherty M. Liposome literature review. Nanobiotechnology and Bioanalysis Group, 2004.
Schmidt W, Roessling G. Novel manufacturing process of hollow polymer microspheres. Chem Eng Sci. 2006;61:4973–81.
Palmowski M, Huppert J, Ladewig G, Hauff P, Reinhardt M, Mueller M, et al. Molecular profiling of angiogenesis with targeted ultrasound imaging: early assessment of antiangiogenic therapy effects. Mol Cancer Ther. 2008;7:101–9.
Saxena V, Sadoqi M, Kumar S, Shao J. Tiny Bubbles—Multifunctional nanoparticles promise early noninvasive tumor detection and tumor destruction within the body. Nanotechnology. Oemagazine, September 2004. http://spie.org/x16543.xml?ArticleID=x16543.
Maeda H, Wu J, Sawa T, Matsumura Y, Hori K. Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J Control Release. 2000;65:271–84.
Sihorkar V, Vyas SP. Biofilm consortia on biomedical and biological surfaces: delivery and targeting strategies. Pharm Res. 2001;18:1247–54.
Fisher NG, Christiansen J, Leong-Poi H, Jayaweera AR, Lindner JR, Kaul S. Myocardial and microcirculatory kinetics of BR-14, a novel third generation intravenous contrast agent. J Am Coll Cardiol. 2002;39:930–7.
Jin M, Zheng Y, Hu Q. Preparation and characterization of bovine serum albumin alginate/chitosan microspheres for oral administration. Asian J Pharm Sci. 2009;4:215–20.
ACKNOWLEDGMENTS
This research was funded by the Asia-Africa Program, Tohoku University, Goho Life Sciences International Fund, and National Research Foundation of South Africa.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
du Toit, L.C., Govender, T., Pillay, V. et al. Investigating the Effect of Polymeric Approaches on Circulation Time and Physical Properties of Nanobubbles. Pharm Res 28, 494–504 (2011). https://doi.org/10.1007/s11095-010-0247-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-010-0247-y