Abstract
Purpose
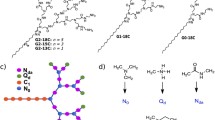
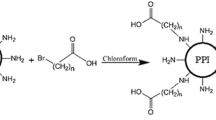
Improved polycation-based non-viral DNA vectors were sought by preparing dendrimers with polyethylenimine cores surrounded by various shells incorporating structural features intended to facilitate steps in transfection mechanisms. Dendrimeric vectors were designed with (a) an outer oligocation shell, intended to facilitate DNA release inside cells, (b) a hydrophobic C-16 alkyl shell, and (c) a polycationic core, the latter two intended to combine lipid-depletion and osmotic burst endosome escape mechanisms, respectively, and were (d) attached through an a acid-cleavable linker reported to hydrolyze at endosomal pH values.
Methods
Vectors and DNA complexes were characterized by dynamic and static light scattering. Flow cytometry was used to quantitate transfection activity and cytotoxicity in CHO–K1 cells.
Results
About 5-fold increased transfection activity was obtained for a vector constructed with an outer shell of oligocations attached through acid-cleavable linkers, relative to a control dendrimer with an acid-stable linker. The most effective oligocation component of outer shells tested was spermine. Neither modification was associated with increased cytotoxicity. This vector design did not permit an evaluation of the benefit of combining endosome release mechanisms.
Conclusion
Using acid-cleavable linkers to attach an outer shell of short, highly-charged oligocations to a PEI-based dendrimeric vector substantially increased transfection efficiency without increasing cytotoxicity.





Similar content being viewed by others
References
Lipinski CA. Drug-like properties and the causes of poor solubility and poor permeability. J Pharmacol Toxicol Methods. 2000;44:235–49.
Lehrman S. Virus treatment questioned after gene therapy death. Nature. 1999;401:517–8.
Volpers C, Kochanek S. Adenoviral vectors for gene transfer and therapy. J Gene Med. 2004;6 Suppl 1:S164–71.
Check E. Regulators split on gene therapy as patient shows signs of cancer. Nature. 2002;419:545–6.
Li Z, Dullmann J, Schiedlmeier B, Schmidt M, von Kalle C, Meyer J, et al. Murine leukemia induced by retroviral gene marking. Science. 2002;296:497.
Merdan T, Kopecek J, Kissel T. Prospects for cationic polymers in gene and oligonucleotide therapy against cancer. Adv Drug Deliv Rev. 2002;54:715–58.
Izsvak Z, Ivics Z, Plasterk RH. Sleeping Beauty, a wide host-range transposon vector for genetic transformation in vertebrates. J Mol Biol. 2000;302:93–102.
Kunath K, von Harpe A, Fischer D, Petersen H, Bickel U, Voigt K, et al. Low-molecular-weight polyethylenimine as a non-viral vector for DNA delivery: comparison of physicochemical properties, transfection efficiency and in vivo distribution with high-molecular-weight polyethylenimine. J Control Release. 2003;89:113–25.
Boussif O, Lezoualc’h F, Zanta MA, Mergny MD, Scherman D, Demeneix B, et al. A versatile vector for gene and oligonucleotide transfer into cells in culture and in vivo: polyethylenimine. Proc Natl Acad Sci U S A. 1995;92:7297–301.
Neu M, Fischer D, Kissel T. Recent advances in rational gene transfer vector design based on poly(ethylene imine) and its derivatives. J Gene Med. 2005;7:992–1009.
Gabrielson NP, Pack DW. Acetylation of polyethylenimine enhances gene delivery via weakened polymer/DNA interactions. Biomacromolecules. 2006;7:2427–35.
Nimesh S, Aggarwal A, Kumar P, Singh Y, Gupta KC, Chandra R. Influence of acyl chain length on transfection mediated by acylated PEI nanoparticles. Int J Pharm. 2007;337:265–74.
Forrest ML, Meister GE, Koerber JT, Pack DW. Partial acetylation of polyethylenimine enhances in vitro gene delivery. Pharm Res. 2004;21:365–71.
Thomas M, Klibanov AM. Enhancing polyethylenimine’s delivery of plasmid DNA into mammalian cells. Proc Natl Acad Sci U S A. 2002;99:14640–5.
Hall HK. Correlation of the base strengths of amines. J Amer Chem Soc. 1957;79:5441–4.
Gosselin MA, Guo W, Lee RJ. Efficient gene transfer using reversibly cross-linked low molecular weight polyethylenimine. Bioconjug Chem. 2001;12:989–94.
Breunig M, Hozsa C, Lungwitz U, Watanabe K, Umeda I, Kato H, et al. Mechanistic investigation of poly(ethylene imine)-based siRNA delivery: disulfide bonds boost intracellular release of the cargo. J Control Release. 2008;130:57–63.
Kloeckner J, Bruzzano S, Ogris M, Wagner E. Gene carriers based on hexanediol diacrylate linked oligoethylenimine: effect of chemical structure of polymer on biological properties. Bioconjug Chem. 2006;17:1339–45.
Zhong Z, Feijen J, Lok MC, Hennink WE, Christensen LV, Yockman JW, et al. Low molecular weight linear polyethylenimine-b-poly(ethylene glycol)-b-polyethylenimine triblock copolymers: synthesis, characterization, and in vitro gene transfer properties. Biomacromolecules. 2005;6:3440–8.
Knorr V, Ogris M, Wagner E. An acid sensitive ketal-based polyethylene glycol-oligoethylenimine copolymer mediates improved transfection efficiency at reduced toxicity. Pharm Res. 2008;25:2937–45.
Shim MS, Kwon YJ. Acid-responsive linear polyethylenimine for efficient, specific, and biocompatible siRNA delivery. Bioconjug Chem. 2009;20:488–99.
Kim S, Choi HS, Jang HS, Suh H, Park J. Hydrophobic modification of polyethylenimine for gene transfectants. Bull Korean Chem Soc. 2001;22:1069–75.
Brownlie A, Uchegbu IF, Schatzlein AG. PEI-based vesicle-polymer hybrid gene delivery system with improved biocompatibility. Int J Pharm. 2004;274:41–52.
Kursa M, Walker GF, Roessler V, Ogris M, Roedl W, Kircheis R, et al. Novel shielded transferrin-polyethylene glycol-polyethylenimine/DNA complexes for systemic tumor-targeted gene transfer. Bioconjug Chem. 2003;14:222–31.
Kircheis R, Schuller S, Brunner S, Ogris M, Heider KH, Zauner W, et al. Polycation-based DNA complexes for tumor-targeted gene delivery in vivo. J Gene Med. 1999;1:111–20.
Tseng WC, Jong CM. Improved stability of polycationic vector by dextran-grafted branched polyethylenimine. Biomacromolecules. 2003;4:1277–84.
Kircheis R, Wightman L, Schreiber A, Robitza B, Rossler V, Kursa M, et al. Polyethylenimine/DNA complexes shielded by transferrin target gene expression to tumors after systemic application. Gene Ther. 2001;8:28–40.
Ogris M, Walker G, Blessing T, Kircheis R, Wolschek M, Wagner E. Tumor-targeted gene therapy: strategies for the preparation of ligand-polyethylene glycol-polyethylenimine/DNA complexes. J Control Release. 2003;91:173–81.
Zhang S, Xu Y, Wang B, Qiao W, Liu D, Li Z. Cationic compounds used in lipoplexes and polyplexes for gene delivery. J Control Release. 2004;100:165–80.
Behr J. The proton sponge: a trick to enter cells viruses did not exploit. Chimia. 1997;51:34–6.
Godbey WT, Wu KK, Mikos AG. Tracking the intracellular path of poly(ethylenimine)/DNA complexes for gene delivery. Proc Natl Acad Sci U S A. 1999;96:5177–81.
Remy-Kristensen A, Clamme JP, Vuilleumier C, Kuhry JG, Mely Y. Role of endocytosis in the transfection of L929 fibroblasts by polyethylenimine/DNA complexes. Biochim Biophys Acta. 2001;1514:21–32.
Srinivasachar K, Neville DM. New protein cross-linking reagents that are cleaved by mild acid. Biochemistry. 1989;28:2501–9.
Ambekar S, Gowda DC. Synthesis of heterobifunctional crosslinking reagents: w-(N-maleimido)alkanoic acid hydrazides. Indian J Chem. 1996;35B:184–6.
Garger SJ, Griffith OM, Grill LK. Rapid purification of plasmid DNA by a single centrifugation in a two-step cesium chloride-ethidium bromide gradient. Biochem Biophys Res Commun. 1983;117:835–42.
Imai K, Toyo’oka T, Watanabe Y. A novel fluorogenic reagent for thiols: ammonium 7-fluorobenzo-2-oxa-1, 3-diazole-4-sulfonate. Anal Biochem. 1983;128:471–3.
Ungaro F, De Rosa G, Miro A, Quaglia F. Spectrophotometric determination of polyethylenimine in the presence of an oligonucleotide for the characterization of controlled release formulations. J Pharm Biomed Anal. 2003;31:143–9.
Wagner HL, Hoeve CAJ. Effect of molecular weight on the refractive increment of polyethylene and n-alkanes. J Polymer Sci A-2. 1971;9:1763–76.
Merdan T. Polyethylenimine and its derivatives: investigation of in vivo fate, subcellular trafficking and development of novel vector systems. Marburg: Philips-Universität Marburg; 2003.
Johnson TW, Klotz IM. Preparation and characterization of some derivatives of poly(ethylenimine). Macromolecules. 1974;7:149–53.
Noeding G, Heitz W. Amphiphilic poly(ethyleneimine) based on long-chain alkyl bromides. Macro Chem Phys. 1998;199:1637–44.
Lin J, Qiu S, Lewis K, Klibanov AM. Bactericidal properties of flat surfaces and nanoparticles derivatized with alkylated polyethylenimines. Biotechnol Prog. 2002;18:1082–6.
Steele TWJ. Oligo-l-lysine-, dextran-, and alkyl-derivatives of polyethylenimine for the development of novel gene transfection vectors. Minneapolis: University of Minnesota; 2006.
Udenfriend S, Stein S, Bohlen P, Dairman W, Leimgruber W, Weigele M. Fluorescamine: a reagent for assay of amino acids, peptides, proteins, and primary amines in the picomole range. Science. 1972;178:871–2.
Weigele M, DeBernardo S, Leimgruber W. Fluorometric assay of secondary amino acids. Biochem Biophys Res Commun. 1973;50:352–6.
Suh J, Paik HJ, Hwang BK. Ionization of polyethylenimine and polyallylamine at various pH’s. Bioorgan Chem. 1994;22:318–27.
Dehshahri A, Oskuee RK, Shier WT, Hatefi A, Ramezani M. Hydrophobized PEI coupled to various oligoamines results in efficient nanocarriers for plasmid DNA transfer. Biomaterials. 2009;30:4187–94.
Oskuee RK, Dehshahri A, Shier WT, Ramezani M. Alkylcarboxylate grafting to polyethylenimine: a simple approach to producing a DNA nano-carrier with low toxicity. J Gene Med. 2009;11:921–32.
Acknowledgements
The authors thank Dr. Robert Vince and University of Minnesota Department of Medicinal Chemistry Developmental Grant in Drug Design #882-1010 for support of this research, and Dr. Mohammad Ramezani for useful discussions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Adapted from the PhD thesis of T.W.J. Steele, University of Minnesota, 2006.
Rights and permissions
About this article
Cite this article
Steele, T.W.J., Shier, W.T. Dendrimeric Alkylated Polyethylenimine Nano-carriers with Acid-Cleavable Outer Cationic Shells Mediate Improved Transfection Efficiency Without Increasing Toxicity. Pharm Res 27, 683–698 (2010). https://doi.org/10.1007/s11095-010-0058-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-010-0058-1