ABSTRACT
Purpose
To evaluate drug-polymer miscibility behavior in four different drug-polymer amorphous solid dispersion systems, namely felodipine-poly(vinyl pyrrolidone) (PVP), nifedipine-PVP, ketoconazole-PVP, and felodipine-poly(acrylic acid) (PAA).
Materials and Methods
Amorphous solid dispersion samples were prepared at different drug-to-polymer ratios and analyzed using differential scanning calorimetry (DSC), mid-infrared (IR) spectroscopy, and powder X-ray diffractometry (PXRD). To help with interpretation of the IR spectra, principal components (PC) analysis was performed. Pair Distribution Functions (PDFs) of the components in the dispersion were determined from the PXRD data, and the pure curves of the components were also extracted from PXRD data using the Pure Curve Resolution Method (PCRM) and compared against experimentally obtained results.
Results
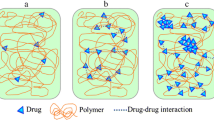
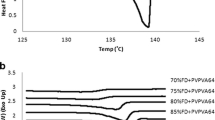
Molecular-level mixing over the complete range of concentration was verified for nifedipine-PVP and felodipine-PVP. For felodipine-PAA, drug-polymer immiscibility was verified for samples containing 30 to 70% polymer, while IR results suggest at least some level of mixing for samples containing 10 and 90% polymer. For ketoconazole-PVP system, partial miscibility is suspected, whereby the presence of one-phase amorphous solid dispersion system could only be unambiguously verified at higher concentrations of polymer.
Conclusions
The three techniques mentioned complement each other in establishing drug-polymer miscibility in amorphous solid dispersion systems. In particular, IR spectroscopy and PXRD are sensitive to changes in local chemical environments and local structure, which makes them especially useful in elucidating the nature of miscibility in binary mixtures when DSC results are inconclusive or variable.












Similar content being viewed by others
References
Hancock BC, Zografi G. Characteristics and Significance of the Amorphous State in Pharmaceutical Systems [Review]. J. Pharm. Sci. 1997;86:1–12.
Hancock BC, Parks M. What is the true solubility advantage for amorphous pharmaceuticals? Pharm Res. 2000;17:397–404.
Leuner C, Dressman J. Improving drug solubility for oral delivery using solid dispersions. Eur J Pharm Biopharm. 2000;50:47–60.
Serajuddin ATM. Solid dispersion of poorly water-soluble drugs: Early promises, subsequent problems, and recent breakthroughs. J. Pharm. Sci. 1999;88:1058–66.
Crowley KJ, Zografi G. The effect of low concentrations of molecularly dispersed poly(vinylpyrrolidone) on indomethacin crystallization from the amorphous state. Pharm Res. 2003;20:1417–22.
Van den Mooter G, Wuyts M, Blaton N, Busson R, Grobet P, Augustijns P, et al. Physical stabilisation of amorphous ketoconazole in solid dispersions with polyvinylpyrrolidone K25. Eur. J. Pharm. Sci. 2001;12:261–9.
Yoshioka M, Hancock BC, Zografi G. Inhibition Of Indomethacin Crystallization In Poly(Vinylpyrrolidone) Coprecipitates. J. Pharm. Sci. 1995;84:983–6.
Yoshihashi Y, Iijima H, Yonemochi E, Terada K. Estimation of physical stability of amorphous solid dispersion using differential scanning calorimetry. J. Therm. Anal. Calorim. 2006;85:689–92.
Marsac PJ, Konno H, Taylor LS. A comparison of the physical stability of amorphous felodipine and nifedipine systems. Pharm Res. 2006;23:2306–16.
Marsac PJ, Konno H, Rumondor ACF, Taylor LS. Recrystallization of nifedipine and felodipine from amorphous molecular level solid dispersions containing poly(vinylpyrrolidone) and sorbed water. Pharm Res. 2008;25:647–56.
Miyazaki T, Yoshioka S, Aso Y, Kojima S. Ability of polyvinylpyrrolidone and polyacrylic acid to inhibit the crystallization of amorphous acetaminophen. J. Pharm. Sci. 2004;93:2710–7.
Bhugra C, Pikal MJ. Role of thermodynamic, molecular, and kinetic factors in crystallization from the amorphous state. J. Pharm. Sci. 2008;97:1329–49.
Matsumoto T, Zografi G. Physical properties of solid molecular dispersions of indomethacin with poly(vinylpyrrolidone) and poly(vinylpyrrolidone-co-vinylacetate) in relation to indomethacin crystallization. Pharm Res. 1999;16:1722–8.
Shamblin SL, Zografi G. The effects of absorbed water on the properties of amorphous mixtures containing sucrose. Pharm Res. 1999;16:1119–24.
Olabisi O, Robeson L, Shaw M. Polymer-polymer Miscibility. San Diego: Academic Press, Inc.; 1979.
Shamblin SL, Taylor LS, Zografi G. Mixing Behavior of Colyophilized Binary Systems. J. Pharm. Sci. 1998;87:694–701.
Pomposo JA, Calahorra E, Eguiazabal I, Cortazar M. Miscibility Behavior of Ternary Poly(Methyl Methacrylate) Poly(Ethyl Methacrylate) Poly(P-Vinylphenol) Blends. Macromolecules. 1993;26:2104–10.
Lu Q, Zografi G. Phase behavior of binary and ternary amorphous mixtures containing indomethacin, citric acid, and PVP. Pharm Res. 1998;15:1202–6.
Bates S, Zografi G, Engers D, Morris K, Crowley K, Newman A. Analysis of amorphous and nanocrystalline solids from their X-ray diffraction patterns. Pharm Res. 2006;23:2333–49.
Newman A, Engers D, Bates S, Ivanisevic I, Kelly RC, Zografi G. Characterization of Amorphous API:Polymer Mixtures Using X-Ray Powder Diffraction. J. Pharm. Sci. 2008;97:4840–56.
Brent RP. Algorithms for Minimization Without Derivatives. Englewood Cliffs, NJ: Prentice Hall; 1973.
I. Ivanisevic, S. Bates, and P. Chen. Novel methods for the assessment of miscibility of amorphous drug-polymer dispersions. J. Pharm. Sci. 2009;98:3373–83.
Konno H, Taylor LS. Influence of different polymers on the crystallization tendency of molecularly dispersed amorphous felodipine. J. Pharm. Sci. 2006;95:2692–705.
Marsac PJ. Molecular Level Understanding of Polymer Induced Formation and Stabilization of Amorphous API, Industrial and Physical Pharmacy. Ph. D. Dissertation: Purdue University, West Lafayette; 2007.
Couchman PR, Karasz FE. Classical thermodynamic discussion of effect of composition on glass-transition temperatures. Macromolecules. 1978;11:117–9.
Rumondor ACF. The Effects of Moisture on Pharmaceutical Amorphous Solid Dispersion Systems, Industrial and Physical Pharmacy. Ph. D. Dissertation: Purdue University, West Lafayette; 2009.
A. C. F. Rumondor, P. Marsac, L. A. Stanford, and L. S. Taylor. Phase Behavior of Poly(vinylpyrrolidone) Containing Amorphous Solid Dispersions in the Presence of Moisture. Mol. Pharm. 2009. doi:10.1021/mp900050c.
Rantanen J, Wikstrom H, Turner R, Taylor LS. Use of in-line near-infrared spectroscopy in combination with chemometrics for improved understanding of pharmaceutical processes. Anal Chem. 2005;77:556–63.
Khougaz K, Clas SD. Crystallization inhibition in solid dispersions of MK-0591 and poly(vinylpyrrolidone) polymers. J. Pharm. Sci. 2000;89:1325–34.
Yalkowsky SH. Solubility and Solubilization in Aqueous Media. New York: Oxford University Press; 1999.
Marcolli C, Luo BP, Peter T. Mixing of the organic aerosol fractions: Liquids as the thermodynamically stable phases. J. Phys. Chem. A. 2004;108:2216–24.
Marsac PJ, Li T, Taylor LS. Estimation of Drug-Polymer Miscibility and Solubility in Amorphous Solid Dispersions Using Experimentally Determined Interaction Parameters. Pharm Res. 2009;26:139–51.
Dong J, Ozaki Y, Nakashima K. Infrared, Raman, and near-infrared spectroscopic evidence for the coexistence of various hydrogen-bond forms in poly(acrylic acid). Macromolecules. 1997;30:1111–7.
Rubinstein M. and Colby. R. H. Polymer Physics: Oxford University Press, New York; 2003.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rumondor, A.C.F., Ivanisevic, I., Bates, S. et al. Evaluation of Drug-Polymer Miscibility in Amorphous Solid Dispersion Systems. Pharm Res 26, 2523–2534 (2009). https://doi.org/10.1007/s11095-009-9970-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-009-9970-7