Abstract
Purpose
Resistance to antiepileptic drugs (AEDs) is the major problem in the treatment of epilepsy. One of the candidate mechanisms of pharmacoresistance is the limitation of AED access to the seizure focus by overexpression of efflux transporters, including P-glycoprotein (Pgp) and multidrug resistance proteins (MRPs). In this respect, it is important to know which AEDs are substrates for such drug transporters in humans.
Methods
In the present study, we used polarized kidney cell lines (LLC, MDCK) transfected with human drug transporters (Pgp, MRP1, MRP2 or MRP5) to evaluate whether the AED topiramate is a substrate for any of these transporters. Known Pgp and MRP substrates were used for comparison.
Results
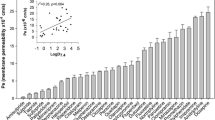
Basolateral-to-apical transport of topiramate, which could be counteracted with the Pgp inhibitor, tariquidar, was determined in Pgp overexpressing LLC cells, whereas topiramate was not transported by any of the MRPs. A comparison with previous experiments in the same transport assay showed that topiramate exhibited the most pronounced Pgp-mediated efflux transport among the AEDS that have been studied as yet.
Conclusions
Thus, these data indicate that brain levels of topiramate may be affected by overexpression of Pgp as determined in patients with intractable epilepsy.



Similar content being viewed by others
REFERENCES
van Passel L, Arif H, Hirsch LJ. Topiramate for the treatment of epilepsy and other nervous system disorders. Expert Rev Neurother. 2006;6:19–31.
Hitiris N, Brodie MJ. Modern antiepileptic drugs: guidelines and beyond. Curr Opin Neurol. 2006;19:175–80.
Schmidt D, Löscher W. Drug resistance in epilepsy: putative neurobiologic and clinical mechanisms. Epilepsia 2005;46:858–77.
Löscher W, Potschka H. Drug resistance in brain diseases and the role of drug efflux transporters. Nat Rev Neurosci. 2005;6:591–602.
Remy S, Beck H. Molecular and cellular mechanisms of pharmacoresistance in epilepsy. Brain 2006;129:18–35.
Löscher W. Animal models of drug-refractory epilepsy. In: Pitkänen A, Schwartzkroin PA, Moshe SL, editors. Models of seizures and epilepsy. San Diego: Elsevier; 2006. p. 551–66.
Potschka H, Volk HA, Löscher W. Pharmacoresistance and expression of multidrug transporter P-glycoprotein in kindled rats. NeuroReport. 2004;19:1657–61.
Kwan P, Brodie MJ. Potential role of drug transporters in the pathogenesis of medically intractable epilepsy. Epilepsia 2005;46:224–35.
Luna-Tortós C, Fedrowitz M, Löscher W. Several major antiepileptic drugs are substrates for human P-glycoprotein. Neuropharmacology 2008;55:1364–75.
Baltes S, Gastens AM, Fedrowitz M, Potschka H, Kaever V, Löscher W. Differences in the transport of the antiepileptic drugs phenytoin, levetiracetam and carbamazepine by human and mouse P-glycoprotein. Neuropharmacology 2007;52:333–46.
Baltes S, Fedrowitz M, Tortos CL, Potschka H, Löscher W. Valproic acid is not a substrate for P-glycoprotein or multidrug resistance proteins 1 and 2 in a number of in vitro and in vivo transport assays. J Pharmacol Exp Ther. 2007;320:331–43.
Rambeck B, Jürgens UH, May TW, Pannek HW, Behne F, Ebner A, et al. Comparison of brain extracellular fluid, brain tissue, cerebrospinal fluid, and serum concentrations of antiepileptic drugs measured intraoperatively in patients with intractable epilepsy. Epilepsia 2006;47:681–94.
Olson DP, Taylor BJ, Ivy SP. Detection of MRP functional activity: calcein AM but not BCECF AM as a Multidrug Resistance-related Protein (MRP1) substrate. Cytometry 2001;46:105–13.
Dogan AL, Legrand O, Faussat AM, Perrot JY, Marie JP. Evaluation and comparison of MRP1 activity with three fluorescent dyes and three modulators in leukemic cell lines. Leuk Res. 2004;28:619–22.
Szakacs G, Jakab K, Antal F, Sarkadi B. Diagnostics of multidrug resistance in cancer. Pathol Oncol Res. 1998;4:251–7.
McAleer MA, Breen MA, White NL, Matthews N. pABC11 (also known as MOAT-C and MRP5), a member of the ABC family of proteins, has anion transporter activity but does not confer multidrug resistance when overexpressed in human embryonic kidney 293 cells. J Biol Chem. 1999;274:23541–8.
Pratt S, Chen V, Perry WI III, Starling JJ, Dantzig AH. Kinetic validation of the use of carboxydichlorofluorescein as a drug surrogate for MRP5-mediated transport. Eur J Pharm Sci. 2006;27:524–32.
Schinkel AH, Wagenaar E, Mol CA, van Deemter L. P-glycoprotein in the blood-brain barrier of mice influences the brain penetration and pharmacological activity of many drugs. J Clin Invest. 1996;97:2517–24.
Yamazaki M, Neway WE, Ohe T, Chen I, Rowe JF, Hochman JH, et al. In vitro substrate identification studies for p-glycoprotein-mediated transport: species difference and predictability of in vivo results. J Pharmacol Exp Ther. 2001;296:723–35.
Hsiao P, Bui T, Ho RJ, Unadkat JD. In vitro-to-in vivo prediction of P-glycoprotein-based drug interactions at the human and rodent blood-brain barrier. Drug Metab Dispos. 2008;36:481–4.
Kühnle M, Egger M, Müller C, Mahringer A, Bernhardt G, Fricker G, et al. Potent and selective inhibitors of breast cancer resistance protein (ABCG2) derived from the p-glycoprotein (ABCB1) modulator tariquidar. J Med Chem. 2009;52:1190–7.
Sills GJ, Kwan P, Butler E, de Lange EC, van den Berg DJ, Brodie MJ. P-glycoprotein-mediated efflux of antiepileptic drugs: preliminary studies in mdr1a knockout mice. Epilepsy Behav. 2002;3:427–32.
Takeuchi T, Yoshitomi S, Higuchi T, Ikemoto K, Niwa S, Ebihara T, et al. Establishment and characterization of the transformants stably-expressing MDR1 derived from various animal species in LLC-PK1. Pharm Res. 2006;23:1460–72.
Kim IW, Booth-Genthe C, Ambudkar SV. Relationship between drugs and functional activity of various mammalian P-glycoproteins (ABCB1). Mini Rev Med Chem. 2008;8:193–200.
Crowe A, Teoh YK. Limited P-glycoprotein mediated efflux for anti-epileptic drugs. J Drug Target. 2006;14:291–300.
Wang Q, Strab R, Kardos P, Ferguson C, Li J, Owen A, et al. Application and limitation of inhibitors in drug-transporter interactions studies. Int J Pharm. 2008;356:12–8.
Lentz KA, Polli JW, Wring SA, Humphreys JE, Polli JE. Influence of passive permeability on apparent P-glycoprotein kinetics. Pharm Res. 2000;17:1456–60.
Santaguida S, Janigro D, Hossain M, Oby E, Rapp E, Cucullo L. Side by side comparison between dynamic versus static models of blood-brain barrier in vitro: a permeability study. Brain Res. 2006;1109:1–13.
Abbott NJ, Ronnback L, Hansson E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat Rev Neurosci. 2006;7:41–53.
Goh LB, Spears KJ, Yao D, Ayrton A, Morgan P, Roland WC, et al. Endogenous drug transporters in in vitro and in vivo models for the prediction of drug disposition in man. Biochem Pharmacol. 2002;64:1569–78.
Borst P, Evers R, Kool M, Wijnholds J. A family of drug transporters: the multidrug resistance- associated proteins. J Natl Cancer Inst. 2000;92:1295–302.
Borst P, de Wolf C, van de Wetering K. Multidrug resistance-associated proteins 3, 4, and 5. Pflugers Arch. 2007;453:661–73.
Dombrowski SM, Desai SY, Marroni M, Cucullo L, Goodrich K, Bingaman W, et al. Overexpression of multiple drug resistance genes in endothelial cells from patients with refractory epilepsy. Epilepsia 2001;42:1501–6.
ACKNOWLEDGEMENTS
We thank Prof. Piet Borst (The Netherlands Cancer Institute) and his group for kindly providing us with the cell lines used in this study. The study was supported by a grant (Lo 274/10-1) from the Deutsche Forschungsgemeinschaft (Bonn, Germany). Carlos Luna-Tortós receives a Ph.D. scholarship from the DAAD (German Academic Exchange Service; Bonn, Germany).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luna-Tortós, C., Rambeck, B., Jürgens, U.H. et al. The Antiepileptic Drug Topiramate is a Substrate for Human P-glycoprotein but Not Multidrug Resistance Proteins. Pharm Res 26, 2464–2470 (2009). https://doi.org/10.1007/s11095-009-9961-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-009-9961-8