Abstract
Purpose
To investigate the influence of mouthpiece geometry on the amount of throat deposition and device retention produced using a dry powder inhaler (Aerolizer®), along with the subsequent effect on the overall inhaler performance.
Materials and Methods
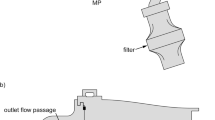
Computational Fluid Dynamics analysis of the flowfield generated in the Aerolizer® with various modified mouthpiece geometries (including cylindrical, conical and oval designs) was used in conjunction with experimental dispersions of mannitol powder using a multi-stage liquid impinger to determine how the overall inhaler performance varied as the mouthpiece geometry was modified.
Results
Geometry of the inhaler mouthpiece had no effect on device retention or the inhaler dispersion performance. In contrast, the mouthpiece geometry strongly affected the amount of throat deposition by controlling the axial component of the exit air flow velocity. The radial motion of the emitted aerosol jet was found to have little effect on throat deposition in representative mouth–throat models. Despite the reduced throat deposition, there was no difference in the overall inhaler performance.
Conclusions
For cases where low throat deposition is a key design parameter, this study demonstrates that the amount of throat deposition can be reduced by making minor modifications to the inhaler mouthpiece design.





Similar content being viewed by others
References
A. R. Clark. Medical aerosol inhalers: past, present, and future. Aerosol Sci. Technol. 22:374–391 (1995).
H.-K. Chan. Inhalation drug delivery devices and emerging technologies. Expert Opin. Ther. Pat. 13:1333–1343 (2003).
C. A. Dunbar, A. J. Hickey, and P. Holzner. Dispersion and characterization of pharmaceutical dry powder aerosols. KONA 16:7–44 (1998).
D. Prime, P. J. Atkins, A. Slater, and B. Sumby. Review of dry powder inhalers. Adv. Drug Deliv. Rev. 26:51–58 (1997).
A. I. Bot, T. E. Tarara, D. J. Smith, S. R. Bot, C. M. Woods, and J. G. Weers. Novel lipid-based hollow-porous microparticles as platform for immunoglobin delivery to the respiratory tract. Pharm. Res. 17:275–283 (2000).
N. Y. K. Chew and H.-K. Chan. Use of solid corrugated particles to enhance powder aerosol performance. Pharm. Res. 18:1570–1577 (2001).
D. A. Edwards, J. Hanes, G. Caponetti, J. Hrkach, A. Ben-Jebria, M. L. Eskew, J. Mintzes, D. Deaver, N. Lotan, and R. Langer. Large porous particles for pulmonary drug delivery. Science 276:1868–1871 (1997).
D. L. French, D. A. Edwards, and R. W. Niven. The influence of formulation on emission, deaggregation and deposition of dry powders for inhalation. J. Aerosol Sci. 27:769–783 (1996).
N. Y. K. Chew and H.-K. Chan. Influence of particle size, air flow, and inhaler device on the dispersion of mannitol powders. Pharm. Res. 16:1098–1103 (1999).
M. Hindle and P. R. Byron. Dose emissions from marketed dry powder inhalers. Int. J. Pharm. 116:169–177 (1995).
H. Steckel and B. W. Müller. In vitro evaluation of dry powder inhalers I: drug deposition of commonly used devices. Int. J. Pharm. 154:19–29 (1997).
L. Borgström, L. Asking, B. Olsson, and L. Thorsson. Throat retention can explain the variability in lung deposition. J. Aerosol Med. 18:99 (2005).
W. H. DeHaan and W. H. Finlay. Predicting extrathoracic deposition from dry powder inhalers. J. Aerosol Sci. 35:309–331 (2004).
B. Grgic, W. H. Finlay, P. K. P. Burnell, and A. F. Heenen. In vitro intersubject and intrasubject deposition measurements in realistic mouth–throat geometries. J. Aerosol Sci. 35:1025–1040 (2004).
W. Stahlhofen, G. Rudolf, and A. C. James. Intercomparison of experimental regional aerosol deposition data. J. Aerosol Med. 2:285–308 (1989).
M. S. Coates, D. F. Fletcher, H.-K. Chan, and J. A. Raper. Effect of design on the performance of a dry powder inhaler using computational fluid dynamics. Part 1: grid structure and mouthpiece length. J. Pharm. Sci. 93:2863–2876 (2004).
B. Grgic, W. H. Finlay, and A. F. Heenen. Regional aerosol deposition and measurements in an idealized mouth and throat. J. Aerosol Sci. 35:21–32 (2004).
F. R. Menter. Two-equation eddy-viscosity models for engineering applications. AIAA J. 32:269–289 (1994).
ANSYS CFX, 2003. http://www.ansys.com/cfx. (accessed 08/01/04).
M. S. Coates, H.-K. Chan, D. F. Fletcher, and J. A. Raper. The role of capsule on the performance of a dry powder inhaler using computational and experimental analyses. Pharm. Res. 22:923–932 (2005).
M. S. Coates, H.-K. Chan, D. F. Fletcher, and J. A. Raper. Influence of air flow on the performance of a dry powder inhaler using computational and experimental analyses. Pharm. Res. 22:1445–1453 (2005).
P. G. Stecher, M. Windholz, and D. S. Leahy. The Merck Index: An encyclopedia of chemicals and drugs. 8th Edition, Merck & Co., Inc., Rahway, 1968.
Y. Zhang, W. H. Finlay, and E. A. Matida. Particle deposition measurements and numerical simulation in a highly idealized mouth–throat. J. Aerosol Sci. 35:789–803 (2004).
A. F. Heenen, W. H. Finlay, B. Grgic, A. Pollard, and P. K. P. Burnell. An investigation of the relationship between the flow field and regional deposition in realistic extra-thoracic airways. J. Aerosol Sci. 35:1013–1023 (2004).
Acknowledgements
This work is funded by a grant from the Australian Research Council. Matthew S. Coates was a recipient of an International Postgraduate Research Scholarship. The authors would like to thank Plastiape S.p.A. for the modification and supply of the inhalers. The authors would also like to acknowledge that the source of the Alberta geometry used in this work was kindly provided from Dr. Finlay’s Aerosol Research Laboratory of Alberta at the University of Alberta, Canada.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Coates, M.S., Chan, HK., Fletcher, D.F. et al. Influence of Mouthpiece Geometry on the Aerosol Delivery Performance of a Dry Powder Inhaler. Pharm Res 24, 1450–1456 (2007). https://doi.org/10.1007/s11095-007-9262-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-007-9262-z