Purpose
The importance of induced crystal disorders like crystallite size, crystal defects, and amorphicity with respect to the dissolution rate of the drug has been discussed in many cases. Thus, the characterization of these properties is of great importance in the pharmaceutical formulation development, although the exact correlation between disorders and dissolution rate is still unclear. The aim of this study was to analyze pharmaceutical tablets with grazing incidence X-ray diffraction, which enables the depth profiling of the crystallographic properties of the tablets. To study and clarify the potential of grazing incidence diffraction in the analysis of pharmaceutical materials, the effect of the compaction process on the surface of tablets was examined.
Methods
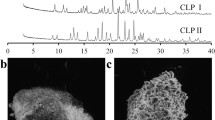
Carbamazepine, tolbutamide, and chlorpropamide tablets, compacted using different compression pressures, were studied using grazing incidence angle X-ray diffraction. The effects of compression on the crystallographic properties were investigated as a function of the distance from the tablet surface.
Results
The surfaces of the tolbutamide and chlorpropamide tablets were disordered due to the compression. The manifestation of the disorder was deduced to be due to amorphicity, small crystallite size, and amount of crystal defects. The changes were mainly on the surface and diminished strongly as a function of the distance from the surface of the tablet. Moreover, the changes were dependent on the compression pressure used. The changes on the surface of the carbamazepine tablets were also due to the compression but these changes were not clearly dependent on the depth nor the compression pressure. The partial phase transition took place in the chlorpropamide tablets due to the compression. The magnitude of the transition was not highest on the surface because amorphization and texturization also took place on the tablet surface during the compression.
Conclusions
The present study proved that grazing incidence X-ray diffraction is a potential novel research tool to reveal crystallographic transformations taking place on the surfaces of the tablets induced, for example, by compression pressure.







Similar content being viewed by others
References
S. Depnath P. Predecki R. Suryanarayanan (2004) ArticleTitleUse of glancing angle X-ray powder diffractometry to depth-profile phase transformations during dissolution of indomethacin and theophylline tablets Pharm. Res. 21 149–159 Occurrence Handle10.1023/B:PHAM.0000012163.89163.f8
A. W. Newman S. R. Byrn (2003) ArticleTitleSolid-state analysis of the active pharmaceutical ingredient in drug products Drug Discov. Today 8 898–905 Occurrence Handle10.1016/S1359-6446(03)02832-0 Occurrence Handle1:CAS:528:DC%2BD3sXnvFSiu7w%3D Occurrence Handle14554018
K. R. Morris U. J. Griesser C. J. Eckhardt J. G. Stowell (2001) ArticleTitleTheoretical approaches to physical transformations of active pharmaceutical ingredients during manufacturing processes Adv. Drug Deliv. Rev. 48 91–114 Occurrence Handle10.1016/S0169-409X(01)00100-4 Occurrence Handle1:CAS:528:DC%2BD3MXivFejsrc%3D Occurrence Handle11325478
H. K. Chan E. Doelker (1985) ArticleTitlePolymorphic transformation of some drugs under compression Drug Dev. Ind. Pharm. 11 135–332
R. Tawashi (1968) ArticleTitleThe dissolution rate of crystalline drugs J. Mond. Pharm. 11 371–379 Occurrence Handle1:CAS:528:DyaF1MXks1Cjsb4%3D
B. A. Brussel Particlevan J. Th. M Hosson ParticleDe (1994) ArticleTitleGlancing angle X-ray diffraction: a different approach Appl. Phys. Lett. 64 1585–1587 Occurrence Handle10.1063/1.111847
M. F. Doerner (1988) ArticleTitleStrain distribution on the aluminum films using X-ray depth profiling J. Appl. Phys. 63 126–131 Occurrence Handle10.1063/1.340503 Occurrence Handle1:CAS:528:DyaL1cXkt1Ciug%3D%3D
Y. Fujii E. Yanase K. Arai (2005) ArticleTitleDepth profiling of the strain distribution in the surface layer using X-ray diffraction at small glancing angle of incidence Appl. Surf. Sci. 244 230–234 Occurrence Handle10.1016/j.apsusc.2004.09.166 Occurrence Handle1:CAS:528:DC%2BD2MXivVKlsbg%3D
N. S. Murthy C. Bednarczyk H. Minor (2000) ArticleTitleDepth-profiles of structure in single- and multilayered commercial polymer films using grazing-incidence X-ray diffraction Polymer 41 277–284 Occurrence Handle10.1016/S0032-3861(99)00137-8 Occurrence Handle1:CAS:528:DyaK1MXntlOltbo%3D
K. Vamsi Krishna V. Dutta (2004) ArticleTitleDepth profiling study of in situ CdCl2 treated CdTe/CdS heterostructure with glancing angle incidence X-ray diffraction Thin Solid Films 450 255–260 Occurrence Handle10.1016/j.tsf.2003.11.110
L. G. Parrat (1954) ArticleTitleSurface studies of solids by total reflection of X-rays Phys. Rev. 95 359–369 Occurrence Handle10.1103/PhysRev.95.359
M. Shibata H. Kokubo K. Morimoto K. Morisaka T. Ishida M. Inoue (1983) ArticleTitleX-ray structural studies and physicochemical properties of cimetidine polymorphism J. Pharm. Sci. 72 1436–1442 Occurrence Handle1:CAS:528:DyaL2cXptFaisQ%3D%3D Occurrence Handle6663481
P. L. D. Wildfong N. A. Morley M. D. Moore K. R. Morris (2005) ArticleTitleQuantitative determination of polymorphic composition in intact compacts by parallel-beam X-ray powder diffractometry: II. Data correction for analysis of phase transformations as a function of pressure J. Pharm. Biomed. Anal. 39 1–7 Occurrence Handle10.1016/j.jpba.2005.03.003 Occurrence Handle1:CAS:528:DC%2BD2MXntVGisLw%3D Occurrence Handle15869857
C. Lefebvre A. M. Guyot-Hermann M. Draguet-Brughmans R. Bouché J. C. Guyot (1986) ArticleTitlePolymorphic transitions of carbamazepine during grinding and compression Drug Dev. Ind. Pharm. 12 1913–1927 Occurrence Handle1:CAS:528:DyaL2sXpt1ym
T. Matsumoto N. Kaneniwa S. Higuchi M. Otsuka (1991) ArticleTitleEffects of temperature and pressure during compression on polymorphic transformation and crushing strength of chlorpropamide tablets J. Pharm. Pharmacol. 43 74–78 Occurrence Handle1:CAS:528:DyaK3MXht1yqtLY%3D Occurrence Handle1672905
K. Kimura F. Hirayama K. Uekama (1999) ArticleTitleCharacterization of tolbutamide polymorphs (Burger's Forms II and IV) and polymorphic transition behavior J. Pharm. Sci. 88 385–391 Occurrence Handle10.1021/js980376z Occurrence Handle1:CAS:528:DyaK1MXhs1Wmu7g%3D Occurrence Handle10187747
T. Roisnel J. Rodríguez-Carvajal (2000) ArticleTitleWinPLOTR: a Windows tool for powder diffraction patterns analysis Mater. Sci. Forum, Proceedings of the EPDIC 7 118–123
F. H. Allen (2002) ArticleTitleThe Cambridge structural database: a quarter of a million crystal structures and rising Acta Crystallogr. B58 380–388 Occurrence Handle1:CAS:528:DC%2BD38XktVOqu74%3D
B. Hancock M. Parks (2000) ArticleTitleWhat is the true solubility advantage for amorphous pharmaceuticals? Pharm. Res. 17 397–404 Occurrence Handle10.1023/A:1007516718048 Occurrence Handle1:CAS:528:DC%2BD3cXktFalsb0%3D Occurrence Handle10870982
B. Hancock G. Zografi (1997) ArticleTitleCharacteristics and significance of the amorphous state in pharmaceutical systems J. Pharm. Sci. 86 1–12 Occurrence Handle10.1021/js9601896 Occurrence Handle1:CAS:528:DyaK28XntlOqt7k%3D Occurrence Handle9002452
H. Burt A. Mitchell (1981) ArticleTitleCrystal defects and dissolution Int. J. Pharm. 9 137–152 Occurrence Handle10.1016/0378-5173(81)90007-7 Occurrence Handle1:CAS:528:DyaL3MXlsFKnurY%3D
V. Levich (1962) Physicochemical Hydrodynamics Prentice Hall New York
M. Nicklasson A-B. Magnusson (1985) ArticleTitleProgram for evaluating drug dissolution kinetics in preformulation Pharm. Res. 2 263–266 Occurrence Handle10.1023/A:1016333432224
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koivisto, M., Heinänen, P., Tanninen, V.P. et al. Depth Profiling of Compression-Induced Disorders and Polymorphic Transition on Tablet Surfaces with Grazing Incidence X-ray Diffraction. Pharm Res 23, 813–820 (2006). https://doi.org/10.1007/s11095-006-9785-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-006-9785-8