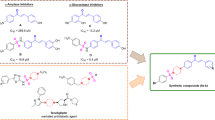
This paper is focused on the in silico and in vitro determination of MAO-B inhibitory activity on recombinant MAO-B enzyme (hMAOB) of newly synthesized substituted N-pyrrolyl hydrazide hydrazones. From the tested substances, only compound DI 5a has shown statistically significant inhibitory activity on human recombinant MAO-B enzyme, comparable with that of selegiline and better than that of melatonin, applied as standards. The molecular docking studies indicated formation of a stable complex in the aromatic “cage” of the enzyme and the 2-hydroxyphenyl moiety of DI 5a. An additional stable water-mediated hydrogen bond is formed with the carbonyl group located at third position in the pyrrole ring.



Similar content being viewed by others
References
P. Duarte, A. Cuadrado and R. León, React. Oxyg. Species, 229 – 259 (2020).
J. P. Johnston, Biochem. Pharmacol., 17, 1285 – 1297 (1968).
M. B. Youdim, W. Maruyamaand and M. Naoi, Drugs Today, 41(6), 369 – 391 (2005).
M. B. H. Youdim, G. G. S. Collins, M. Sandler, et al., Nature, 236, 225 – 228 (1972).
M. B. Youdim, J. Neural Transm., 125(11), 1719 – 1733 (2018).
P. O. Patil and S. B. Bari, Arab. J. Chem., 7(6), 857 – 884, (2014).
A. C. Tripathi, S. Upadhyay, S. Paliwal and S. K. Saraf, Eur. J. Med. Chem., 145, 445 – 497 (2018).
M. O. Ogunrombi, S. F. Malan, G. Terre’Blanche, et al., Bioorg. Med. Chem., 16(5), 2463 – 2472 (2008).
D. G. Tzankova, S. P. Vladimirova, D. S. Aluani, et al., Acta Pharm., 70(3), 303 – 324 (2020).
O. M. Bautista-Aguilera, G. Esteban, I. Bolea, et al., Eur. J. Med. Chem., 75, 82 – 95 (2014).
E. Mateev, I. Valkova, M. Georgieva and A. Zlatkov, Int. J. Pharm. Pharm. Sci., 13(8), 32 – 35 (2021).
T. A. Halgren, R. B. Murphy, R. A. Friesner, et al., J. Med. Chem., 47(7), 1750 – 1759 (2004).
M. B. Youdim, D. Edmondson and K. F. Tipton, Nat. Rev. Neurosci., 7(4), 295 – 309 (2006).
G. G. S. Collins, M. Sandler, E. D. Williams and M. B. H. Youdim, Nature, 225(5235), 817 – 820 (1970).
J. P. Petzer, Jr. N. Castagnoli, M. A. Schwarzschild, et al., Neurotherapeutics, 6(1), 41 – 151 (2009).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tzankova, D., Mateev, E., Vladimirova, S. et al. IN SILICO and IN VITRO Estimation of MAO-B Inhibitory Activity of Newly Synthesized Hydrazones Bearing a Pyrrole Heterocyclic System. Pharm Chem J 57, 1444–1448 (2023). https://doi.org/10.1007/s11094-023-03008-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-023-03008-7