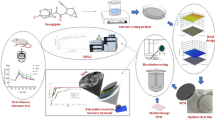
An efficient, perceptive, sensitive, accurate and economical RP-UPLC method with tunable UV detector has been developed for the simultaneous estimation of telmisartan and azelnidipine in bulk powders and their combined film-coated bilayer tablet dosage form. The proposed method employs Agilent Zorbax Stable Bond (SB) packing C8 (100 mm × 2.1 mm, 2 μm) column, a mobile phase consisting of 0.01 N potassium dihydrogen orthophosphate (pH 4.8) and acetonitrile in 70:30 v/v ratio pumped at a flow rate of 0.3 mL/min, and the UV detector tuned to 257 nm wavelength, which reliably separates all analytes with good resolution. Telmisartan and azelnidipine were eluted from the column at 0.675 and 1.601 min, respectively. Linearity of the telmisartan and azelnidipine determination was observed in the range of 20 – 120 μg/mL and 2 – 12 μg/mL, respectively. Computed values of the validation parameters confidently show that the method is specific, accurate and precise with high sensitivity. Exploration of the analytes under various forced degradation conditions with the help of the proposed method confirms stability-indicating character of the method. The developed method has high efficiency in separating telmisartan and azelnidipine and ensures their stability-indicating assay with good sensitivity and specificity, thus being adapted to the pharmaceutical industry applications.




Similar content being viewed by others
References
P. Gosse, Vasc. Health Risk Manag., 2, 195 – 201 (2006).
W. Wienen, M. Entzeroth, J. C. A. V. Meel, et al., Cardiovasc. Drug Rev., 18, 127 – 154 (2000).
S. Nandi, S. Nag, T. Ash, et al., Med. Res. Chronicles, 7, 366 – 370 (2020).
G. Guido, Q. T. Fosca, and M. Giuseppe, J. Renin Angiotensin Aldosterone Syst., 9, 66 – 74 (2008).
K. Shimada, K. Miyauchi, and H. Daida, Expert Rev. Cardiovasc. Ther., 13, 23 – 31 (2015).
M. Watanabe, T. Hirano, S. Okamoto, et al., Hypertens. Res., 33, 43 – 48 (2010).
V. U. Shewale, S. S. Aher, and R. B. Saudagar, J. Drug Deliv. Ther., 9, 1002 – 1005 (2019).
B. Upendra, H. Dhaked, and A. K. Danodia, Int. J. Drug Regul. Aff., 1, 61 – 64 (2013).
P. Ashok, S. T. Narenderan, S. N. Meyyanathan, et al., Int. J. Appl. Pharm., 237 – 240 (2019).
V. P. Kurade, M. G. Pai, and R. Gude, Indian J. Pharm. Sci., 71, 148 – 151 (2009).
S. Shaina, S. Varinder, R. Sandeep, et al., J. Pharm. Technol. Res. Manag., 4, 63 – 79 (2016).
N. Doshi, A. Sheth, A. Sharma, et al., J. Chem. Pharm. Res., 2, 252 – 263 (2010).
V. U. Barge, R. B. Gaikwad, F. M. Chaudhari, et al., Int. J. Pharm. Clin. Res., 10, 219 – 223 (2018).
A. R. Chabukswar, S. C. Jagdale, S. V. Kumbhar, et al., Res. J. Pharm. Technol., 3, 1227 – 1230 (2010).
M. L. Surekha, G. K. Swamy, and G. L. Ashwini, Int. J. Drug Dev. Res., 4, 200 – 205 (2012).
S. V. Prasad, G. P. Satish, and V. M. Kiran, J. Anal. Bioanal. Tech., 03 (2012).
A. K. Palakurthi, T. Dongala, and L. N. R. Katakam, Pract. Lab. Med., 21 (2020).
S. Nalwade, V. Ranga Reddy, D. Durga Rao, et al., Sci. Pharm., 79, 69 – 84 (2011).
V. B. Ravi, J. K. Inamadugu, N. R. Pilli, et al., J. Pharm. Anal., 2, 319 – 326 (2012).
P. Sengupta, B. Chatterjee, U. K. Mandal, et al., J. Pharm. Anal., 7, 381 – 387 (2017).
K. D. Raskapur, M. P. Mrunali, and D. C. Anandkumari, Int. J. Pharm. Pharm. Sci., 4, 238 – 240 (2011).
S. Muralidharan, S. Parasuraman, and V. Venugopal, J. Pharm. Belg., 1, 43 – 45 (2015).
D. Prabhakar, J. Sreekanth, and K. N. Jayaveera, Int. J. Chemtech Res., 11,7 – 12 (2018).
K. P. Jayvadan and K. P. Nilam, Sci. Pharm., 82, 541 – 554 (2014).
K. Manish, Umesh Chandra, G. Pankaj, et al., Int. J. Pharm. Sci. Drug Res., 13 (2021).
K. Kishore and K. Somasekhar Reddy, Bull. Env. Pharmacol. Life Sci., 10, 19 – 27 (2021).
International Council for Harmonisation (ICH). Quality Guidelines ICH. https://www.ich.org/page/quality-guidelines (accessed January 18, 2021).
Analytical Procedures and Methods Validation for Drugs and Biologics, Analytical Procedures: Guidance for Industry, U. S. Department of Health and Human Services Food and Drug Administration (2015).
M. Blessy, R. D. Patel, P. N. Prajapati, et al., J. Pharm. Anal., 4, 159 – 165 (2014).
K. K. Otha, S. P. K. Reddy, V. K. Raju, et al., Int. Res. J. Pharm., 4, 78 – 85 (2013).
G. Ngwa, Drug Deliv. Technol., 10, 56 – 59 (2010).
British Pharmacopoeia (BP) 2016, https://www.pharmacopoeia.com/the-british-pharmacopoeia (accessed January 27, 2021).
Acknowledgments
The authors are thankful to the Department of Pharmaceutical Analysis, Sri Padmavati Mahila Visvavidyalayam (Tirupati Andhrapradesh, India) for encouragement.
Competing Interest
The authors declare that they have no conflicts of interest.
Authors’ Contributions
All authors contributed equally in design of the work and corresponding author played vital role in manuscript preparation. All authors have read and approved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Swetha, A., Ramya, K.B. New Stability Indicating RP-UPLC Method for Simultaneous Estimation of Telmisartan and Azelnidipine in Bulk and Combined Bilayer Film-Coated Tablet Dosage Form. Pharm Chem J 56, 1544–1551 (2023). https://doi.org/10.1007/s11094-023-02826-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-023-02826-z