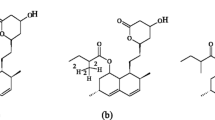
The present work has been done as part of optimization procedures aimed at developing a sensitive HPLC-MS method with good precision and accuracy for the quantification of atorvastatin and rosuvastatin in pharmaceutical and clinical samples. Amixture of methanol and water in a 70:30 ratio by volume was selected as the mobile phase and its pH was set at 3. The oven temperature was 25°C and the sample injection volume was set at 1 μL. ESI-MS interface was operated in the positive ionization mode at block temp 400°C, interface temp 350°C, DL temp 250, CID gas Ar 270 kpa, Neb gas N2 flow at 2.0 L/min. drying gas N2 flow at 10 L/min, heating gas Zero Air flow at 10.0 L/min. The analytical method validation was carried out as prescribed by the USP and ICH guidelines to provide reproducible results. Pharmacokinetics of atorvastatin from conventional (40 mg) tablets was evaluated in patients aged between 24 and 32 years. Results showed that the proposed method was rapid, both drugs were separated within 10 min. Linearity was evaluated at 0 – 100 ng/mL and showed the response to be linear for both drugs. Thus, the method proved to be rapid, specific, precise, and sensitive for simultaneous analysis of atorvastatin and rosuvastatin in biological and pharmaceutical samples.




Similar content being viewed by others
References
A. G. Olsson, F. McTaggart, and A. Raza, Cardiovasc. Drug Rev., 20(4), 303 – 328 (2002).
E. S. Istvan and J. Deisenhofer, Science, 292 (5519), 1160 – 1164, (2001).
F. P. Gomes, P. L. García, J. M. Porto Alves, et al., Anal. Lett., 42(12), 1784 – 1804, (2009).
T. R. Kumar, N. R. Shitut, P. K. Kumar, et al., Biomed. Chromatogr., 20(9), 881 – 887, (2006).
S. Vittal, N. R. Shitut, T. R. Kumar, et al., Biomed. Chromatogr., 20(11), 1252 – 1259, (2006).
Y. Shah, Z. Iqbal, Z. L. Ahmad, et al., J. Chromatogr. B, 879(9), 557 – 563, (2011).
C. K. Hull, A. D. Penman, C. K. Smith, et al., J. Chromatogr. B, 772(2), 219 – 228, (2002).
S. S. Singh, K. Sharma, H. Patel, et al., J. Braz. Chem. Soc., 16(5), 944 – 950, (2005).
K. A. Oudhoff, T. Sangster, E. Thomas, et al., J. Chromatogr. B, 832(2), 191 – 196, (2006).
D. H. Xu, Z R. Ruan, Q. Zhou, et al., Mass Spectrom., 20(16), 2369 – 2375, (2006).
K. Lan, X. Jiang, Y. Li, et al., J. Pharm. Biomed. Anal., 44(2), 540 – 546, (2007).
J. Gao, D. Zhong, X. Duan, et al., J. Chromatogr. B, 856(1–2), 35 – 40, (2007).
R. K. Trivedi, R. R. Kallem, R. Mullangi, et al., J. Pharm. Biomed. Anal., 39(3–4), 661 – 669, (2005).
C. K. Hull, P. D. Martin, M. J. Warwick, et al., J. Pharm. Biomed. Anal., 35(3), 609 – 614, (2004).
H. Lee, C. Ho, M. Hu, et al., Biomed. Chromatogr., 27(11), 1369 – 1374, (2013).
J. S. Macwan, I. A. Ionita, and F. Akhlaghi, Anal. Bioanal. Chem., 402(3), 1 – 11, (2012).
ICH Guidelines Q2 (R1), Validation of Analytical Procedures: Text and Methodology, CPMP / ICH / 381 / 95, European Medicines Agency: London (1995).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hussain, A., Zakria, M., Tariq, S.A. et al. Simultaneous Analysis of Atorvastatin and Rosuvastatin by LC-MS: Development, Validation, and Application of the Proposed Method to Analysis of Atorvastatin Pharmacokinetics in Pakistani Population. Pharm Chem J 56, 1149–1156 (2022). https://doi.org/10.1007/s11094-022-02766-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-022-02766-0