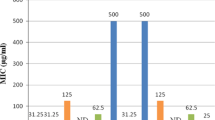
Twelve novel1, 2-diaminopropane side chain modified 4-aminoquinoline Mannich bases were synthesized and characterized by a number of analytical and spectroscopic techniques. The molecules were subsequently screened for in-vitro antimalarial activity against chloroquine sensitive strain of Plasmodium falciparum (3D7). On antimalarial activity screening, all target compounds (9a–9l) showed MIC between 15.6 – 125 μg/mL. Two compounds among all, namely 7-chloro-N-(1-(3-(dibenzylamino)-1-p-tolylpropylamino) propan-2-yl)quinolin-4-amine 9b (MIC = 15.6 μg/mL or 2.590 ± 0.04 μM) and 7-chloro-N-(1-(3- (dibenzylamino)-1-(4-methoxyphenyl)propylamino)propan-2-yl)quinolin-4-amine 9c (MIC = 15.6 μg/mL or 2.398 ± 0.04 μM) were found moderately potent against chloroquine sensitive strain of P. falciparum (3D7) compared to chloroquine (MIC = 0.4 μg/mL or 0.106 ± 0.01μM).
Similar content being viewed by others
References
J. D. Martinez, M. T. Parker, K. E. Fultz, et al., in: Burger’s Medicinal Chemistry and Drug Discovery, John Wiley & Sons, New York (2003), Vol. 5, pp. 920 – 931.
N. White, Plasmodium knowlesi: the Fifth Human Malaria Parasite, University of Chicago Press (2008).
A. V. Pandey, H. Bisht, V. K. Babbarwal, et al., Biochem. J., 355(2), 333 – 338 (2001).
A. C. Chou, R. Chevli, and C. D. Fitch, Biochemistry, 19(8), 1543 – 1549 (1980).
T. J. Egan and H. M. Marques, Coord. Chem. Rev., 190, 493 – 517 (1999).
A. Dorn, R. Stoffel, H. Matile, et al., Nature, 374(6519), 269 – 271 (1995).
D. J. Sullivan, I. Y. Gluzman, D. G. Russell, and D. E. Goldberg, Proc. Natl. Acad. Sci., 93(21), 11865 – 11870 (1996).
T. J. Egan, Targets, 2(3), 115 – 124 (2003).
T. J. Egan, R. Hunter, C. H. Kaschula, et al., J. Med. Chem., 43(2), 283 – 291 (2000).
C. H. Kaschula, T. J. Egan, R. Hunter, et al., J. Med. Chem., 45(16), 3531 – 3539 (2002).
R. G. Ridley, W. Hofheinz, H. Matile, et al., Antimicrob. Agents Chemother., 40(8), 1846 – 1854 (1996).
P. B. Madrid, N. T. Wilson, J. L. DeRisi, et al., J. Comb. Chem., 6(3), 437 – 442 (2004).
A. Sparatore, N. Basilico, S. Parapini, et al., Bioorg. Med. Chem., 13(18), 5338 – 5345 (2005).
M. Freitag, M. Kaiser, T. Larsen, et al., Bioorg. Med. Chem., 15(7), 2782 – 2788 (2007).
A. Ryckebusch, M.-A. Debreu-Fontaine, E. Mouray, et al., Bioorg. Med. Chem. Lett., 15(2), 297 – 302 (2005).
C. C. Musonda, S. Little, V. Yardley, et al., Bioorg. Med. Chem. Lett., 17(17), 4733 – 4736 (2007).
D. De, F. M. Krogstad, L. D. Byers, et al., J. Med. Chem., 41(25), 4918 – 4926 (1998).
P. A. Stocks, K. J. Raynes, P. G. Bray, et al., J. Med. Chem., 45(23), 4975 – 4983 (2002).
P. M. O’Neill, D. J. Willock, S. R. Hawley, et al., J. Med. Chem., 40(4), 437 – 448 (1997).
D. W. Mathieson, Interpretation of Organic Spectra, Academic Press, New York (1965).
R. M. Silverstein and G. C. Bassler, J. Chem. Educ., 39(11), 546 (1962).
B. Furniss, A. Hannaford, P. Smith, et al. Vogel’s Textbook of Practical Organic Chemistry, ELBS Longman, London (1989).
N. I. Wenzel, N. Chavain, Y. Wang, et al., J. Med. Chem., 53(8), 3214 – 3226 (2010).
L. M. Antinarelli, A. M. Carmo, F. R. Pavan, et al., Bioorg. Med. Chem. Lett., 2(1), 16 (2012).
A. Kumar, K. Srivastava, S. R. Kumar, et al., Eur. J. Med. Chem., 46(2), 676 – 690 (2011).
K. Rieckmann, G. Campbell, L. Sax, et al., Lancet, 311(8054), 22 – 23 (1978).
S. Puri and N. Singh, Exp. Parasitol., 94(1), 8 – 14 (2000).
B. S. Kalra, S. Chawla, P. Gupta, et al., Indian J. Pharmacol., 38(1), 5 (2006).
W. Trager and J. B. Jensen, Science, 193(4254), 673 – 675 (1976).
C. Lambros and J. P. Vanderberg, J. Parasitol., 65(3), 418 – 420 (1979).
Acknowledgments
The authors are thankful to Dr. C. R. Pillai, Emeritus Scientist and Dr. Anup Anvikar, Director, National Institute of Malaria Research (Indian Council of Medical Research), New Delhi for providing antimalarial screening facilities and training. The authors also are thankful to S. A. I. F., Punjab University, Chandigarh, India for providing spectroscopic data.
CONFLICT OF INTEREST
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, B., Chetia, D. & Kumawat, M.K. Synthesis and In Vitro Antimalarial Activity Evaluation of Some New 1,2-Diaminopropane Side-Chain-Modified 4-Aminoquinoline Mannich Bases. Pharm Chem J 55, 724–731 (2021). https://doi.org/10.1007/s11094-021-02484-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-021-02484-z