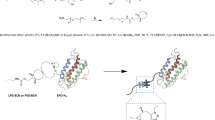
A conjugate of erythropoietin β with 30 kDa linear methoxy poly(ethylene glycol) characterized by the presence of an azo spacer between the protein fragment and poly(ethylene glycol) was developed for import-substitution of Mircera drug in the Russian Federation. The erythropoietic activity of the synthesized conjugate was determined. Its pharmacokinetics in CBA mice were found to be comparable to those of Mircera. The bioequivalence of the proposed azo conjugate and Mircera was established based on the calculated drug pharmacokinetic parameters in mice.




Similar content being viewed by others
References
European Pharmacopoeia, 8.0 Ed. (2014).
V. Yu. Shilo, Nefrol. Dial., 3 – 4, 192 – 198 (2008).
M. Buemi, L. Nostro, and A. Romeo, Cardiovasc. Hematol. Agents Med. Chem., 4(4), 299 (2006).
L. T. Goodnough, B. Skikne, and C. Brugnara, Blood, 96, 823 (2000).
Z. Cai, D. J. Manalo, G.Wei, et al., Circulation, 108, 79 (2003).
V. M. Ermolenko and A. Yu. Nikolaev, Ter. Arkh., 62(11), 141 (1990).
A. J. Erslev, J. Caro, E. Kansu, and R. Silver, Br. J. Haematol., 45(1), 65 (1980).
A. W. Flake, M. R. Harrison, N. S. Adzick, and E. D. Zanjani, Blood, 70(2), 542 (1987).
G. Weiss and L. T. Goodnough, N. Engl. J. Med., 352(10), 1011 (2005).
J. P. Kaltwasser, U. Kessler, R. Gottschalk, et al., J. Rheumatol., 28, 2430 (2001).
R. T. Means, Curr. Hematol. Rep., 2, 116 (2003).
S. T. Murphy and P. S. Parfrey, Semin. Nephrol., 20, 350 (2000).
A. D. Pavlov and E. F. Morshakova, Regulation of Erythropoiesis: Physiological and Clinical Aspects [in Russian], Meditsina, Moscow (1987), p. 272.
A. E. G. Raine (ed.), Advanced Renal Medicine, Oxford University Press, Oxford (1992), p. 484.
L. T. Goodnough, T. G. Monk, and C. L. Andriole, N. Engl. J. Med., 336(13), 933 (1997).
V. Yu. Shilo and N. N. Khasabov, Lechashchii Vrach, 1, 25 (2008).
A. J. Collins, R. M. Brenner, J. J. Ofman, et al., Am. J. Kidney Dis., 46(3), 481 (2005).
A. L. De Francisco,W. Sulowicz, M. Klinger, et al., Int. J. Clin. Pract., 60(12), 1687 (2006).
D. T. Gilbertson, J. P. Ebben, R. N. Foley, et al., Clin. J. Am. Soc. Nephrol., 3(1), 133 (2008).
R. E. Smith, I. A. Jaiyesimi, L. A. Meza, et al., Br. J. Cancer, 84, s1, 24 (2001).
B. Canaud, G. Mingardi, J. Braun, et al., Nephrol. Dial. Transplant., 23(11), 3654 (2008).
J. Burg, A. Engel, et al., Int. Pat. WO2002049673; PCT Int. Appl., Jun. 27, 2002.
J. Burg, B. Hilger, and H.-P. Josel, Int. Pat. WO2001002017; PCT Int. Appl., Jan. 11, 2001.
T. Nakamura, Y. Sekimori, et al., US Pat. Appl. 20060276634; PCT Int. Appl., Dec. 7, 2006.
F. M. Veronese, PEGylated Protein Drugs: Basic Science and Clinical Applications, Birkhauser Verlag, Basel (2009).
I. G. Nikitin, I. E. Baikova, and L. M. Gogova, Lech. Delo, 4, 18 (2005).
V. Shilo, Vrach, 2009(6), 39 (2009).
H. G. Higgins and D. Fraser, Aust. J. Sci. Res., 5, 736 (1952).
H. Pauly, Z. Physiol. Chem., 94, 284 (1915).
D. V. Lonshakov, S. V. Sheremet’ev, E. M. Belosludtseva, et al., RSC Adv., 5, 42903 (2015).
S. V. Sheremet’ev, S. A. Korovkin, et al., RU Pat. 2,439,091, Jan. 10, 2012.
S. V. Sheremet’ev, V. V. Zverev, et al., RU Pat. 2,433,134, Nov. 10, 2011.
S. V. Sheremet’ev, S. A. Korovkin, et al., Eurasian Pat. 018116, May 30, 2013.
Handbook for Experimental (Preclinical) Study of New Drugs [in Russian], 2nd Ed., Revised and Suppl., Meditsina, Moscow (2005).
Guideline on bioanalytical method validation, EMEA/CHMP/EWP/192217/2009, Rev. 1 Corr. 2, Committee for Medicinal Products for Human Use, European Medicines Agency (2015).
S. E. Rosenbaum, Basic Pharmacokinetics and Pharmacodynamics: An Integrated Textbook and Computer Simulations, John Wiley and Sons, Hoboken, New Jersey (2011).
F. M. Veronese, Biomaterials, 22, 405 (2001).
R. F. Finn and N. R. Siegel, Int. Pat. WO2006024953, Mar. 9, 2006.
M. Reslow, Int. Pat. WO2007025988, Mar. 8, 2007.
C. O. Beauchamp, S. L. Gonias, D. P. Menapace, and S. V. Pizzo, Anal. Biochem., 131, 25 (1983).
J. P. Balthasar, Am. J. Pharm. Educ., 63, 194 (1999).
Handbook for Drug Review [in Russian], Vol. 1, IV, Grif i K, Moscow (2013).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 55, No. 7, pp. 30 – 37, July, 2021.
Rights and permissions
About this article
Cite this article
Sheremet’ev, S.V., Lonshakov, D.V., Belosludtseva, E.M. et al. Synthesis and Pharmacological Properties of the Azo-Conjugate of Erythropoietin β with Poly(Ethylene Glycol). Pharm Chem J 55, 698–705 (2021). https://doi.org/10.1007/s11094-021-02480-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-021-02480-3