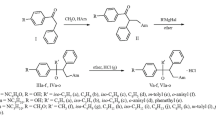
Tertiary piperazine aminoalcohols were synthesized by reacting 1-(4-butoxyphenyl)-3-(4-substituted piperazin-1-yl)-2-phenylpropan-1-ones with alkyl(aryl)magnesium halides. Some of the final compounds were converted into dihydrochlorides. The influence of the synthesized compounds on C-180 mouse tumor DNA methylation was studied in vitro by incubating drug solutions (3 × 10–6 M) with tumor homogenates at 37°C (thermostatted) for 24 h. Rather high activity (51.6% inhibition of DNA methylation) was observed for 1-(4-butoxyphenyl)-3-[4-(4-fluoropheny)piperazin-1-yl]-1,2-diphenylpropan-1-ol containing a 1-phenyl radical. Introduction of a methoxy group into the ortho-position of the phenyl radical decreased the activity to 37.5%.
Similar content being viewed by others
References
E. A. Tatarkova, Vestn. AGU, 1, No. 133, 74 – 79 (2014).
P. M. Das and R. Singal, J. Clin. Oncol., 22(22), 4632 – 4642 (2004).
A. U. Isakhanyan, G. A. Gevorgyan, S. G. Chshmarityan, et al., Zh. Org. Khim., 52(4), 608 – 611 (2016).
G. A. Gevorgyan, A. U. Isakhanyan, N. K. Gasparyan, et al., “Data on the Pharmacological Properties of Tertiary Arylaliphatic Aminopropanols,” in: Progress in Organic and Pharmaceutical Chemistry [in Russian], Erevan (2012), pp. 87 – 99.
N. Z. Akopyan, O. A. Papoyan, G. A. Gevorgyan, and G. A. Panosyan, Zh. Org. Khim., 83(11), 1873 – 1877 (2013).
B. F. Vanyushin, A. N. Masin, V. R. Vasiliev, et al., Biochim. Biophys. Acta, 299, 397 (1973).
Author information
Authors and Affiliations
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 55, No. 2, pp. 29 – 32, February, 2021.
Rights and permissions
About this article
Cite this article
Gevorgyan, G.A., Hakobyan, N.Z., Agababyan, A.G. et al. Synthesis of Tertiary Piperazine Aminoalcohols and their Dihydrochlorides and their Influence on DNA Methylation Processes In Vitro. Pharm Chem J 55, 138–141 (2021). https://doi.org/10.1007/s11094-021-02391-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-021-02391-3