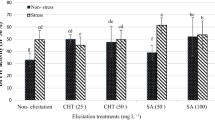
Saffron possesses important medicinal properties, all of which are due to the secondary metabolites such as crocin, picrocrocin, and safranal. This study aims at investigating biosynthesis of the secondary metabolites during growth of saffron callus cells in a suspension culture, and examining the expression of genes controlling the metabolites. Sterilized saffron corms were cultured in a MS½ medium supplemented by 2-4-D and BAP. The calluses were induced after fourteen days, and then put into the suspension culture after being four times subcultured. From the 8th to 14th day after establishment of the suspension culture, the amount of secondary metabolites was measured by HPLC, and gene expressions were analyzed with real-time PCR for three samples taken once a day at the same time from the suspension. It was found that the amount of secondary metabolites and expression of the corresponding genes (CsLYC, CsBCH and CsGT2) were saturated on the 12th day after establishment of the suspension. For the most effective stimulation of genes and biosynthesis of apocarotenoids, suitable elicitors and precursors should be added within 72 h before saturation of the synthesis rate which happens on the 12th day after establishment of the suspension.


Similar content being viewed by others
References
P. Winterhalter and M. Straubinger, Food Rev. Int., 16(1), 39 – 59 (2000).
R. Castillo, J. A. Fernandez, and L. Gomez-Gomez, Plant Physiol., 139(2), 674 – 689 (2005).
X. Wang, Z. Wang, J. Dong, et al., Genes Genet. Syst., 84(6), 397 – 405 (2009).
G. Britton, Overview of carotenoid biosynthesi. in: Carotenoids, Biosynthesis and Metabolism, G. Britton, S. Liaaen-Jensen, and H. Pfander (Eds.), Basel: Birkhäuser Verlag (1998), pp. 13 – 147.
H. Pfander and H. Schurtenberger, Phytochemistry, 21(5), 1039 – 1042 (1982).
S. A. Teale, F. X. Webster, and A. Zhang, New aggregation pheromone for the bark beetle Ips pini and uses thereof, US Patent 5167955 (1992).
F. Bouvier, C. Suire, J. Mutterer, et al., Plant Cell, 15(1), 47 – 62 (2003).
C. Dufresne, F. Cormier, and S. Dorion. Planta Med., 63(2), 150 – 153 (1997).
O. Ahrazem, A. Rubio Moraga, R. C. Lopez, et al., J. Exp. Bot., 61(1), 105 – 119 (2010).
J. I. Mir, N. Ahmed, A. H. Wafai, et al., Physiol. Mol. Biol. Plants, 18(4), 371 – 375 (2012).
A. R. Moraga, J. L. Rambla, O. Ahrazem, et al., Phytochemistry, 70(8), 1009 – 1016 (2009).
J. M. Pezzuto. Natural product cancer chemoprotective agents, in: Recent Advances in Phytochemistry: Phytochemistry of Medicinal Plants, J. T. Arnason, R. Mata, and J. T. Romeo (Eds.), New York: Plenum Press (1995), pp. 19 – 45.
H. Dornenburg and D. Knorr, Food Biotechnol., 10(1), 75 – 92 (1996).
A. W. Alfermann, and M. Petersen, Plant Cell Tissue Org. Cult., 43(2), 199 – 205 (1995).
G. A. Ravishankar, N. Bhyalakshmi, and S. Ramachandra Rao, Production of food additives, in: Biotechnology: Secondary Metabolites, K. G. Ramawat and J. M. Merillon (Eds.), New Delhi: Oxford IBH (1995), pp. 89 – 110.
K. J. Livak and T. D. Schmittgen, Methods, 25(4), 402 – 408 (2001).
H. Himeno and K. Sano. Agric. Biol. Chem., 51(9), 2395 – 2400 (1987).
W. M. Freeman, S. J. Walker, and K. E. Vrana, Biotechniques, 26(1), 112 – 122 (1999).
T. Taherkhani, R. Asghari Zakaria, M. Omidi, et al., Nat. Prod. Res., 33(4), 486 – 493 (2019).
T. Taherkhani, R. Asghari Zakariya, M. Omidi, et al., J. Genet., 13(1), 11 – 18 (2018).
Acknowledgments
The authors are thankful to the National Institute of Genetic Engineering and Biotechnology of Iran for kindly providing laboratory facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Taherkhani, T., Zakaria, R.A., Omidi, M. et al. Expression of Genes Involved in the Biosynthesis of Crocin and Safranal in Cell Suspension Culture of Saffron (Crocus sativus). Pharm Chem J 54, 1045–1050 (2021). https://doi.org/10.1007/s11094-021-02317-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-021-02317-z