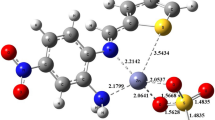
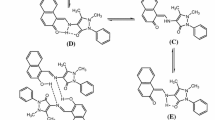
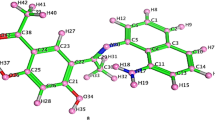
Amino acid derived Schiff base ligand [1-({[(Z)-(2-hydroxynaphthalen-1-yl)methylidene]-amino}methyl)-cyclohexyl]acetic acid (H-HMAC) and its metal complexes with composition [M(HMAC)2] [M = Co(II), Ni(II), Cu(II), and Zn(II)] reported previously were studied for their thermal degradation, antimicrobial and inhibitory activities against the enzymes like urease, α-chymotrypsin, acetylcholinesterase and butyrylcholinesterase. Only a zinc-based compound of the zwitter-ion ligand has been found to inhibit the urease activity with IC50= 0.04 ± 0.01 μ M (value ± SEM), which is 400 times better than the standard reference drug used. Metal coordination with zinc improved the bioactivity and deserved for selective urease inhibitor development. The inhibitory activity was structurally rationalized by carrying out the molecular modeling studies using AutoDock software. The same metal complex was also observed to inhibit the activity of Candida albicans. The thermal degradation studies suggest that the order of stability and activation energies of all compounds is as follows: Cu < H-HMAC < Ni < Co = Zn and E*Zn> E*Co> E*Ni> E*Cu, respectively.










Similar content being viewed by others
References
A. V. Bar, Rev. Recent Clin. Trials, 5(3), 174 – 178 (2010).
K.-Y. Ho, T. J. Gan, and A. S. Habib, Pain, 126(1 – 3), 91 – 101 (2006).
M. A. Rose and P. C. Kam, Anaesthesia, 57, 451 – 462 (2002).
P. J. Wiffen, H. J. McQuay, J. E. Edwards, and R. A. Moore, Cochrane Database Syst. Rev., CD005452 (2005).
M. Backonja, A. Beydoun, K. R. Edwards, et al., JAMA, 280, 1831 – 1836 (1998).
M. I. Bennett and K. H. Simpson, Palliat Med., 18, 5 – 11 (2004).
M. Rowbotham, N. Harden, B. Stacey, P. Bernstein, and L. Magnus-Miller, JAMA, 280, 1837 – 1842 (1998).
A. C.van de Vusse, S. G. Stomp-van den Berg, A. H. Kessels, and W. E. Weber, BMC Neurol., 4, 13 (2004).
Z.-Q. Liu and D. Wu, J. Phys. Org. Chem., 22, 308 – 312 (2009).
Y.-Z. Tang and Z.-Q. Liu, Cell Biochem. Funct., 26, 185 – 191 (2008).
Li Y.-F., Liu Z.-Q., Eur. J. Pharm. Sci., 44, 158 – 163 (2011).
M. Ikram, S.-U. Rehman, S. Rehman, et al., Inorg. Chim. Acta, 390, 210 – 216 (2012).
P. Datta, A. P. Mukhopadhyay, P. Manna, et al., J. Inorg. Biochem., 105, 577 – 588 (2011).
M. Ikram, S. Rehman, M. Ali, et al., Thermochim. Acta, 562C, 22 – 28 (2013).
M. Ikram, S. Rehman, M. Ali, et al., Thermochim. Acta, 555, 72 – 80 (2013).
P. G. Cozzi, Chem. Soc. Rev., 33, 410 – 421 (2004).
H. Adams, N. A. Bailey, I. S. Baird, et al., Inorg. Chim. Acta, 101, 7 – 12 (1985).
R. Atkins, G. Brewer, E. Kokot, et al., Inorg. Chem., 24, 127 – 134 (1985).
B. D. Clercq and F. Verpoort, Macromolecules, 35, 8943 – 8947 (2002).
T. Opstal and F. Verpoort, Angew. Chem. Int. Ed., 42, 2876 – 2879 (2003).
T. Opstal and F. Verpoort, Synlett, 6, 935 – 942 (2002).
S. N. Pal and S. Pal, Inorg. Chem., 40, 4807-4810 (2001).
B. D. Clercq and F. Verpoort, Adv. Synth. Catal., 34, 639 – 648 (2002).
B. D. Clercq, F. Lefebvre, and F. Verpoort, Appl. Catal. A 247, 345 – 364 (2003).
R. I. Kureshy, N. H. Khan, S. H. R. Abdi, et al., J. Mol. Catal. A: Chem., 150, 175 – 183 (1999).
M. Ikram, S. Rehman, A. Khan, et al., Inorg. Chim. Acta, 428, 117 – 126 (2015).
M. W. Weatherburn, Anal Chem., 39, 971 – 974 (1967).
I. Khan, S. Ali, S. Hameed, et al., Eur. J. Med. Chem., 45, 5200 – 5207 (2010).
R. J. P. Cannell, S. J. Kellam, A. M. Owsianka, and J. M. Walker, Planta Med., 54, 10 – 14 (1988).
G. L. Ellman, Arch. Biochem. Biophys., 74, 443 – 450 (1958).
O. Trott and A. J. Olson, J. Comput. Chem., 31, 455 – 461 (2010).
Discovery Studio Modeling Environment, Release 3.5, Accelrys Software Inc., San Diego (2012).
Atta-ur-Rahman, M. I. Choudhary, and W. J. Thomsen, Bioassay Techniques for Drug Development, Harwood Academic, Amsterdam, The Netherlands (2001).
H. H. Horowitz and G. Metzger, Anal. Chem., 35(10), 1464 – 1468 (1963).
M. Olszak-Humienik and J. Mozejko, Thermochim. Acta, 344, 73 – 79 (2000).
A. Coblentz, J. Am. Geriatr. Soc. 16(9), 1039 – 1046 (1968).
M. Arfan, M. Ali, H. Ahmad, et al., J. Enz. Inh. Med. Chem., 25, 296 – 299 (2010).
R. J. P. Cannell, S. J. Kellam, A. M. Owsianka, and J. M. Walker, Planta Med., 54, 10 – 14 (1988).
L. Zhang, S. B. Mulrooney, A. F. K. Leung, et al., BioMetals, 19, 503 – 511 (2006).
P. E.Wilcox, Chymotrypsinogens—Chymotrypsins, in: Methods in Enzymology, 19, 64 – 108 (1970).
F. A. Cotton, G. Wilkinson, and C. A. Murillo, Advanced Inorganic Chemistry, Wiley, New York (1999).
R. H. Holm, P. Kennepohl, and E. I. Solomon, Chem. Rev., 96, 2239 – 2314 (1996).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ikram, M., Rehman, S., Akhtar, M.N. et al. Selective Urease Inhibitory and Antimicrobial Activities of Transition Metal Complexes of Amino Acid Bearing Schiff Base Ligand: Thermal Degradation Behavior of Complexes. Pharm Chem J 54, 469–477 (2020). https://doi.org/10.1007/s11094-020-02224-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-020-02224-9