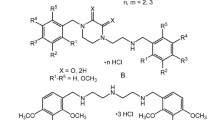
A new group of potential pFOX inhibitors among linear methoxyphenyltriazaalkanes were designed and synthesized. Cardiotropic activity in rodent experiments was found for most of the synthesized compounds. The most active compound N1-(2,3,4-trimethoxybenzyl)-N2-{2-[(2,3,4-trimethoxybenzyl)amino]ethyl}-1,2-ethanediamine trihydrochloride (LM-1) combined anti-ischemic, antiarrhythmic, and antifibrillatory activities (1 mg/kg, i.v.) and low toxicity (LD50 = 119 mg/kg, mice, i.p.) and was selected for development as a potential cardiotropic drug.



Similar content being viewed by others
References
L. M. McComick, A. C. Kydd, and D. P. Dutka, Cardiovasc. Hematol. Agents Med. Chem., 10(4), 319 – 324 (2012).
A. N. Grossman, L. H. Opie, J. R. Beshansky, et al., Circulation, 127(9), 1040 – 1048 (2013).
V. M. Olesova, O. Yu. Markatyuk, Yu. Yu. Yurova, and A. G. Obrezan, Kardiologiya, 53(1), 66 – 71 (2013).
G. D. Lopaschuk, J. R. Ussher, C. D Folmes, et al., Physiol. Rev., 90(1), 207 – 258 (2010).
V. Lionetti, W. C. Stanley, and F. A. Recchia, Cardiovasc. Res., 90(2), 202 – 209 (2011).
J. S. Jaswal, W. Keung, W. Wang, et al., Biochim. Biophys. Acta, 1813(7), 1333 – 1350 (2011).
J. S. Ingwall, Cardiovasc. Res., 81(3), 412 – 419 (2009).
M. van Bilsen, P. J. H. Smeets, A. J. Gilde, and G. J. van der Vusse, Cardiovasc. Res., 61(2), 218 – 226 (2004).
B. Bhandari and L. Subramanian, Recent Pat. Cardiovasc. Drug Discovery, 2(1), 35 – 39 (2007).
Y. H. Fang, L. Piao, Z. Hong, et al., J. Mol. Med. (Berlin), 90(1), 31 – 43 (2012).
P. F. Kantor, A. Lucien, R. Kozak, and G. D. Lopaschuk, Circ. Res., 86(5), 580 – 588 (2000).
M. Aldakkak, A. K. Camara, J. S. Heisner, et al., Pharmacol. Res., 64(4), 381 – 392 (2011).
K. M. Wyatt, C. Skene, K. Veitch, et al., Biochem. Pharmacol., 50(10), 1599 – 1606 (1995).
M. Imai, S. Rastogi, N. Sharma, et al., Cardiovasc. Drugs Ther., 21(1), 9 – 15 (2007).
P. Partownavid, S. Umar, J. Li, et al., Crit. Care Med., 40(8), 2431 – 2437 (2012).
G. Fragasso, A. Salerno, R. Spoladore, et al., Curr. Pharm. Des., 15(8), 857 – 862 (2009).
B. Hu, W. Li, T. Xu, et al., Clin. Cardiol., 34(6), 395 – 400 (2011).
L. Zhang, Y. Lu, H. Jiang, et al., J. Am. Coll. Cardiol., 59(10), 913 – 922 (2012).
E. Vizzardi, A. D’Aloia, F. Quinzani, et al., J. Cardiovasc. Pharmacol. Ther., 17(4), 353 – 356 (2012).
S. Sossalla and L. S. Maier, Pharmacol. Ther., 133(3), 311 – 323 (2012).
S. Yamamoto, K. Matsui, M. Sasabe, and N. Ohashi, J. Cardiovasc. Pharmacol., 39(2), 234 – 241 (2002).
A. N. Mironov, Handbook for Preclinical Drug Trials [in Russian], Part 1, Grif i K, Moscow, 2013, pp. 385 – 416.
I. A. Ilyushkina, A. N. Berchatova, I. A. D’yachenko, et al., Biomeditsina, No. 2, 6 – 13 (2012).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 53, No. 6, pp. 16 – 23, June, 2019.
Rights and permissions
About this article
Cite this article
Mokrov, G.V., Likhosherstov, A.M., Barchukov, V.V. et al. Synthesis and Cardiotropic Activity of Linear Methoxyphenyltriazaalkanes. Pharm Chem J 53, 500–506 (2019). https://doi.org/10.1007/s11094-019-02027-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-019-02027-7