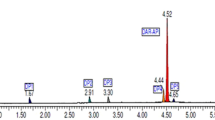
Stability study for dolutegravir bulk drug was performed and the degradation products formed were identified by chromatography (HPLC, LCMS) and spectroscopy (ESI-MS, FTIR, 1H and 13C NMR) techniques. Degradation of the drug in acidic and peroxide environment yielded one common major degradant f ((2,4-difluorophenyl) methanamine) with a mass peak at m/z = 182.44. In addition to this, five more minor degradation products (a, b, c, d and e) were formed. The structure interpretation showed that the drug degraded to its synthetic precursor and no extra structures were formed. Hence, it was suggested that drug dolutegravir should be kept away from acidic and oxygen rich conditions.









Similar content being viewed by others
References
British National Formulary: BNF 69 (69th Edition), British Medical Association (2015), 429.ISBN 9780857111562.
S. Kanters, M. Vitoria, M. Doherty, et al., Lancet HIV, 3, 510–520 (2016).
N. Mulligan, B. Best, E. Capparelli, et al., in: Conference on Retroviruses and Opportunistic Infections (2016, Boston, MA, USA), Abstract 438.
G. B. Bhavar, S. S. Pekamwar, K. B. Aher, et al., Sci. Pharm., 84(2), 305 – 320 (2016).
G. B. Bhavar, K. B. Aher, R. S. Thorat, et al., Malay. J. Anal. Sci., 19(6), 1156 – 1163 (2015).
N. M. Rao and D. G. Sankar, Future J. Pharm. Sci., 1(2), 73 – 77 (2015).
X. Wang, S. D. Penchala, A. Amara, et al., Ther. Drug Monitor., 38(3), 327 – 31 (2016).
ICH Q2 (R1), Validation of Analytical Procedures: Text and Methodology. International Conference on Harmonization, IFPMA, World Health Organization: Geneva (2009).
M. Nenad, Lovro Selic, and C. Anja, Patent WO2016113372A1 (2016).
Conflict of Interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, T.N.V.G., Vidyadhara, S., Narkhede, N.A. et al. Study of Dolutegravir Degradation and Spectroscopic Identification of Products by LCMS, 1H and 13C NMR Techniques. Pharm Chem J 53, 368–375 (2019). https://doi.org/10.1007/s11094-019-02007-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-019-02007-x