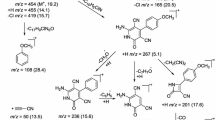
A method for the synthesis of 1-amino-3,3-dimethyl-3,4-dihydronaphthaline-2-carbonitrile (β-aminonitrile) was developed and used to synthesize Schiff bases, thioureide and chloracetamide derivatives. This latter was cyclized to 5,5-dimethyl-2-chloromethyl-5,6-dihydrobenzo[h]quinazoline-4(3H)-one, which was used to synthesize a series of 2-sulfanylmethyl and 2-aminomethyl derivatives. 2-Chloromethylbenzo[h]quinazoline in the presence of a base was found to form condensed heterocyclic compounds with seven rings: 1,1,11,11-tetramethyl-1,2,11,12-tetrahydropyrazino[2,1-b:5,4-b′]di(benzo[h]quinazoline)-10,16(8H,18H)-di one. The antitumor properties of the compounds synthesized here were studied in an ascites carcinoma model and the antibacterial properties were studied using the agar diffusion method.
Similar content being viewed by others
References
A. Wissner, M. Floyd, B. D. Johnson, et al., J. Med. Chem., 48(24), 7560 – 7581 (2005).
M. Kidwai, D. Bhatnagarom, R. Kumarom, et al., Chem. Pharm. Bul. Tokyo, 58(10), 1320 – 1323 (2010).
S. Eathiraj, R. Palma, M. Hirschi, et al., J. Biol. Chem., 286, 20677 – 20687 (2011).
I. Sovadinova, L. Blaha, J. Janošek, et al., Environmental Toxicol. Chem., 25(5), 1291 – 1297, No. 2006).
J. L. Liang, S. E. Parom, Y. Kwon, et al., Bioorg. Med. Chem., 20(16), 4962 – 4967 (2012).
P. Selvam, N. Murugesh, M. Chandramohan, et al., Ind. J. Pharm Sci., 72(6), 806 – 809 (2010).
C. Brullo, M. Rocca, P. Fossa, E. Cichero, et al., Bioorg. Med. Chem. Let., 22, 1125 – 1129 (2012).
R. Gupta and R. P. Chaudhary, Phosphorus, Sulfur Silicon, 187(6), 735 – 742 (2012).
N. Chidananda, B. Poojary, V. Sumangala, et al., Ind. J. Het. Chem., 20, 337 – 342 (2011).
N. Sati, S. Kumar, M. S. M. Rawat, et al., Ind. J. Pharm. Sci., 71(5), 572 – 575, No. 2009).
A.-G. E. Amr, H. H. Sayed, M. M. Abdulla, et al., Arch. Pharmazie, 338(9), 433 – 440, No. 2005).
H. Ohtomo. T. Tagata, K. Sasaki, et al., Tetrahedron, 63(51), 12541 – 12546 (2007).
H. K. Maurya, R. Vema, S. Alam, et al., Bioorg. Med. Chem. Lett., 23(21), 5844 – 5849 (2013).
A. I. Markosyan, S. A. Gabrielyan, G. A. Panosyan, et al., Khim.-Farm. Zh., 42(2), 6 – 9 (2008).
A. I. Markosyan, S. V. Dilanyan, R. S. Sukasyan, et al., Khim.-Farm. Zh., 42(3), 16 – 19 (2008).
A. I. Markosyan, S. V. Dilanyan, F. G. Arsenyan, et al., Khim.-Farm. Zh., 44(3), 3 – 5 (2010).
A. I. Markosyan, S. V. Dilanyan, S. A. Gabrielyan, et al., Khim.-Farm. Zh., 44(7), 14 – 16 (2010).
A. I. Markosyan, S. A. Gabrielyan, F. G. Arsenyan, et al., Khim.-Farm. Zh., 44(8), 3 – 6 (2010).
A. I. Markosyan, S. A. Gabrielyan, F. G. Arsenyan, et al., Khim.-Farm. Zh., 44(8), 7 – 10 (2010).
A. I. Markosyan, S. A. Gabrielyan, F. G. Arsenyan, et al., Khim.-Farm. Zh., 48(12), 15 – 18 (2014).
Guidelines for Preclinical Studies of Medicines [in Russian], Moscow (2012), pp. 509 – 524.
Z. P. Sof’ina, A. B. Syrkin, A. Goldin, et al., Experimental Assessment of Antitumor Substances in the USSR and USA [in Russian], Meditsina, Moscow (1980).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 53, No. 1, pp. 17 – 23, January, 2019.
Rights and permissions
About this article
Cite this article
Markosyan, A.I., Airapetyan, K.K., Gabrielyan, S.A. et al. Synthesis and Antitumor and Antibacterial Activity of Novel Dihydronaphthaline and Dihydrobenzo[H]Quinazoline Derivatives. Pharm Chem J 53, 15–22 (2019). https://doi.org/10.1007/s11094-019-01948-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-019-01948-7