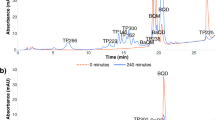
Considering the pharmacological activities of 5-benzyl-1,3,4-oxadiazole-2-thiol (OXPA), the present study aimed to develop and validate a UV-spectroscopic method according to the ICH guidelines to be used to study degradation profile of the compound. The method was developed at 263 nm, the wavelength giving maximum absorbance. Linearity of the method was evaluated by analyzing a series of standard solutions. Then, recovery, accuracy and sensitivity were determined. The compound was subjected to a number of stress conditions to determine it in the presence of degradants (specificity). The proposed method showed linearity in a concentration range (0.33 - 40.0 μg/mL) with correlation coefficient (R2) = 0.998. The recovery was within 100.63 – 102.46%, intraday and inter-day accuracy were found to be 99.10 – 102.46% with relative standard deviation less than 2%, and the LOD and LOQ were found to be 0.099 and 0.330 μg/mL, respectively. The OXPA was degraded with the passage of time under acidic conditions and oxidized completely in 35% H2O2, whereas the degradation was 58.47% in 3% H2O2. Dry heat (80°C) exposure for 48 h resulted in 1.09% degradation, whereas UV exposure resulted in complete loss of the compound. These results indicate that the method is simple, sensitive, accurate and specific, hence may be used to carry out quantitative work on OXPA.










Similar content being viewed by others
References
J. Bostrom, A. Hogner, A. Llinas and E. Wellner et al, J. Med. Chem., 55, 1817 (2012).
K. C. Nagaraj, Chaluvaraju, M. S. Niranjan, and S. Kiran, Int. J. Pharm. Pharm. Sci., 3, 9 (2011).
J. Sun, J. A. Makawana, and H. L. Zhu, Bioorg & Med. Chemistry., 19, 4895–4902 (2011).
S. Kavitha, S. Gnanavel and K. Kannan, Asian. J. Pharm. Clin. Res., 7, 11 (2014).
J. J. Piala and H. L. Yale, US Patent 3141022, Chem. Abstr., 61, 8317 (1964).
A. Almasirad, S. A. Tabatabai, M. Faizi et al, Acta Chim. Slov., 54 , 317 – 324 (2007).
I. Angilini, L. Angilini and F. Sparac, British Patent, 196, 91:161, 801. Chem. Abstr., 71, 112936 (1969).
D. A. Kennedy and L. A. Summers, J. Heterocyclic Chem., 18(2), 409 – 410 (1981).
H. L. Yale and K. Losee, J. Med. Chem., 9(4), 478 – 483 (1966).
A. J. Vlietinck and V. J. Ram, J. Heterocyclic Chem., 25(9), 253 – 259 (1988).
S. Bahadur and K. K. Pandey, J. Indian Chem. Soc., 57, 447(1980).
S. A. F. Rostom, M. A. Shalaby and M. A. EL-Demellawy, Eur. J. Med. Chem., 38(11 – 12), 959 – 974 (2003).
D. H. Boschelli, D. T. Connor, D. A. Bornemeier, et al, J.Med. Chem., 36, 1802 (1993).
A. Cansýz, M. Koparýr, and A. Demirdag, Molecules., 9(4), 204 – 212 (2004).
M. Koparir, A. Çetin, and A. Cansýz, Molecules., 10(2), 475 – 480 (2005).
S. N. Hemavathi, B. K. Vishukumar, and R. K. M. Lokanatha, Int. J. Pharm. Pharm Sci., 3(4), 110 – 114 (2011).
S. Kumar, Turk. J. Chem., 35, 99 2011).
A. Rehman, S. Z. Siddiqui, M. A. Abbasi, et al, Int. J.Pharm. Pharm. Sci., 4(2), 676 (2012).
ICH Q2 B, International Conference on Harmonization (ICH), Validation of Analytical Procedures: Methodology, IFPMA, Geneva (1997).
T. Hussain, M. K. Shahzad, K. Hayat, et al., Pharm. Chem. J., 50(5), 289 – 295 (2016).
ICH Q1 A (R2), International Conference on Harmonization (ICH). Stability Testing of New Drug Substances and Products, IFPMA, Geneva (2003).
ICH Guidance for Industry, Q1B, Photo stability Testing of New drug substances and products, IFPMA, Geneva (2006).
D. W. Reynolds, K. L. Facchine, J. F. Mullaney et al, Pharm. Technol., 2, 48 – 56 (2002).
S. W. Baertschi, Pharmaceutical Stress Testing, Predicting Drug Degradation, Taylor & Francis Group, Boca Raton (2005), pp. 13.
E. P. Nesynov and A. P. Grekov, Russ. Chem. Rev., 33(10), 508 (1964).
M. S. Charde, J. Kumar, A. S. Welankiwar and R. D. Chakole. Int. J. Adv. Pharm.., 3(4), 2 (2013).
G. Ngwa, Drug. Deliv. Technol., 10(15), 1 – 4 (2010).
A. Hetzheim and K. Mockel, Adv. Heterocyclic Chem., 7, 183 – 224 (1967).
H. Khalilullah, M. J. Ahsan, Md. Hedaitullah, et al., Mini-Rev. Med. Chem., 12, 789 – 801 (2012).
J. A. Claisse, M. W. Foxton, G. Gregory, et al., J. Chem. Soc. Perkin Trans., 45, 2241 – 2249 (1973).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qamar, S., Hussain, K., Bukhari, N.I. et al. Method Development and Stress Degradation Profile of 5-Benzyl-1,3,4-Oxadiazole-2-Thiol Studied by UV Spectroscopy. Pharm Chem J 52, 278–283 (2018). https://doi.org/10.1007/s11094-018-1806-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-018-1806-5