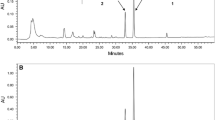
Chaga can be extracted with EtOAc to afford ~0.88% extracted substances, of which ~58% are lipophilic compounds with the remainder being phenolic compounds. Tetracyclic triterpenes, sterols and their esters, sesquiterpenes (azulenes), bicyclic monoterpenes (iridoids), phenolcarboxylic acids, simple phenols, and flavonoids were identified in the extract. Stilbene-type compounds, a phenanthrene derivative, and 9(11)-dihydroergosteryl benzoate were observed in chaga for the first time.

Similar content being viewed by others
References
M. A. Sysoeva, V. R. Khabibrakhmanova, G. I. Kyyamova, et al., Khim. Rastit. Syr’ya, No. 4, 117 – 122 (2009).
M. A. Sysoeva, V. R. Khabibrakhmanova, V. S. Gamayurova, et al., Khim. Rastit. Syr’ya, No. 1, 111 – 114 (2008).
A. N. Shivrina, Biosynthetic Products of Higher Fungi and Their Use [in Russian], Leningrad, Moscow (1966).
State Pharmacopoeia of the USSR, No. 2, Meditsina, Moscow (1990).
State Pharmacopoeia of the USSR, No. 2, Meditsina, Moscow (1987).
M. N. Zaprometov, Principles of the Biochemistry of Phenolic Compounds [in Russian], Vysshaya Shkola, Moscow (1974).
E. V. Beketov, A. A. Abramov, O. V. Nesterov, et al., Vestn. Mosk. Univ., Ser. 2, 46(4), 259 – 262 (2005).
A. A. Lobanova, V. V. Budaeva, and G. V. Sakovich, Khim. Rastit. Syr’ya, No. 1, 47 – 52 (2004).
E. N. Zhukovich, M. Yu. Semenova, L. A. Sharikova, et al., Khim.-farm. Zh., 44(3), 35 – 37 (2010); Pharm. Chem. J., 44(3), 144 – 146 (2010).
L. V. Antipov and N. N. Bezryadin, Physical Methods for Control of Meat-Industry Raw Materials and Products [in Russian], GI-ORD, St. Petersburg (2006).
M. A. Sysoeva, V. R. Khabibrakhmanova, V. S. Gamayurova, et al., Khim. Rastit. Syr’ya, No. 3, 119 – 122 (2008).
M. Kates, Techniques of Lipidology: Isolation, Analysis, and Identification of Lipids, Elsevier, New York (1972), 610 pp.
E. Stahl (ed.), Thin-Layer Chromatography: A Laboratory Handbook [translated from the German], Academic, New York (1965), 553 pp.
S. E. Severin and G. A. Solov’ev, Practicum for Biochemistry [in Russian], Izd. MGU, Moscow (1989).
M. M. Anisimova, V. A. Kurkin, and V. N. Ezhkov, Izv. Samar. Nauchn. Tsentra RAN, 1(8), 2011 – 2014 (2010).
G. L. Ryzhova, S. S. Kravtsova, and S. A. Matasova, Khim.-farm. Zh., 31(10), 44 – 47 (1997); Pharm. Chem. J., 31(10), 551 – 554 (1997).
M. A. Burmasova and M. A. Sysoeva, Khim. Rastit. Syr’ya, No. 1, 149 – 152 (2012).
W. Mazurkiewicz, Acta Pol. Pharm., No. 67, 397 – 406 (2010).
W.-F. Zheng, Mycosystema, No. 27, 575 – 581 (2008).
Y. J. Kim, J. Korean Soc. Appl. Biol. Chem., No. 54, 287 – 294 (2011).
L. R. Ivanova, L. I. Butenko, and L. V. Ligai, Khim. Rastit. Syr’ya, No. 4, 131 – 133 (2010).
A. N. Shivrina and E. V. Lovyagina, Feed Proteins and Physiologically Active Substances for Animal Husbandry [in Russian], Moscow-Leningrad (1965), pp. 59 – 64.
R.-S. Ludwiczak and U. Wrecino, Rocz. Chem., No. 36, 497 – 502 (1962).
E. V. Lovyagina and A. N. Shivrina, Biokhimiya, No. 5, 794 – 800 (1962).
A. Blazhei and L. Shutyi, Phenolic Compounds of Plant Origin [in Russian], Mir, Moscow (1977).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 51, No. 12, pp. 21 – 23, December, 2017.
Rights and permissions
About this article
Cite this article
Kyyamova, G.I., Khabibrakhmanova, V.R. & Sysoeva, M.A. Hydrophobic Constituents Extracted from Chaga by Ethylacetate. Pharm Chem J 51, 1085–1087 (2018). https://doi.org/10.1007/s11094-018-1745-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-018-1745-1



